3. Assuming we start with 200 mg of cyclopentadiene and 300 mg of maleic anhydride, and 375 mg of the cycloadduct anhydride is obtained. Which is the limiting reagent? Calculate the percent yield for the anhydride.
3. Assuming we start with 200 mg of cyclopentadiene and 300 mg of maleic anhydride, and 375 mg of the cycloadduct anhydride is obtained. Which is the limiting reagent? Calculate the percent yield for the anhydride.
Chapter10: Organohalides
Section10.2: Preparing Alkyl Halides From Alkanes: Radical Halogenation
Problem 4P: Taking the relative reactivities of 1°, 2°, and 3° hydrogen atoms into account, what product(s)...
Related questions
Question
Can i get help with number 3

Transcribed Image Text:е ехо
dride. Which one will be favored?
3. Assuming we start with 200 mg of cyclopentadiene and 300 mg of maleic anhydride,
and 375 mg of the cycloadduct anhydride is obtained. Which is the limiting reagent?
Calculate the percent yield for the anhydride.
mnlete hoW.Could you separate the desired
2.
Expert Solution
Step 1
The reaction of cyclopentadiene with maleic anhydride giving cycloadduct anhydride can be given as: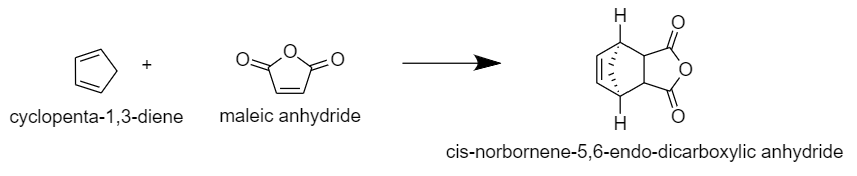
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning