3. Balance the equation and determine how much ammonium sulfate is made from 5 moles of sulfuric acid H2SO4 + NH4OH --> H20 + (NH4)2S04 О 12 O 10 O 15
3. Balance the equation and determine how much ammonium sulfate is made from 5 moles of sulfuric acid H2SO4 + NH4OH --> H20 + (NH4)2S04 О 12 O 10 O 15
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.9QAP
Related questions
Question
100%
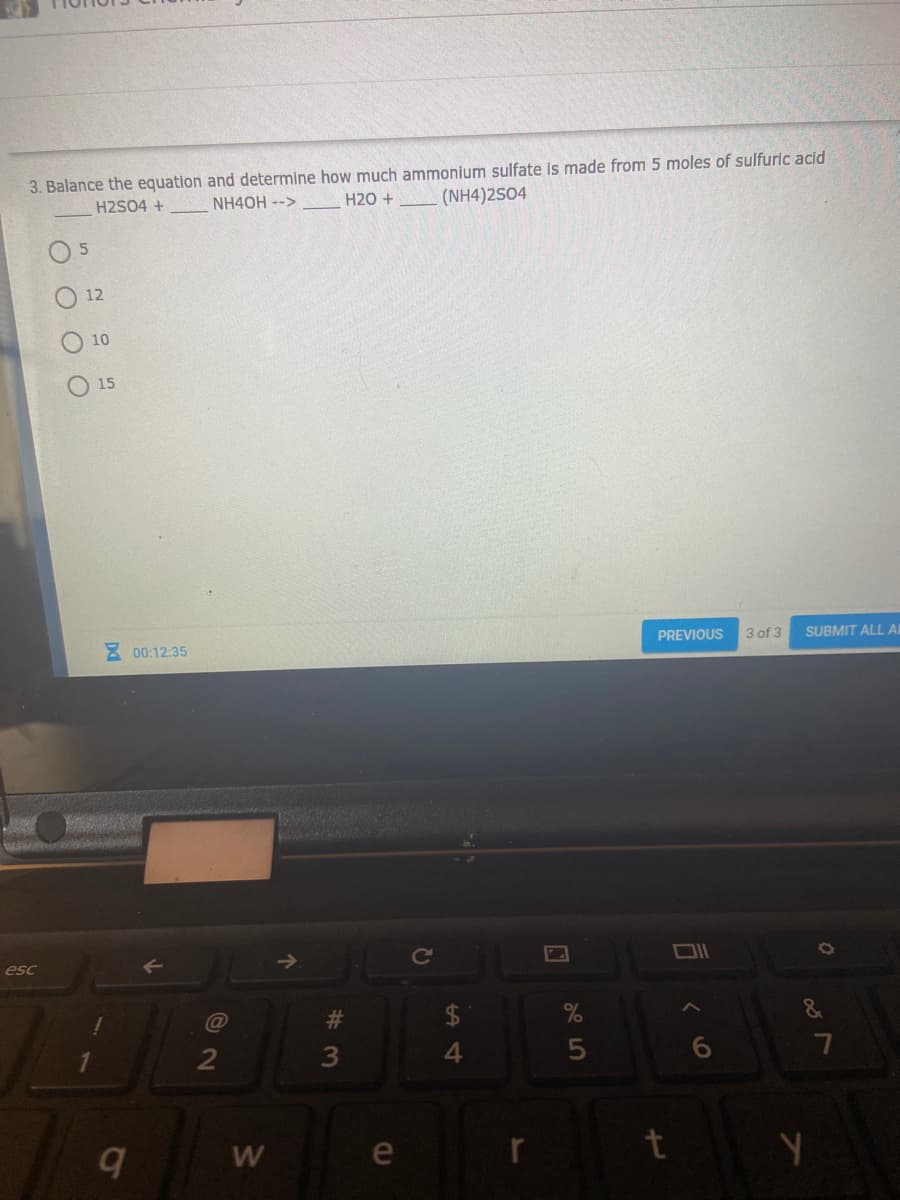
Transcribed Image Text:3. Balance the equation and determine how much ammonium sulfate is made from 5 moles of sulfuric acid
H2SO4 +
NH4OH -->
H20 +
(NH4)2S04
O 5
12
10
15
E 00:12:35
PREVIOUS
3 of 3
SUBMIT ALL AI
esc
23
$
&
4.
e
Expert Solution
Step 1
The balanced chemical equation for the reaction between sulfuric acid and ammonium hydroxide is
H2SO4 + 2 NH4OH → 2 H2O + (NH4)2SO4
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning



Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning