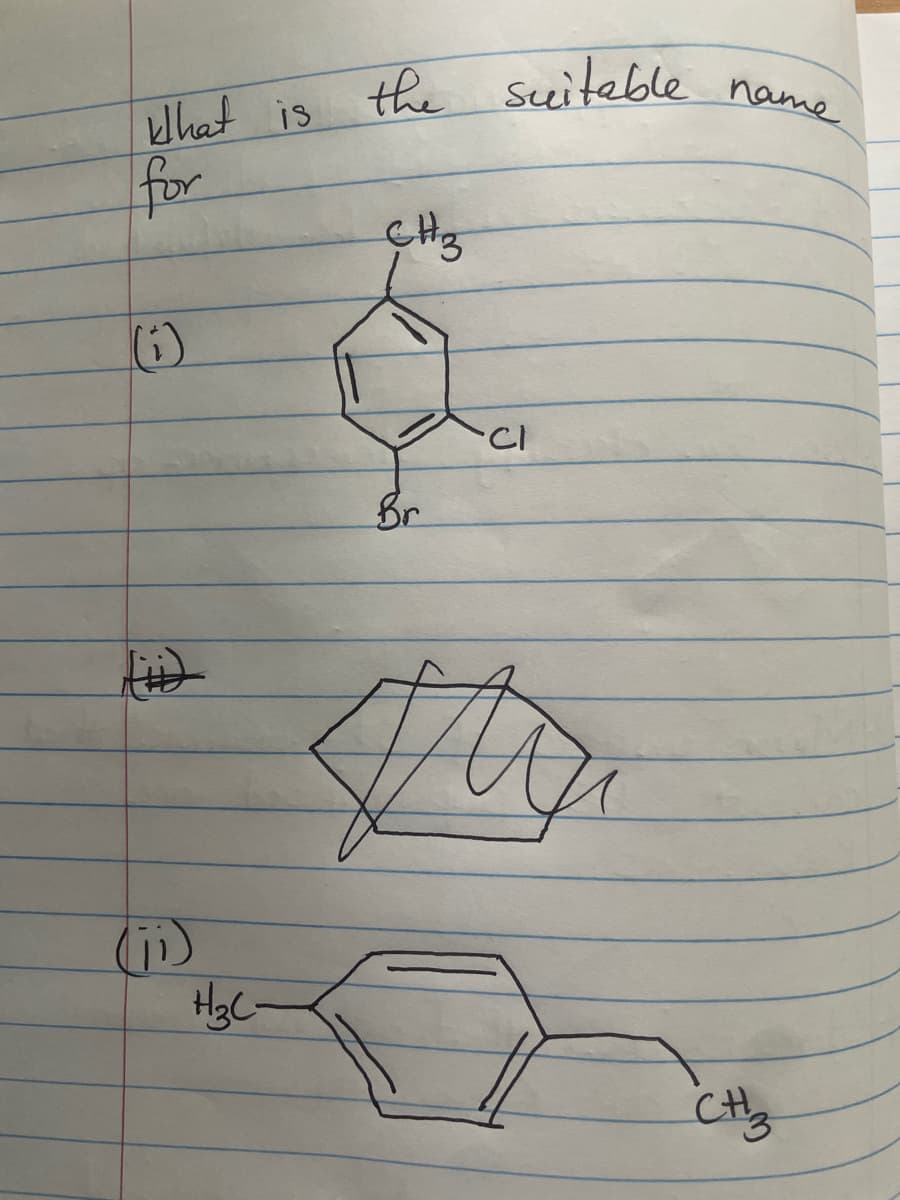
Q: C6H100, IR (cm-1): 2110, 3300 cm1 (sharp), 1H NMR (8): 3.31 (3H, s); 2.41 (1H, s), 1.43 (6H, s) H- I…
A:
Q: A sample containing NaOH only was titrated with standard 0.1280 M HCl, requiring 25.45 mL to reach a…
A:
Q: What could explain the simultaneous increases in the price of lithium and the production of lithuim.
A: Lithium is an alkali element belongs to s block of periodic table .
Q: what is the mole ratio of ammonia to ammonium chloride in a buffer with a pH of 9.00?
A: The answer to the following question is-
Q: (c) Calculate the n-electron energy of the pi electrons a pentadiene radical
A:
Q: In which energy range(s) would you expect to see absorption in an Infrared (IR) spectrum of dimethyl…
A:
Q: Transmitance Transmittance % Transmittance Please match the structure to the IR spectra. (letter)…
A:
Q: Identify the irreducible representations to which all five d orbitals of the central Xe atom in XeF4…
A:
Q: CI O 1. NaBH4 (0.25 eq.), CH3OH 2. H₂O, HCI 1. NaBH4 (0.50 eq.), CH3OH 2. DCI, D₂O
A: The above organic transformation can be carried out as follows
Q: A 25.0 mL sample of a 0.100M solution of aqueous trimethylamine is titrated with a 0.125M solution…
A: Strong acid + weak base ---> salt + water pH = 1/2(pkw - pkb- log C) Molarity = no. of moles of…
Q: The actual ionization energy of He is 2372.3 kJ/mol. Which of the following can be said about the…
A: We need to describe the shielding effect in helium.
Q: A researcher isolated 10 g of a Natural Product from a local plant. In an attempt to determine the…
A: 1) Polarimeter used by researcher to obtain the reading. A polarimeter is used to measure the…
Q: xn A Part A: Balanced Chemical Equations (must include physical states) Balanced Total Equation…
A:
Q: 1.50g of hydrocarbon undergoes complete combustion to produce 4.40g of CO2 and 2.50g of H2O. What is…
A: mass of carbon dioxide = 4.40 g mass of water = 2.50 g
Q: 1. Consider the reaction sequence given below: Compound C 1. LDA, THF, -78°C 2. Br NaOEt Br Compound…
A:
Q: Which ion could be used to precipitate Ba2+ from a solution of Ba(NO3)2? 1) Sr²+ 2) CIO4 3) SO4²- 4)…
A:
Q: The addition of 3.15 g of Ba(OH)₂ 8H₂O to a solution of 1.52 g of NH SCN in 100 g of water in a…
A:
Q: A certain metal M forms a soluble nitrate salt M(NO3)₂. Suppose the left half cell of a galvanic…
A: In the given Galvanic cell, the electrode is the same in both the half-cells which is the metal, M.…
Q: 1. How would you draw the condensed structural diagram for oct-3-yne 2. How would you draw the line…
A: Given, 1. How would you draw the condensed structural diagram for oct-3-yne 2. How would you draw…
Q: The freezing point of a 0.20 m solution of magnesium sulfate (MgSO4) is −1.8°C. What is the value of…
A:
Q: It said "You have calculated the amount of reactant which has been consumed, not the amount…
A: The half llif a reaction is the time taken by reactant to reduce the concentration to one half of…
Q: Draw a structural formula for 3-chloro-3-methylhexane.
A: Given -> 3-chloro-3-methylhexane
Q: 1. A student dissolves 2.006 g of FeSO4 7 H₂O in 40 mL of sulfuric acid and then dilutes it to…
A: #Q.1: Given the mass of FeSO4.7H2O initially taken to prepare the solution = 2.006 g The volume of…
Q: Hydrogen-powered fuel cells are seen increasingly as "clean" sources of energy and are now used in…
A: The volume occupied by 1 mol of gas at standard temperature and pressure (STP) is 22.4 litres.
Q: 1A H2A Li Be Na Mg 3B 48 58 68 7B K Ca Sc This ion is a(n) 8A 3A 4A 5A 6A 7A He BCN OF NE Si P S Cl…
A:
Q: PREDICT THE PRODUCT Provide the product(s) for the reactions shown below. No partial credit will be…
A: The product of the above organic transformation is given below, here NaBH4, LiAlH4 acts as reducing…
Q: Combustion of ethanol (C₂H6O) is a well know chemical reaction, where in the presence of O2 carbon…
A: Given, Ethanol ( C2H6O) react with O2 to produced carbon dioxide (CO2 ) and water (H2O) . The…
Q: The concentration of A was monitored for the reaction A → P and the following data were obtained.…
A:
Q: Fill in the orbital energy diagram for the oxide ion. E 2s 1s 2p-
A:
Q: You are studying the % CI in FeCl2. You have 5.53 g of Ferrous chloride (FeCl2) sample and after a…
A:
Q: You have 8.45 mol of the compound potassium hydrogen phosphate, K₂HPO4. How many potassium ions are…
A:
Q: 1. them. 3.2 CHEMISTRY HOLIDAY WORK 2022 Nalubula is investigating changes in mass of substances…
A: Mass of Zinc metal = 100 g Mass of sulfuric acid = 200 g
Q: Balloons may contain tiny microscopic holes, which allow the gas particles within to escape via…
A: When gas comes out of a small hole, we normally termed it as effusion. From Graham's law of…
Q: Draw structural formulas for organic products A and B in the window below. Cul H₂C=CHBr Li pentane A…
A: When alkylhalide is treated with Lithium (Li) it gives an organometallic compound alkyl lithium…
Q: molality of the 0.75 mol of salt (NaCl) dissolved in 155 g of water?
A: Given, Moles = 0.75 mol Mass of solvent = 155 g = 0.155 Kg Molality = mole/mass of solvent(Kg)
Q: Write electron configurations to show the first two ionization processes for lithium. (Express your…
A:
Q: 4. Predict the products: ин от + ін гоём 2 + f ян д дн - - он он - он он
A: The answer to the following question is-
Q: Using only the periodic table arrange the following elements in order of increasing ionization…
A:
Q: Please explain question 1 and 8 1. How many degrees of unsaturation are present in your unknown…
A: Benzene undergo Electrophilic substitution reaction in which an electrophile replaces H of its ring.
Q: Consider the following hypothetical equation. A2B3 + 3 C2D --> A2D3 + 3 C2B OR A2 B3 + 3 C2 D…
A:
Q: a b CH CH₂ CH₂ CH₂
A:
Q: Calculate the weight of pure sodium carbonate that is necessary to prepare 2.806 L of 0.223 N…
A: We need to find molarity of the solution . We assume molar mass of sodium-carbonate in 106 g/mol,…
Q: NH3(aq) + H₂O(1) = NH(aq) + OH- (aq) The equilibrium hydroxide concentration in a 0.0220 M NH3…
A: Concept based on the pH calculation of the the given concentrated solution.
Q: CHEMISTRY RATE WILL BE GIVEN! NO LONG EXPLANATION NEEDED. PLEASE ANSWR ALL
A:
Q: a This question uses He to explore the ability of one electron to shield another from the positive…
A: The formula for the energy of an electron is: E = -NA Rhc * Z2n2 where NA Rhc = 1312 kJ/mol Z =…
Q: A substance decomposes according to first order kinetics. The rate constants at various temperatures…
A: In the given question the activation energy for a first order reaction has been asked where the data…
Q: For a certain first order reaction, it takes 449 seconds for the concentration of reactant to…
A:
Q: C9H9N IR (cm-1): 1H NMR (5): 13C NMR (5): 3050, 2950, 2240, 1630 7.5 (2H, d), 7.1 (2H, d), 2.3 (2H,…
A:
Q: A 25.00 mL sample of 0.280 M LiOH is titrated with 0.750 M HCl at 25 °C. a. Calculate the initial…
A: Given, Molarity of LiOH = 0.280 M. Volume of LiOH = 25.00 mL. Molarity of HCl = 0.750 M. The…
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A:
Step by step
Solved in 2 steps with 2 images

- How B shows no effervescence as O2 is produced from decomposition of h202 I supposeA compound X is bitter in taste. It is a component of washing powder& reacts withdil. HCI to produce brisk effervescence dur to colourless, odourless gas Y wich turns lime water milky due to formation of Z. When excs of CO2is passed, ilkinessdisappears due to formation of P. Identify X, Y, And Z & P.Kp of this quation 2A+3B=4C+5D
- What are the respective concentrations (M) of Fe3+ and I- afforded by dissolving 0.200 mol FeI3 in water and diluting to 725 mL?Which of the following compound/s are correct in the given statements? 1.What compound/s will be able to get the change in the color of AlCl3 Crystal? If tert-butyl chloride is added in AlCl3. 2. Which compound will react in water under under light conditions with Br2? 3. Which compound will react in Kl under under dark conditions with I2?Which of the following is not one of the characteristics of edtan?A. Stability in alkaline environmentB. High capacity for making complexesC. Reacts with +2 and +3 valence nonmetal ions at a ratio of 1: 1D. It is resistant to hydrolysis
- The Palladium complex shown in the reaction scheme below will catalyze the addition of SiH4 to an alkene. Identify the reaction types. A. B. C. .D. E.?One and only one combination of cobalt salt and solvent should have turned blue, thusindicating the presence of [CoCl4]2− ion. Explain why only this combination underwent a reactionand the others did not.What is the process: a. Converts 214Bi to 214Po b. Converts 40K into 40Ar

