Q: Draw the resonance structures ? AY H;C-N CH3 BY CH3
A: A. The resonance structures of the given compound are,
Q: 2. Now write the condensed formulas for the following bond-line structures. la он Н HO.
A:
Q: d)Draw the resonance structures for the following: ÇHa H20
A: The above structure is for a protonated tertiary butanol. For a molecule to show resonance, the…
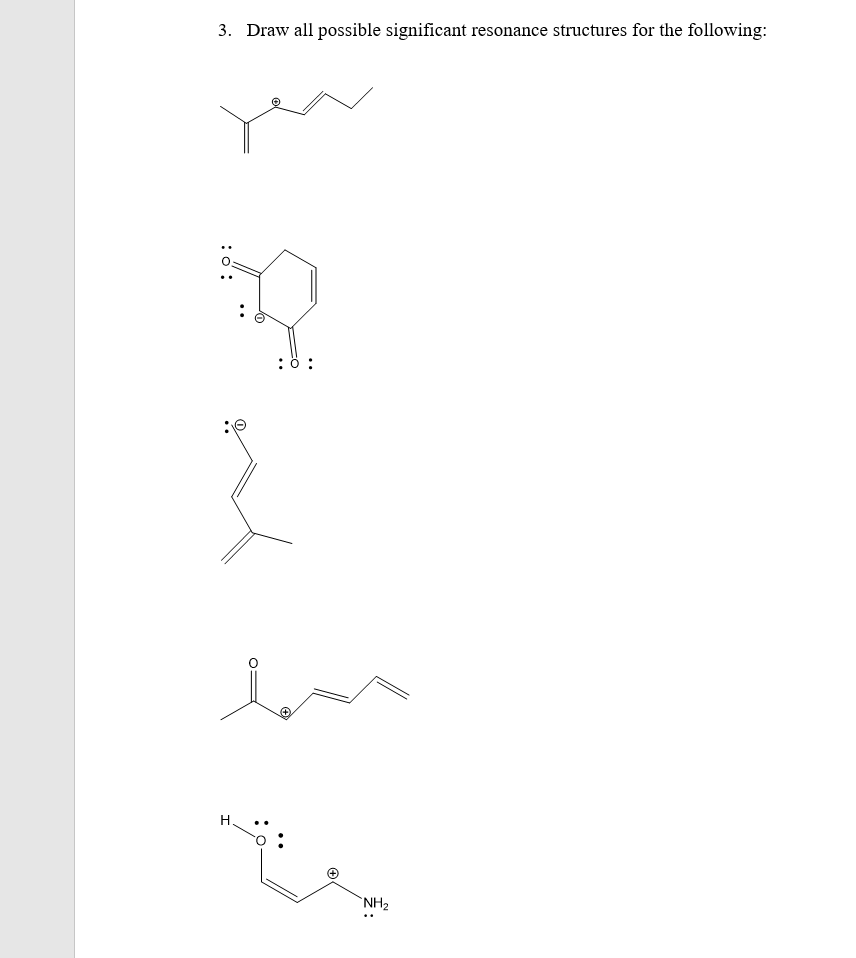
Q: For the following draw out all possible resonance structures using propre bond line notation . It is…
A: Resonance is a way of describing bonding in certain molecules or ions by the combination of several…
Q: 1. Draw three resonance structures with separation of charges for the following compound: NM22 MeO,
A:
Q: Draw at least 10 more resonance structures for acetaminophen, the active pain reliever in Tylenol.
A: In acetaminophen, the resonance takes place between the acetamino and hydroxyl groups along with the…
Q: 9. Draw all resonance structures of the following compounds, showing electron movement with curved…
A:
Q: a) Draw the structure below and the two other important resonance forms. b) Circle the most…
A: A question based on introduction to organic chemistry that is to be accomplished.
Q: 3. Draw all resonance structures for the following radicals.
A: Geometry of free radicals - ' shallow pyramid' Partially filled p orbital can participate in…
Q: Arrange the compounds in increasing order according to its availability of lone pair of N
A:
Q: Use resonance structures to explain why the compound shown below is considered to be antiaromatic.
A:
Q: Draw a second resonance structure for nitromethane, a compound used in drag racing fuels and in the…
A: Resonance is the term that is used to explain the delocalization of electrons in a compound. It is…
Q: H. (+) H3C CH3 CI
A:
Q: Draw
A: Dear student I have given answer to your question in the image format.
Q: II. (8 Points) Draw resonance structures for each of the following: а. с. b. d.
A:
Q: Part B: Draw the correct structure for th appropriate number of hydrogens. (2 m
A:
Q: Draw a second resonance form for the structure shown below. CH3 H₂C хон H
A:
Q: 2) Draw a second resonance structure for the following structures and then an overall resonance…
A:
Q: ecide whether each row is a set of resonance resonance structures? structures yes O no O yes O no O…
A: Resonance : involves transfer of electrons only. A. Here, methyl group is shifted. That's why these…
Q: Add curved arrows to show how the first resonance structure can be converted to the second.
A: a) Please find below the curved arrows showing how the first resonance structure can be converted to…
Q: 2) Draw two more resonance structures for the following:
A: Resonating structure
Q: 6. Which of the following does NOT depict resonance structures? (1) CH3-0-CH CH3 CH, 0 CH-CH, (2)…
A: Correct answer is (3) Resonating structures are correct in option (3) but arrow used should double…
Q: 2. Draw a resonance structure for the compound below.
A: The solution is as follows :
Q: 7. a) Draw the resonance arrows to form the structure on the right, b) Circle the major resonance…
A: The transformation given is,
Q: For each example, specify whether the two structures are resonance contributors to the same…
A: Resonance occurs only when there is something in conjugation. Conjugation occurs only there are…
Q: 1. Draw the contributing resonance structures for the following. :ö ЕН
A: While drawing resonating structures, position of atoms remains unchanged only position of electrons…
Q: 6. Draw resonance structures for the following species: (a) CH2 (b) :0: (c) H. C H нн H
A:
Q: Rank the resonance structures below from most to least important. :0: :0: H. H. H. 3) 2) 1)
A: Resonance structure: When more than one Lewis structure can be drawn, then the molecule or ion is…
Q: Draw a second resonance form for the structure shown below.
A: Resonance is a method of representation of bonds in various molecules or ions. Resonance structure…
Q: Draw additional resonance structures for each anion.
A: The resonance structure of an ion is obtained by the movement of delocalized electrons on the ion.…
Q: aw a second resonance form for the structure shown below. H. H3C CH3 N. ČH3
A: Resonance effect is a primary effect to give stabilization to a compound. In resonating structure…
Q: 1. Draw all resonance structures for the following molecules. CF3 a. b.
A: Resonance is defined as transfer of charge from one atom to another atom
Q: 1. Draw three resonance structures with separation of charges for the following compound: NME2 Meo.
A: Here, lone pair of nitrogen, of NMe2 goes to resonance. Because N is less electronegative than…
Q: With reference to compound A drawn below, label each compound as an isomer, a resonance structure,…
A: Isomer are those compounds which have same molecular formula but different structure. Resonating…
Q: Draw all possible resonance structures for the following cation, and indicate which structure makes…
A:
Q: Show how these resonance structures can be interconverted H3C H3C H3C CI H3C
A:
Q: With reference to compound A drawn below, label compound B as an isomer, a resonance structure, or…
A: Isomer of any compound is the structure with same molecular formula and different arrangement of…
Q: 2. Draw 2 resonance structures for the molecule below including arrows!
A:
Q: 1. Draw three resonance structures with separation of charges for the following compound: NM@2 Meo.
A: Since you have asked multiple questions, we will answer only first question for you. In order to get…
Q: Draw the contributing resonance structures for the following. E H
A: Given structure of the intermediate is : Draw the resonance contributing structures of the…
Q: 4. Draw all resonance structures for each compound below (including arrow pushing) and indicate…
A:
Q: :: :0: О.
A:
Q: 5. |Draw any relevant resonance structures for the compounds below. Use arrow pushing to show…
A: a) Given structure is:
Q: 9) Draw all the possible resonance structures for the following molecules. H. a) b) c) H2N-C=CH
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Which of the following is a resonance structure of the compound below? H3C CH3 H2C-CHOH H,CHC CH2…
A: Given data : Acetone Resonance Structure need to be write. Resonance Structure : Whenever a single…
Q: H;C-CH-C-CH=CH–CN
A: Delocalization of pi electrons in compound is known as resonance and the structure which are…
Q: 2. Which of the following pairs represent resonance structures? H2N & *HN & NH NH2 & H3C CH3 H3C CH2…
A: Of the given pairs of compound, some are pair of resonance structures and some are isomers.…
Q: NaOH (A) Include ALL relevant resonance structures CN (1 equiv) -CO2ET 1) (A) (B) (C) Heat 2) H3O*
A: The explanation of the organic reactions are given below
Q: Provide the following 2 resonance structures and indicate whether or not they're major or minor
A: Resonating structures with a lesser charged atoms is more stable.
Step by step
Solved in 2 steps with 1 images

- I need to indicate how the first resonance structure (the one with #4 above it) turns into the second one with arrows, but I cannot figure it out.Provide one more resonance form for the molecule below.Fill in the multiple bonds in the following model of naphthalene, C10H8 (gravy = C, ivory = H). How many resonance structures does naphthalene have? Draw them.
- For each example, specify whether the two structures are resonance contributors to the same resonance hybrid.pls help ASAP! “which term best describes the relationship between the following two molecules?”A. Find the moleular formula of diazomethane. b. Draw iits lewis structure and resonance forms as neccessary c. Explain why diazomethane is not very stable
- Rank the following in order of decreasing importance as a contributing resonance structure to the molecular structure of acetone, CH3COCH3 (more important > less important)Make a continuous model for C4H10 by using 4 black 4 hole carbon atoms, 10 white one hole hydrogen atoms, and 13 pink bonds. Then write in wedge dash notation C4H10 and add in missing hydrogen atoms.1. Add nonbonding electron pairs to each atom where it is implied by the line-angle drawings shown below. 2. Use the curved arrow formalism to generate four additional, valid resonance structures for each compound. 3. Rank your resonance structures in order of increasing contribution to the overall nature of the molecule.
- Draw a second resonance structure and the hybrid for each species, and then rank the two resonance structures and the hybrid in order of increasing stability.What are the next favourable resonance structures being asked for?2. Draw the each structures. For each structure, obtain the following information:a. Chargeb. Bond dipolesc. Over-all dipole

