3. Mechanisms i) Consider the following reaction: Br. Br PPH3 R H. CBr4 R H. This is a part of the so-called Corey-Fuchs reaction, which converts an aldehyde into the corresponding dibromoalkene. The first step of the mechanism goes as follows: Ph Ph-P : Br-CB 3 Ph a) Draw the products in the boxes according to the arrowpushing shown. b) Consider this step as a bimolecular nucleophilic substitution (SN2). Identify the nucleophile electrophile and leaving group in this step. Nucleophile Electrophile Leaving Group
3. Mechanisms i) Consider the following reaction: Br. Br PPH3 R H. CBr4 R H. This is a part of the so-called Corey-Fuchs reaction, which converts an aldehyde into the corresponding dibromoalkene. The first step of the mechanism goes as follows: Ph Ph-P : Br-CB 3 Ph a) Draw the products in the boxes according to the arrowpushing shown. b) Consider this step as a bimolecular nucleophilic substitution (SN2). Identify the nucleophile electrophile and leaving group in this step. Nucleophile Electrophile Leaving Group
Chapter9: Alkynes: An Introduction To Organic Synthesis
Section9.SE: Something Extra
Problem 49AP: Occasionally, a chemist might need to invert the stereochemistry of an alkene—that is, to convert...
Related questions
Question
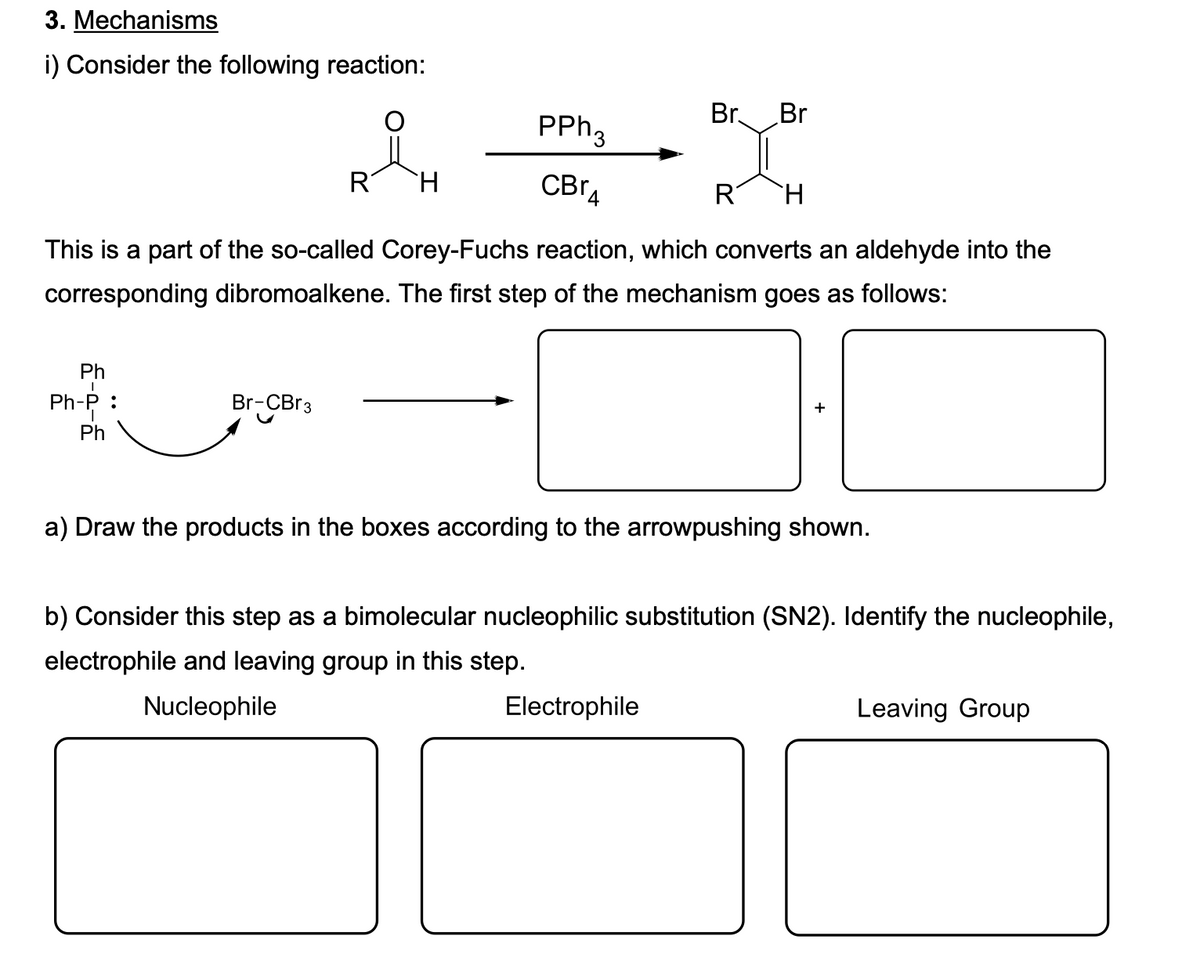
Transcribed Image Text:3. Mechanisms
i) Consider the following reaction:
PPH3
Br.
Br
R
CBrA
R
This is a part of the so-called Corey-Fuchs reaction, which converts an aldehyde into the
corresponding dibromoalkene. The first step of the mechanism goes as follows:
Ph
Ph-P :
Br-CBr3
Ph
a) Draw the products in the boxes according to the arrowpushing shown.
b) Consider this step as a bimolecular nucleophilic substitution (SN2). Identify the nucleophile,
electrophile and leaving group in this step.
Nucleophile
Electrophile
Leaving Group
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 6 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

