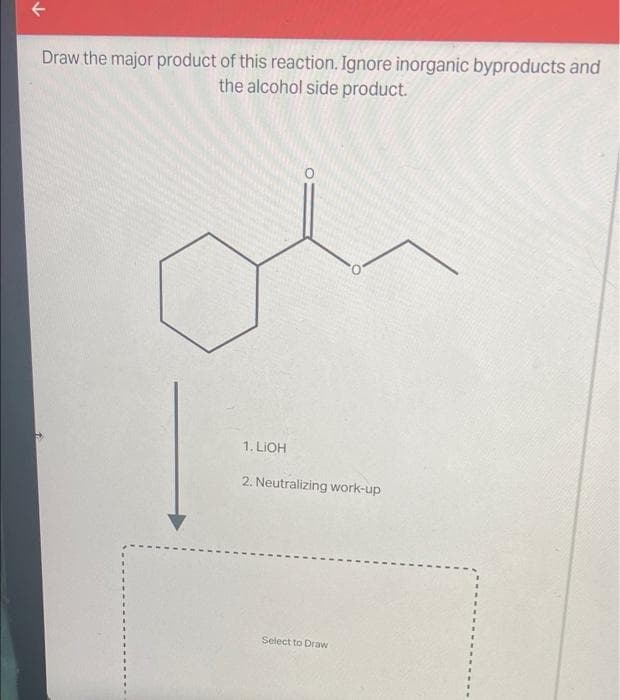
Draw the major product of this reaction. Ignore inorganic byproducts and the alcohol side product. 1. LIOH 2. Neutralizing work-up Select to Draw
Q: For the simplest serial first order reactions A→ R→S; the concentration of component A -increases…
A: A first-order reaction is one in which the rate of the…
Q: What is true considering the bonding of atoms in clusters? O When atoms are bound in small clusters…
A: The correct answer is 1) When atoms are bound in small clusters (i.e., nanoparticles), their…
Q: H₂N-CH- answer: CH₂ OH 1 H____CH-C CH₂ OH 2 CHIC- CH₂ CH-CH CH3 3 -OH
A: A protein is formed through the peptide bond. These peptide bonds are formed by the amino acids. The…
Q: A sample of gas has a volume of 597 ml at a pressure of 4.19 atm. The gas is allowed to expand and…
A:
Q: Draw a Lewis structure for HC C-CHO. Draw the Lewis dot structure for the given compound. Include…
A: propiolaldehyde Chemical Formula: C3H2O Exact Mass: 54.01 Molecular Weight: 54.05 m/z: 54.01…
Q: a. Choose the complete Lewis structure for the following partially completed Lewis structure. F…
A: Lewis structure is a way of representing the structure of a molecule in which bond pairs (shared…
Q: 1. The region of electromagnetic spectrum for nuclear magnetic resonance is: a) Microwave b) UV-rays…
A: EMR - ELECTROMAGNETIC RADIATIONS Type of EMR Region of spectrumMicrowave -…
Q: If you start with 5 grams of lithium chloride (LiCl = 42.4 grams/mol), how many grams of calcium…
A: Number of moles is calculated by dividing given mass with molecular mass. In the given question it…
Q: Using the kinetic molecular theory, explain why the pressure of a gas decreases as the temperature…
A: According to the kinetic molecular theory, gases consist of particles (atoms or molecules) that are…
Q: The balanced chemical equation for the reaction between iodine vapour and fluorine gas is: 3I2(g) +…
A:
Q: The first step in the reaction of Alka-Seltzer with stomach acid consists of one mole of sodium…
A:
Q: 3. Write oxidation and reduction half reactions and the net ionic equations for: a. copper(II)…
A: A redox reaction is defined as the reaction which shows oxidation and reduction reaction…
Q: 5. Calculate the concentration of the hydrochloric acid if a 20.00 mL sample is titrated with…
A:
Q: For n=4, what are the possible sublevels? a. 1,2,3,4 b. 0,1 c. 0,1,2 d. 0,1,2,3 e. 0
A:
Q: How much time does it take to reduce the concentration of NO2 by half by looking at the reaction…
A: Initial NO2 concentration = 0.05 MTime taken to reduce concentration to half = ?
Q: 6.8 Consider the displacements of the atoms in the trigonal bipyramidal molecule PF5. Calculate the…
A: Vibrational modes of molecule define the shapes that a structure will vibrate in when excited. There…
Q: Calculate the solubility at 25 °C of Zn(OH)₂ in pure water and in a 0.0120M ZnSO4 solution. You'll…
A:
Q: Which of the following atoms and ions is (are) isoelectronic with C1³+: Ne, Ca2+, Ar, Si, Si4+,…
A: All the isoelectronic species have the same number of electrons.
Q: The picture below is a representation of a set of: O 3 0 04 335 O 5 1. sp orbitals 2. sp² orbitals…
A: An orbital is a three dimensional description of the most likely location of an electron around an…
Q: What is the name of the compound with the formula Cu₂O ? What is the name of the compound with the…
A: The inorganic compounds do not form by the combination of C and H only. There may be other atoms,…
Q: Obtain cyclopentanamine from O-(cyclopentylmethyl)hydroxylamine. Comment the reaction.
A: The structure of -(cyclopentylmethyl)hydroxylamine is as follows:
Q: 2. For each pair of the following compounds, identify which compound and conditions would react more…
A: Biomolecular nucleophilic substitution reaction (SN2 reaction) involves the attack of nucleophile…
Q: 6. For each of the following acid base reactions shown below, draw curved arrows to show proton…
A: Sodium hydroxide (NaOH) is a very strong base, thus it can easily abstract the acidic proton on…
Q: His musboe eri lo ricillim eq enq 8. The label on a bottle of "sports drink" indicates that the…
A: Amount of sodium ion in the drink = 50 mgAmount of potassium ion in the drink = 55 mgVolume of the…
Q: Which product would form for the reaction below? OH Me HB (aq) Br Me Me Me Br. Me Me Br ? Me Me O Me…
A:
Q: 150.0 mL of 0.500 mol/L sodium hydroxide, NaOH (aq), is required to titrate 125.0 mL of hydrochloric…
A: This is strong acid HCl - strong base NaOH titration. And give neutralization reactionNaOH(aq) +…
Q: Macmillan Learning A fossil is found to have a "Clevel of 63.0% compared to living organisms. How…
A:
Q: a. Consider the drug naproxen, a nonsteroidal anti-inflammatory drug sold under the brand name…
A: Drug molecules are givenWe have to check the correct statements. We have to find the configuration…
Q: Obtain 2-methylbutanoic acid from diethyl malonate. Comment the reactions briefly.
A: Diethyl malonate is basically a 1,3 diester compound. It is an important starting material for the…
Q: A substance has a low melting point (i.e. below room temperature). It does not conduct electricity…
A: Metals (due to free electrons) and ionic compounds (in aqueous medium produce ions) conduct…
Q: Each of 10 stereoisomeric sugar derivatives can be prepared via a multiple-step synthesis starting…
A: As 1L is the enantiomer of 1D, hence 1L is the non-superimposable mirror image of 1D.
Q: 4. A student tested the pH of 0.1 mol/L solutions of hydrochloric acid and acetic acid. The pH of…
A: The question is based on the concept of pH of the solution. it is defined as a negative logarithm of…
Q: What is the pH change when 40.0 mL of 0.100 M NaOH solution is added to 40 mL of 0.100 M H3PO4…
A: Volume of 0.1 M NaOH solution = 40 mLVolume of 0.1 M H3PO4 solution = 40 mL
Q: Active Ingredient Raw Materials 2 kgs. Salicylic acid and 2.5 kgs. acetic anhydride Aspirin…
A: 3 subdivision questions are based on mole concept. Each of them are to be answered by the respective…
Q: Overlap in the atomic orbitals shown below will result in what type of bonding molecular orbital?…
A: This is formed by the lateral overlapping of two p-orbitals
Q: The reaction conditions to carry out the production of cyclopentene using bromocyclopentane as the…
A:
Q: Consider three isolated ions: Mg2+, O²- and Na*. Which is true about these ions? A. All of the ions…
A: Atomic radius is the distance between the central nucleus and the outermost electron. Atomic radius…
Q: Classify each of the following amines as primary, secondary or tertiary. CH₂ -NH- CHỊCH,CH, CHINH, |…
A: RNH2 = Primary, because N-atom is linked to one alkyl group(R) and two H-atoms.R2NH = secondary…
Q: Z 'N HO™ H₂O heat
A:
Q: a. How does the combustion of coal to generate electricity contribute to acid precipitation? (Refer…
A: a). Because some contaminants are released into the atmosphere when coal is burned to produce power,…
Q: Pressure affects the rate of reaction. TRUE or FALSE
A: Effect of pressure on reaction rate.
Q: A marine biologist is preparing a deep-sea submersible for a dive. The sub stores breathing air…
A: The width of the sphere, w = 69.0 cm Volume of air, V1 = 3900 L at a pressure (P1) of 1 atm
Q: How many core and valence electrons does one atom of iodine (1) have? 7 core and 36 valence 46 core…
A: Outermost shell containing electrons are known as valence electrons and except valence electrons…
Q: Energy Which letter represents the location of reaction intermediates? a) B and E b) C m Oc) B d) C…
A: Reaction intermediate or an intermediate is a molecular entity that is formed from the reactants but…
Q: H₂C For the following structural formula, pick the structure that accurately represents its MOST…
A:
Q: Question 1 Isomer A B CH 3 OSI(CH3)3 Which isomer is chiral? Isomer B CH3 OSI(CH3)3
A: A compound is said to be chiral if it has a chiral carbon which has four different groups attached…
Q: What is the splitting pattern expected for the proton at C-2 in the following compound? HH He H H Br…
A: 1H NMR spectroscopy is used to determine the structure of unknown compound in organic chemistry. The…
Q: 1) Which of the following elements is a metal? A) nitrogen 2) Which of the following elements is a…
A: 1.nitrogen is a non metalic element Which belongs to group 15.2.fluorine is also a non metal which…
Q: ) The critical point coordinates of hexane (C6H14) are 234.5°C, 3020 kPa, and its normal boiling…
A: The question is based on the concept of phase transition.we need to calculate the pressure asked in…
Q: Reaction #2 Figure #2 Br compound A KOH, ethanol heat لمی میں نہیں ہیں compound B compound C…
A:
Step by step
Solved in 3 steps with 2 images


