Chapter8: Sampling, Standardization, And Calibration
Section: Chapter Questions
Problem 8.8QAP
Related questions
Question
100%
answer 3 and 4 only
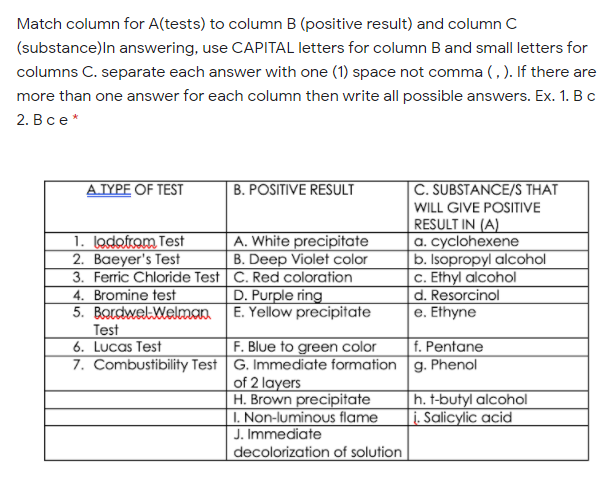
Transcribed Image Text:Match column for A(tests) to column B (positive result) and column C
(substance)ln answering, use CAPITAL letters for column B and small letters for
columns C. separate each answer with one (1) space not comma (, ). If there are
more than one answer for each column then write all possible answers. Ex. 1. Bc
2. В се*
AJYPE OF TEST
B. POSITIVE RESULT
C. SUBSTANCE/S THAT
WILL GIVE POSITIVE
1. lodofram Test
2. Baeyer's Test
3. Ferric Chloride Test C. Red coloration
4. Bromine test
5. BordwelWelman.
Test
6. Lucas Test
RESULT IN (A)
a. cyclohexene
|b. Isopropyl alcohol
c. Ethyl alcohol
d. Resorcinol
e. Ethyne
A. White precipitate
B. Deep Violet color
D. Purple ring
E. Yellow precipitate
F. Blue to green color
7. Combustibility Test G. Immediate formation g. Phenol
of 2 layers
H. Brown precipitate
I. Non-luminous flame
J. Immediate
decolorization of solution
f. Pentane
h. t-butyl alcohol
i. Salicylic acid
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you





