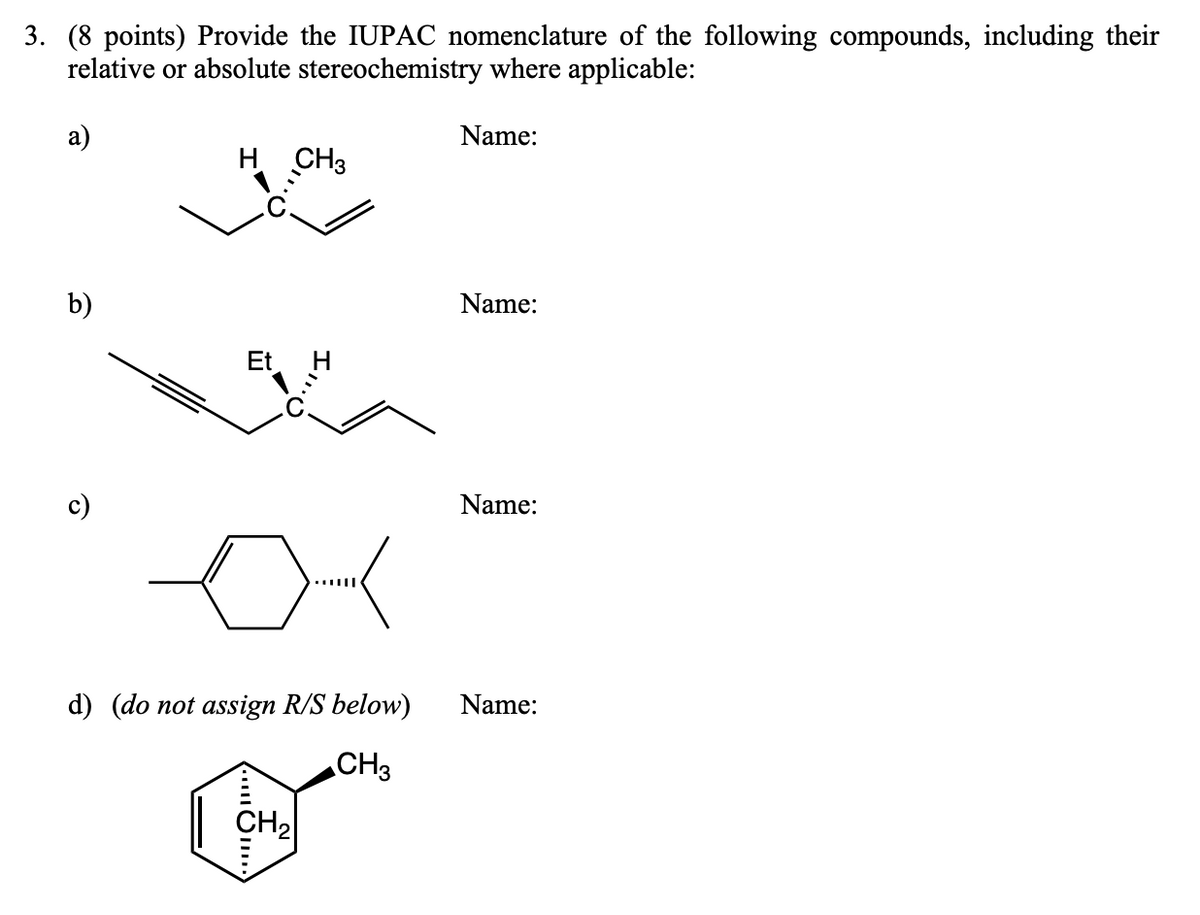
3. (8 points) Provide the IUPAC nomenclature of the following compounds, including their relative or absolute stereochemistry where applicable: a) H CH3 Name: b) Name: Et Name: d) (do not assign R/S below) Name: CH3 CH₂
Q: Please don't provide handwritten solution. Can you perform this multistep synthesis (no arrow…
A: In case of any doubt please feel free to ask.
Q: 3. Which of the following compounds can exist as cis-trans isomers? Draw each cis-trans pair. a)…
A: In chemistry, alkenes can exist as cis and trans isomers. Shown below are the structures of the…
Q: 2. Tetrodotoxin (TTX) is a potent neurotoxin. Its name derives from Tetraodontiformes, an order that…
A: Objective: Inspired by the structure and function of tetrodotoxin (TTX), the proposed research…
Q: 2.F. 2.G. 2.H. diene + R2BH diene + BH3 RBH 2 H H B. B. A s + BH3 RBH2 9-BBN Page 3 of 4
A:
Q: Calculate the pH at the equivalence point in titration 0.100 M solutions of each of the following…
A: Step 1:Step 2:
Q: Problems: 1. Predict the product and show the mechanism for the following intramolecular reaction. H…
A: This reaction is completed in four steps of the reaction mechanism. Step 1: In the first step, the…
Q: None
A: The difference in boiling points between carbon tetrachloride (CCl4) and chloroform (CHCl3) can be…
Q: Complete solutions need
A: Given: MnO4(aq)−+C2O4(aq)2−→MnO2(aq)+CO3(aq)2−Step 1: Write the…
Q: What is the pH at the equivalence point in the titration of a 28.6 mL sample of a 0.490 M aqueous…
A: Step 1:The volume of the aqueous hydrofluoric acid is 28.6 mL and the concentration is 0.490…
Q: Review Homework REQUIRED (NOT Pie Progress) Question 18 of 46 (2 points) | Question Attempt: 1 of…
A:
Q: Suppose that in a certain chemical process the reaction time y (hr) is related to the temperature…
A:
Q: Explain why phenol has a lower pKa than ethanol. OH OH phenol ethanol
A: Phenol does indeed have a lower pKa than ethanol, meaning it's a stronger acid. Here's why:Stability…
Q: 3) In the box below, provide the starting material(s) necessary to synthesize the provided product…
A: Step 1: Step 2: Step 3: Step 4:
Q: Organic Chemistry problem. Please help. Please provide a reasonable product structure for the…
A: Step 1: Information: This is the example of Alpha substituted reaction going to carbanion or…
Q: Determine whether the following are aromatic, nonaromatic or antiaromatic.
A: Step 1:A nonaromatic compound is a molecule that does not meet all the criteria for aromaticity.…
Q: Which of the following carbocations is/are likely to rearrange? IV I II Ob. III Oc II and V Od IV
A: Here, we are aware of the following carbocation stability: Whereas secondary carbocation is more…
Q: None
A: Part 2: Explanation:1. For the CH2CH compound, the CH proton will exhibit a triplet splitting…
Q: 3. Coal, which is primarily carbon, can be converted to natural gas, primarily CH4, by the reaction…
A: The forward reaction produces CH₄, while the reverse reaction produces C and H₂.To favor the…
Q: None
A:
Q: If I were trying to separate two compounds that eluted closely together. How would theirseparation…
A: A crooked column can significantly hinder the separation of two closely eluting compounds in column…
Q: using the table below can you help me calculate A) the concentration of weak acid (HC2H302) using…
A: Sure, The concentration of the weak acid (HC2H3O2) and conjugate base (C2H3O2) in the solutions of…
Q: Demonstrate that the probability of occupying a given vibrational energy level does not depend on…
A: Step 1:Step 2:Step 3:Step 4:
Q: Draw the products for the following reactions. Hint: I am asking for ALL products. Br MeOH heat H₂O…
A:
Q: In a titration experiment, 15.0 mL of 0.15 M Ba(OH)2 is required to titrate a 20.0 mL of an unknown…
A: Step 1: First ,Write the balanced chemical equation 2HNO₃ + Ba(OH)₂ → Ba(NO₃)₂ + 2H₂O Determine…
Q: Drawing Lewis Structures For each molecule, calculate the total number of valence electrons. Then,…
A: In general, we can count the total valence electrons of molecules by getting the summation of the…
Q: Solution for the uploaded question please.
A: Step 1:Step 2:Step 3:
Q: Base Base Meid Acid CH3CH2COOH(aq) + H2O()CH3CH2COO(aq) + H30*(aq) 2. Propanoic acid, CH2CH2COOH, is…
A:
Q: Show work, thank you!
A: The attached image mostly shows gray screen. No question was asked. It would be better to…
Q: Consider the branched alkane below and complete the following tasks based on its structure. Part: 0…
A:
Q: The hemiacetal reaction is also reversible and can also be catalyzed by either acid or base.…
A: In case of any doubt please feel free to ask.
Q: <HW 4: Chapter 14 (Aldehydes and Ketones) - Attempt 1 Question 4 Part A Spell out the common name of…
A: Step 1: Information : As the given example having functional group RCOR It indicates the presence…
Q: Please look at the image attached Please please please answer everything super super fast
A: --- ---The first slide is an introduction.The title of this article is "Understanding Sun Protection…
Q: The chart shows the names of the different bands of the electromagnetic spectrum. Which of the bands…
A: Step 1:From the spectrum, The wavelength of radiowaves are of the order of, 1The wavelength of…
Q: Problem 77 of 80 Submit Curved arrows are used to illustrate the flow of electrons. Using the…
A:
Q: Calculate the pH when 45.0 mL of 0.155 M NaOH is mixed with 20.0 mL of 0.310 M HF (Ka = 6.8 × 10⁻⁴).
A: Step 1:A buffer solution can be made by partially neutralizing the acid with a strong acid. This…
Q: 1,3-Cyclopentadiene has to be used quickly after it is made, or stored cold. Which reaction below is…
A: cyclopentadiene reacts with water and produces the 1,2 addition. In the above reaction,…
Q: please answer in text form and in proper format answer with must explanation , calculation for each…
A: The reaction shown in the image is a conversion of an alcohol to an alkyl chloride using phosphorus…
Q: Draw two resonance structures for the species below. :0: CH₂
A:
Q: Please step by step solutions
A: Concentration of ammonia is 1-2(0.00314) = 0.99372
Q: Macmillan Learning Consider the reaction. I CH3 3 NaOH CH3 What are the fatty acid products of the…
A: Saponification is the process in which triglycerides are combined with a strong base to form fatty…
Q: 19. What is the product? a. b. Li/NH3 ? ethanol d. e. not a.-d.
A:
Q: Organic Chemistry problem. Please help solve. Thank you.What is the mechanism by which the reaction…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: This is an excerpt from an academic journal article about TymeBank, a digital bank in South Africa.…
Q: Identify the Michael donor and Michael acceptor that could be used to prepare the following compound…
A:
Q: Identify the structure of 3-phenylpentane 1 ΠΙ CHÍCH,CH HẠCH CH a. I IV O > d. II IV. OH СЊСЊ СНСН…
A:
Q: 5. What are the spin system of the following compounds: (a) Cl₂CHCH2CHC₁₂ (b) CF3C=C-H (c) CI- H H H…
A: Second image In Nuclear Magnetic Resonance (NMR) spectroscopy, the term "spin system" refers to a…
Q: Use acetylene as a starting material and design a synthesis for pentanal
A: Step 1: Step 2: Step 3: Step 4:
Q: Compound X has the molecular formula C7H14. Hydrogenation of compound X produces…
A: Step 1: Information: Give one data the compound X having molecular formula C7H14 Hydrogenation of…
Q: The correct IUPAC name for this compound is : H₂C. H&G HyG CH₂ O a.…
A: Approach to solving the question:In naming hydrocarbons, we need to consider the longest parent…
Q: Dinitrogen tetraoxide is a colorless gas at room temperature. It can dissociate into nitrogen…
A: The equilibrium:N2O4 ←→ 2NO2The expression for equilibrium constant,K = [NO2]2 = constant…
Please Write Step by Step Answer
Otherwise i give DISLIKE !!
Step by step
Solved in 2 steps with 2 images

- In some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.In some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1- phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Whichproduct predominates—the product of inversion or the product of retention of conguration? (c) Suggest an explanation for this phenomenon.In some nucleophilic substitutions under SN1 conditions, completeracemization does not occur and a small excess of one enantiomer ispresent. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data.(b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.
- Addition of HCl to alkene X forms two alkyl halides Y and Z. (A) Label Y and Z as a kinetic or thermodynamic product and explain why. (B) Explain why the addition of HCl occurs at the exocyclic C=C, rather than the other C=CFor the following molecule: A. Draw both chair conformations and label substitutents as axial or equatorial B. Indicate which conformation is more stable using equilibrium arrows C. Predict the major product of the reaction with sodium methoxide D. Predict the major product of the reaction with potassium tert-butoxideIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.
- what is degree unstaturation of the following ? and draw 2 structures per each number8. The anti-formation of (5)-3-phenyl-2-butanone is included. Draw the attack produce of NaBHa by the face if of this conformation.For each of the following: I) draw the structures of all product(s), both constitutional and stereoisomers; ii.) Indicate the stereocenters with and asterisk;



