Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter14: Rotational And Vibrational Spectroscopy
Section: Chapter Questions
Problem 14.24E: Which of the following molecules should have pure rotational spectra? a Dimethyltriacetylene,...
Related questions
Question
For each of the following: I) draw the structures of all product(s), both constitutional and stereoisomers; ii.) Indicate the stereocenters with and asterisk;
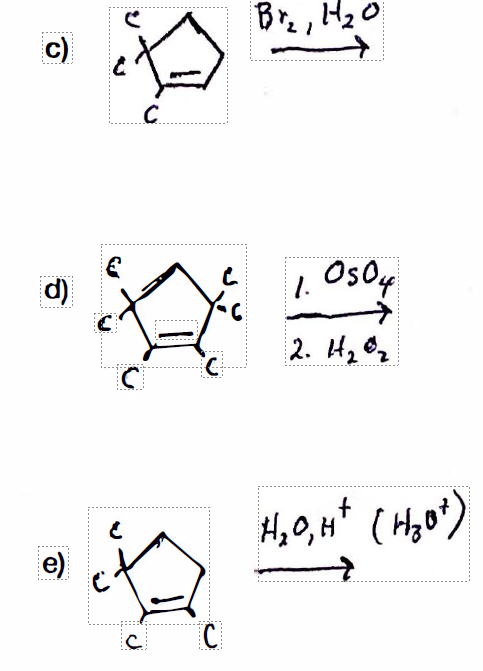
Transcribed Image Text:Bre, H20
c)
C.
d)
1. Os0y
2. Hz Oz
4,0, H* ( H,o*)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,