Q: Macmillan Learning A fossil is found to have a "Clevel of 63.0% compared to living organisms. How…
A:
Q: A 3.88 gram sample of C6H2103 contains how many O atoms? Use 3 significant figures and scientific…
A: We have to find the number of oxygen atom in 3.88 gram of C6H21O3.
Q: Using the information from the table below, what should be the unit for k? Total reaction order 0 1…
A:
Q: For the simplest serial first order reactions A→ R→S; the concentration of component A -increases…
A: A question based on reaction rates. 4 options for the general serial first order reaction is given…
Q: The freezing point of benzene is 5.5 °C. What is the molar mass of a nonlonizing solute if 1.394…
A: Freezing point of benzene (Tf(benzene)) = 5.5°CFreezing point of solution (Tf(solution)) =…
Q: The unknown reaction: A -> P has been measured at the following temperatures: Temp = 298 K Initial…
A: Order of reaction cannot determined theoretically, its a experimental measurement. From the given…
Q: Which of the following is/meso compounds? 애 애 ОН 어 ОН ОН || ОН ОН ОН ОН 4 ОН ОН |||
A: Meso compound is stereoisomer which has either plane of symmetry or center of symmetry present due…
Q: Provide the correct IUPAC name for the following compounds and state the function group. Image…
A: It is based on the concept of nomenclature Here we are required to predict the IUPAC name of the…
Q: The volume of the HCI solution is 80.5 mL and the volume of the 0.10 M NaOH titrant at the…
A: Volume of the HCI solution = 80.5 mL volume of the 0.10 M NaOH titrant at the equivalence point =…
Q: 1) Classify the unsaturated. i) Classify the 1º, 2º, 3º. State a your molecule as being saturated on…
A: Unsaturated carbons are those which is connected to other carbons by a double bond or triple bond…
Q: Radioactive Uranium 325U will decay in the following sequence: First an a decay, next a B-decay,…
A: The given types of radioactive decays are discussed below decay: It is a type of radioactive decay,…
Q: Describe the factors that influence the rate of a chemical reaction and explain their effects using…
A: Introduction:The rate of a chemical reaction is influenced by several factors, including reactant…
Q: In the context of chemical reactions, explain the concept of kinetic versus thermodynamic control…
A: The concept of kinetic versus thermodynamic control in chemical reactions explores the factors that…
Q: H— Н H Н Н. Н (11) H H
A: The ideal bond angel of a organic molecules can be easily determined by counting the total number of…
Q: Ammonia is synthesized from nitrogen gas and hydrogen gas at 400 degrees Celsius and 350 kPa. If…
A: Nitrogen reacts with hydrogen to form ammonia. The equation for the balanced chemical reaction is…
Q: C1. Subject :- Chemistry
A: The final quantity of reaction's species for any reversible reaction is computed using equilibrium…
Q: N₂O4(g) 2NO₂(g) K = 6.14 x 10-3 @ 298 K; "X" = 0.186 The sealed reaction vessel was charged with…
A: The equilibrium reaction is as follows:The initial concentration of = 2.50 MThe initial…
Q: Arrange the compounds below in order of increasing boiling point. 2-methylheptane, octane and…
A: 1. Inter molecular force of attraction : in alkanes boiling point depends upon london forces . if…
Q: Question 6 2 Name 3 4 Periplanone B How many chirality centers are in this molecule? H₂C Structure…
A:
Q: 12.44 Classify each of the following alcohols as primary (1°), secondary (2°), or tertiary (3°):…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: Using just a periodic table (not a table of electronegativities), decide which of these is likely to…
A: Answer:-This question is answered by using the simple concept of polarity of the covalent bond.
Q: Question 16 CH3 H..!! C Br CH₂OH Determine if the stereocenter(s) are R- or S- O The stereocenter is…
A: To determine the stereochemistry, we can check the following -Whether the lowest priority group is…
Q: K,, for lead (II) chloride, PbCi,, is 1.6 x 10. Calculate the molar solubility of PbCI, a) b) in…
A:
Q: How many core and valence electrons does one atom of iodine (1) have? 7 core and 36 valence 46 core…
A: Outermost shell containing electrons are known as valence electrons and except valence electrons…
Q: Which of these molecules can hydrogen bond with water? 0. www O O NH₂ Br Br N Br
A: Hydrogen bonding is a type of intermolecular attraction that occurs between a hydrogen atom bonded…
Q: a. How many grams of iron(III) nitrite, Fe(NO2)3, are present in 1.82 moles of this compound? grams…
A:
Q: What type (redox, acid-base, precipitation) are the reactions below? For the redox reactions,…
A: A) FeCl3 + Cu ----->FeCl2 + CuClThis reaction is displacement reaction and also redox reactionB)…
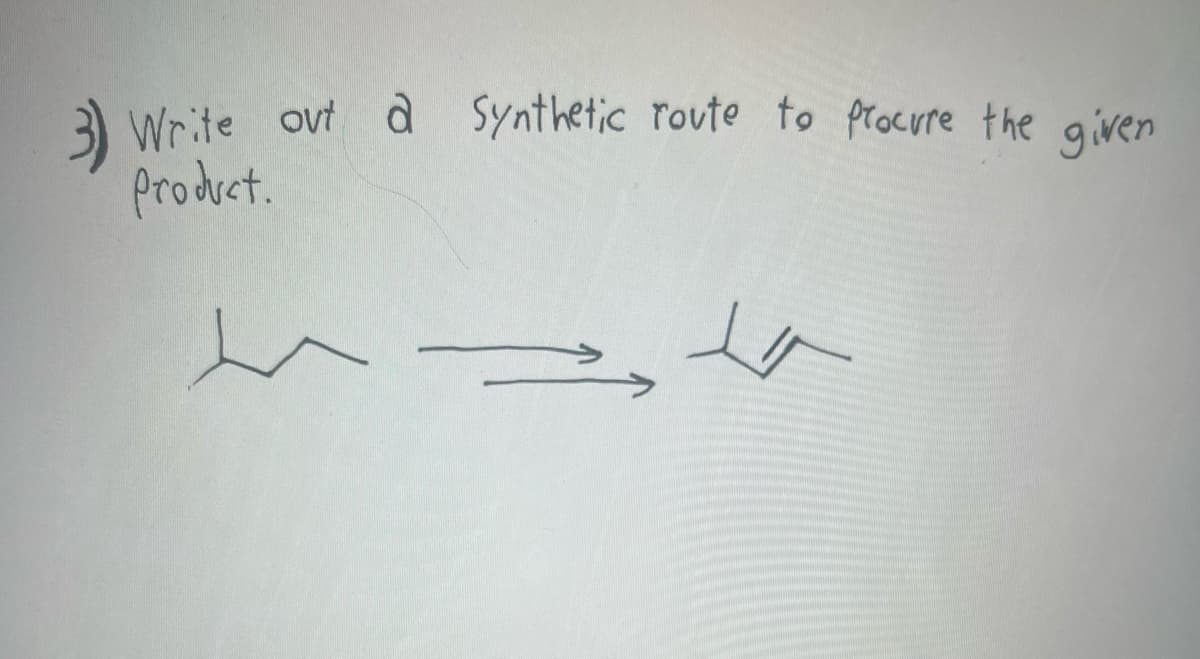
Q: Current Attempt in Progress Suggest an efficient synthesis for the following transformation: 4 The…
A: Alkylhalide after elimination gives alkeneAlkene reacts with O3 followed by DMS to produce two…
Q: The active ingredient of Benadryl© Chesty Forte Cough Liquid is Guaiphenesin an expectorant drug…
A: Volume of Benadryl Chesty Forte cough syrup = 200 mLAmount of Guaiphenesin = 4 g
Q: Consider the electron configuration for fluorine (F) and the electron configuration for chlorine…
A: Electron configuration of fluorine (F) is,1s2 2s2 2p5Electron configuration of chlorine (Cl) is,1s2…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product.…
A: This is an example of basic hydrolysis of ester
Q: NH₂
A: 1). First step is friedal craft alkylation2). Nitration reaction with the help of HNO3/H2SO43). Then…
Q: 1. Which of the following compounds contains an ether functional group? a. Ethanol b. Acetic acid c.…
A: Functional group-The atom or group of atoms show characteristics properties of organic…
Q: Which of the following 3 point molecules has the greatest hyperconjugation effect? с een A Br B Br…
A: Carbocation stability: Hyperconjugation is a key factor in stabilizing reactive intermediates in the…
Q: What is the net charge of the tetrapeptide C-R-Y-K at pH 11.5
A: We must take into account the pKa values of the relevant amino acids in order to calculate the net…
Q: Convert the following chair cyclohexane structure into a valid flat line-bond drawing: Brcl H3CO Br"…
A:
Q: Structure A Br H3C...C H CH₂CH3 Structure B CH 3 Hmo Br CH₂CH3 Determine the relationship between…
A:
Q: Question 1 Isomer A B CH 3 OSI(CH3)3 Which isomer is chiral? Isomer B CH3 OSI(CH3)3
A: A compound is said to be chiral if it has a chiral carbon which has four different groups attached…
Q: Calculate the molality of a solution produced by dissolving 15.2 g of calcium chloride in 345 g of…
A: Definition of Molality: It is defined as number of moles of solute dissolve in 1 kg of solvent .…
Q: The following set up is arranged in a laboratory. Vessel 1 Vessel 2 Assuming the volume of the…
A: At constant temperature, the volume of a given amount of gas is inversely proportional to the…
Q: With detailed mechanisms and explanations please
A:
Q: The natural abundance of the three stable isotopes are 90.92% isotope A (19.9924 amu), 0.3104%…
A:
Q: Propose an efficient synthesis for the following transformation: o The transformation above can be…
A: This question is answered by using the simple concept of chemical reactions of organic compounds…
Q: 3. C,H,O, is infinitely miscible (soluble) in water. Ethylene glycol is a nonelectrolyte that is…
A: The depression in the freezing point () is given by the formula, i is the van't hoff factorm is…
Q: What is the best explanation for why metals are ductile? Valence electrons carry charge anywhere…
A: Ductility is the property of a material that allows it to be stretched or drawn out into a wire or…
Q: Draw the product formed when the structure shown below undergoes a reaction with diluted water…
A: This is reaction of alkene with diluted H2SO4 .This is addition reaction of alkene.In this reaction,…
Q: Determine the product(s) formed in the following reactions. A. B. OH -C=CH-CH₂ OCH3 -C=C-CH3 CH3OH…
A: A) Alkyne reareact with H2O/H2SO4 and gives Ketone moiety. Now the ketone will…
Q: Fe203 + 3 CO → 2 Fe + 3 CO2 If the reaction uses 55.5 liters of CO gas, how many molecules is this?
A:
Q: Classify each aqueous solution as acidic, basic, or neutral at 25 °C. Acidic pH=3.57 [H]-3.8 x 10"…
A: For acedic solutionspH < 7 [H+] > 1.0 x 10-7 and [OH-] < 1.0 x 10-7For basic solutionspH…
Q: O LOH H₂SO4, A & 1) 2 equiv 2) H₂O MgBr
A: In a Grignard reagent, an alkyl group is attached to the electropositive metal, magnesium, which…
Step by step
Solved in 3 steps with 1 images

- Perform a retrosynthetic analysis and suggest a synthesis of the target molecule (on the left) from the given starting material (on the right). (any reagents maybe usedProvide the retrosynthetic analysis and synthetic route for the following Compound.In synthesis of benzoic acid with Grignard reagent, benzene is often detected as an impurity. How is benzene formed?
- Provide reagents and conditions for the following synthetic tranformation. (Please don't provide handwritten solution)Supply the synthetic routes to the compounds show using starting material with six or fewer carbon atoms. Usage of triphenylphosphine as a reagent may be needed.Suggest a reasonable mechanism for the following reactions. (Use arrows to show movement of electrons)
- The analgesic acetaminophen is synthesized by treating 4-aminophenol with one equivalent of acetic anhydride. Complete the equation for the formation of acetaminophen. a. Acetaminophen: b. Additional Product pls add the name for both, thanks!Write a mechanism for the following reaction:Propose a reasonable synthetic route.

