3-Chloro-2-methylpropene reacts with sodium methoxide in methanol to form 3-methoxy-2-methylpropene. For each of the following changes in the reaction conditions, state whether the reaction rate would increase, decrease, or remain the same. Explain your reasoning. In some cases the identity of the major organic product would be expected to change; in such cases, give the expected major product. (a) dimethyl sulfoxide (DMSO) is used in place of methanol (b) methanol is used by itself without sodium methoxide (c) 1-chloro-2,2,-dimethylpropane is used in place of 3-chloro-2-methylpropene
3-Chloro-2-methylpropene reacts with sodium methoxide in methanol to form 3-methoxy-2-methylpropene. For each of the following changes in the reaction conditions, state whether the reaction rate would increase, decrease, or remain the same. Explain your reasoning. In some cases the identity of the major organic product would be expected to change; in such cases, give the expected major product. (a) dimethyl sulfoxide (DMSO) is used in place of methanol (b) methanol is used by itself without sodium methoxide (c) 1-chloro-2,2,-dimethylpropane is used in place of 3-chloro-2-methylpropene
Macroscale and Microscale Organic Experiments
7th Edition
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Kenneth L. Williamson, Katherine M. Masters
Chapter17: Nucleophilic Substitution Reactions Of Alkyl Halides
Section: Chapter Questions
Problem 3Q
Related questions
Question
3-Chloro-2-methylpropene reacts with sodium methoxide in methanol to form 3-methoxy-2-methylpropene.
For each of the following changes in the reaction conditions, state whether the reaction rate would increase, decrease, or remain the same. Explain your reasoning. In some cases the identity of the major organic product would be expected to change; in such cases, give the expected major product.
(a) dimethyl sulfoxide (DMSO) is used in place of methanol
(b) methanol is used by itself without sodium methoxide
(c) 1-chloro-2,2,-dimethylpropane is used in place of 3-chloro-2-methylpropene
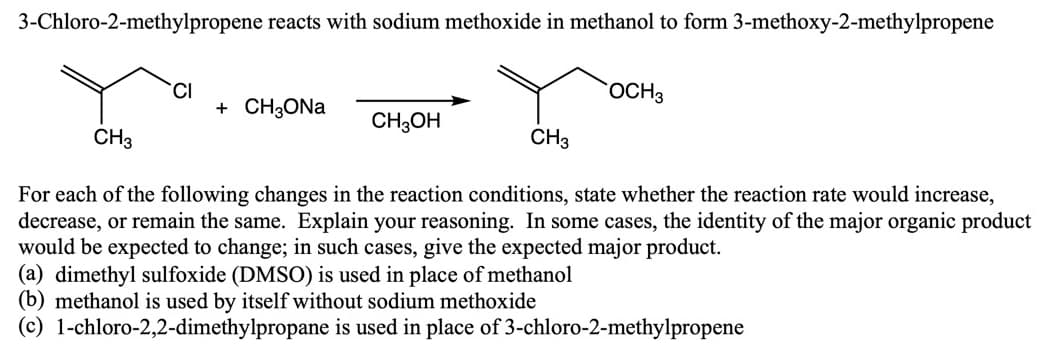
Transcribed Image Text:3-Chloro-2-methylpropene reacts with sodium methoxide in methanol to form 3-methoxy-2-methylpropene
CI
+ CH3ONA
OCH3
CH3OH
CH3
CH3
For each of the following changes in the reaction conditions, state whether the reaction rate would increase,
decrease, or remain the same. Explain your reasoning. In some cases, the identity of the major organic product
would be expected to change; in such cases, give the expected major product.
(a) dimethyl sulfoxide (DMSO) is used in place of methanol
(b) methanol is used by itself without sodium methoxide
(c) 1-chloro-2,2-dimethylpropane is used in place of 3-chloro-2-methylpropene
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning