3) "For one smalt bass which could have been bought at any trustworthy market for sixty-five cents, or, possibly, some poor little kitten-fish-offspring of a catfish{-whose mother's milk is not yet dry upon its lips." The author's diction in the sentence makes the audience A) feel sorry for the catfish B) feel worried for the smat[ bas. Cheofeel excitement for the fisherman. "Dr feel apathy for tne mother catfish.
3) "For one smalt bass which could have been bought at any trustworthy market for sixty-five cents, or, possibly, some poor little kitten-fish-offspring of a catfish{-whose mother's milk is not yet dry upon its lips." The author's diction in the sentence makes the audience A) feel sorry for the catfish B) feel worried for the smat[ bas. Cheofeel excitement for the fisherman. "Dr feel apathy for tne mother catfish.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 80AP
Related questions
Question
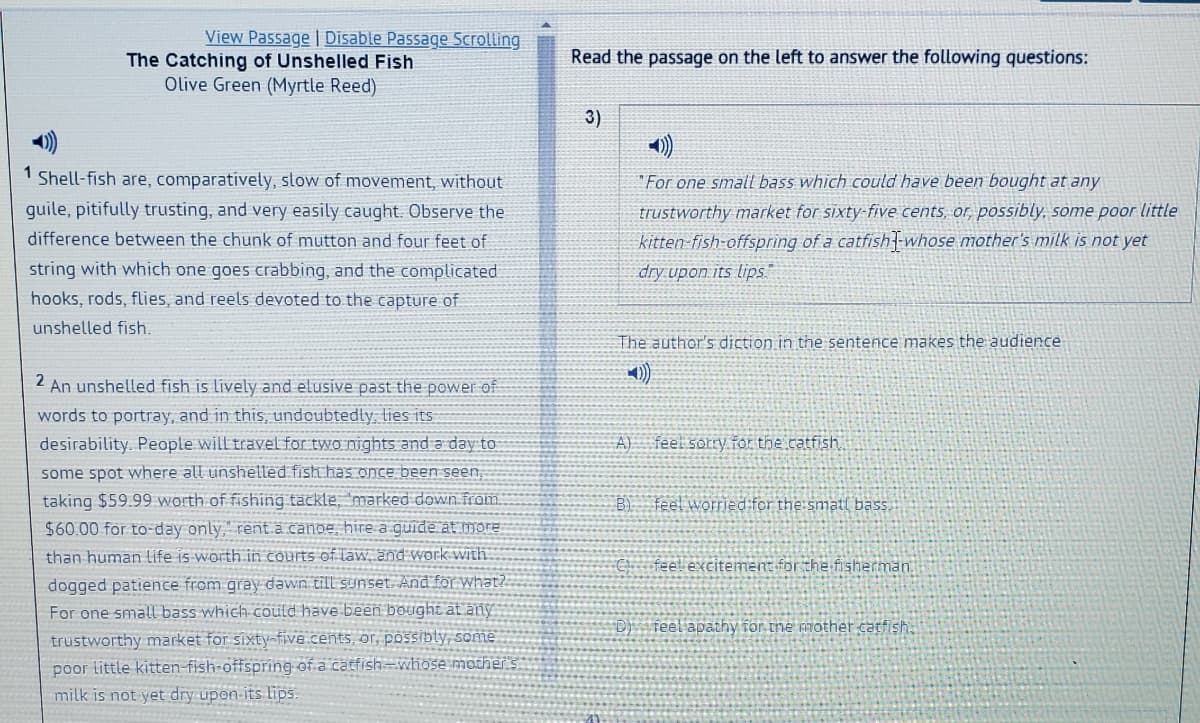
Transcribed Image Text:View Passage | Disable Passage Scrolling
Read the passage on the left to answer the following questions:
The Catching of Unshelled Fish
Olive Green (Myrtle Reed)
3)
1 Shell-fish are, comparatively, slow of movement, without
"For one small bass which could have been bought at any
guile, pitifully trusting, and very easily caught. Observe the
trustworthy market for sixty-five cents, or, possibly, some poor little
kitten-fish-offspring of a catfish--whose mother's milk is not yet
difference between the chunk of mutton and four feet of
string with which one goes crabbing, and the complicated
dry upon its lips."
hooks, rods, flies, and reels devoted to the capture of
unshelled fish.
The author's diction in the sentence makes the audience
))
2
An unshelled fish is lively and elusive past the power of
words to portray, and in this, undoubtedty, lies its
desirability. People will travel for two nights and a day to
A)
feel sorry for the catfish
some spot where all unshelled fish has once been seen,
taking $59.99 worth of fishing tackle, 'marked down from
B) feel worried for the smatl bass.
$60.00 for to-day only," rent.a.canoe, hire a guide at more
than human life is worth in courts of läw, and work With
C feel excitement for the fisherman.
dogged patience from gray dawn till sunset. And for what?.
For one small bass which could have been bought at anỹ
D) feel apathy for the mother catfish,
trustworthy market for sixty-five cents, or, pOSSibly, some
poor little kitten-fish-offspring of a catfish=whose mother s
milk is not yet dry upon-its lips.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning