Q: Which of the following is a strong acid? O acetic acid O hydrochloric acid hydrofluoric acid O…
A: Which of the following is a strong acid ?
Q: Which aldohexoses could be obtained if D-erythrose undergoes 2 cycles of the Fischer-Killani Chain…
A: D- Erythrose undergoes 2 cycles of the Fischer Killani chain extension produces D-allose, D-altrose,…
Q: Which of the following structures is (R)-4-ethyloctane? IV IV
A: In this question we have to tell the structure of the compound.
Q: The pK, value for HF is 3.14. Would a buffer prepared from HF and NaF with a pH of 1.14 be…
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Suppose that an in-ground swimming pool has a length and width of 22.0 ft and 39.5 ft, respectively,…
A:
Q: When [Cu2+] = 1.46 M, the observed cell potential at 298 K for an electrochemical cell with the…
A: The overall reaction : Cu2+(aq) + Mn(s) ---> Cu(s) + Mn2+(aq) [Cu2+] = 1.46 M Ecell = 1.620 V…
Q: CI N(CH3)2 NH(CH3)2 `N' 'N
A:
Q: 0.01 mol of solid NaOH was added to a 500 mL solution containing 0.55 M C6H5CH2COOH and 0.75…
A:
Q: 1. What determines the frequency and period of an oscillating mass?
A: As you didn't specify question number you need hence I'm answering first question only. If you need…
Q: Iron(III) chloride and potassium bromide. This is the net ionic equation I got, but I could easily…
A: The net ionic equation is written after eliminating the common ions on both sides of equation and…
Q: Two 30.0 mL samples, one 0.100 M HCl and the other 0.100 M HF, wore titrated with 0.200 M KOH.…
A: A.) , B) Given acids are monoprotic ( Have one acidic H ) and base is KOH , so volume needed at…
Q: If you ‘overshot’ the endpoint of your standardization titrations, how will the calculated…
A: A question based on acid-base titrations that is to be accomplished.
Q: 1. For functional group and hydrocarbon each: -Give definition: -Give classification: -Give…
A: Functional groups ad hydrocarbons are part of organic chemistry.
Q: During the heat addition process, the temperature of a system increases by 50 °F. Express Your ans…
A:
Q: Match the following descriptions to the correct type of reaction. There are two answers that not be…
A:
Q: APITAL Letter of the correc- th statements are true 1st statement is false and 1st statement is true…
A: 10) Spontaneity is determined by Gibbs free energy change. If □G is negative then reaction is…
Q: image is an example of what type of unit cell? body-centered cubic O face-centered hexagonal…
A:
Q: When the Pb2+ concentration is 1.02 M, the observed cell potential at 298K for an electrochemical…
A:
Q: What is the emf of this cell under standard conditions? Express your answer using three significant…
A:
Q: When the vapor pressure of solution decreases, What will happen to the boiling polll and freezing…
A: vapor pressure of solution decreases means that solution having more number of solute particles. as…
Q: Please help with this question question 9
A:
Q: (b) Solution R contains the mildly soluble hydroxide of trivalent metal X as well as the highly…
A: We have find out the molarity of X in solution R.
Q: mpis leit eck my Click the "draw structure" button to launch the drawing utility. Draw the product…
A:
Q: The compound having the HIIGHEST percentage of cyclic hemiacetal at equilibrium is Compound The…
A:
Q: For each compound below, indicate which tests you would expect to be positive and which you would…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: What is the oxidation state and the coordination number of cobalt in [Co(H2O)4CI2]*? O Oxidation…
A:
Q: Bromine-88 is radioactive and has a half life of 16.3 seconds. Calculate the activity of a 6.7 mg…
A:
Q: What are the wavelength ranges for the following (assume 3 sigs.): (a) the AM radio band (540-1600…
A: What are the wavelength ranges for the following---
Q: Radioactive Carbon-14 The percentage Pof radioactive carbon- t/5700 14 remaining in a fossil after t…
A: #Radioactive carbon-14: Given the percentage of radioactive carbon-14 remaining after t years, P =…
Q: Define the following terms a. Aldose b. ketose c. Aldohexose d. Ketohexose
A: An aldose is a monosaccharide with a carbon backbone chain with a carbonyl group on the endmost…
Q: For each of the two electrochemical systems below write each half reaction and then calculate the…
A:
Q: Which of the following is a strong acid? O acetic acid O hydrochloric acid O hydrofluoric acid O…
A: acid which can dissociate 100% into ions in a solution called strong acid acid which can not…
Q: The combustion of pentane produces heat according to the equation C5H12(l) + 8O2(g) ® 5CO2(g) +…
A: ★ Answer : 156.71 grams CO2 are produced.
Q: Br 1. Pd°Ln, EtN, MECN, Me Me 2. MCPBA, CH2Cl2 3. HCIO, EIOH provide step by step analysis for each…
A: 1. a. Heck coupling. Epoxidation. Acid catalyzed epoxide opening with ethanol. (epoxide is opened at…
Q: Given the [OH^-] value of the soil sample = 3.5x10^-4 mol/ L. What is the classification of this…
A:
Q: 4.3 Lesson: PHYSICAL PROPERTIES OF SOLUTIONS (SOLUTION STOICHIOMETRY AND COLLIGATIVE PROPERTIES OF…
A: Given, Volume = 5 mL =0.0050 L Osmotic pressure (P) = 8.02 atm T =40 °c = 313 K R = 0.0821…
Q: Give the expected products for the aldol condensations of pentan-3-one. Draw the molecule(s) on the…
A: Aldol condensation: The alpha hydrogen of carbonyl compounds is acidic in nature and it is removed…
Q: Name the following compounds by the IUPAC system. The name must indicate the stereochemistry of the…
A:
Q: Rate Law Assignment 1. What is the rate law and rate constant for the reaction written below? 2NO…
A:
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A:
Q: E. E.
A: Conductors are those which do not have an energy gap between the valence and conduction band.…
Q: Which of the following is a major product of the Hofmann degradation shown in the box? LN(CH3)2 CH3I…
A: The reaction proceeds by the Hofmann elimination reaction mechanism. In the Hofmann elimination (or…
Q: For a certain equilibrium reaction, the following data are given AH = -57.2 kJ/mol K = 1.2 x 104…
A: ∆H = -57.2 KJ/mol. K = 1.2 × 104 The statements supported by the given data = ?
Q: ed under standard conditions, when the reaction is st ate the non-standard Free Energy change (AG)…
A: Given, □G°rxn = 2×(-16.6) = -33.2 kJ/mol Q = (1.5)2/(1.0)(0.06)3 = 625 □G = □G° + RT×ln(Q)
Q: Balance the equation for the first reaction - the synthesis of 3-mtrophthalhydrazide from 3-…
A: The correct answer is given below
Q: A Lewis structure for ozone (O3) is shown on the left below. Draw another equivalent resonance…
A:
Q: What is the property of a material that makes it transparent? Why does mineral such as emerald…
A:
Q: What is the coordination number of Ru in [Ru(NH3)5(H2O)]Cl2? 1 5 6 8
A:
Q: What arenium intermediates are formed in the ortho- and meta-substituted products of the following…
A: Given reaction is :
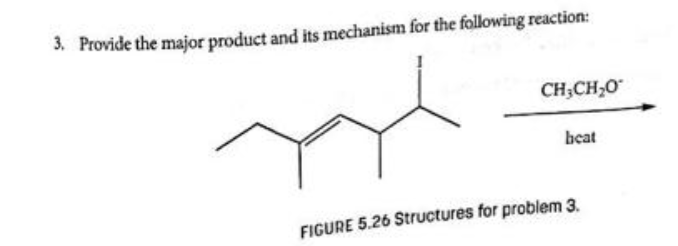
Step by step
Solved in 2 steps with 2 images

- Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaF5..8. What is the expected major for the following reaction ?6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd
- In each reaction box, place the best reagent and conditions from the list below. (A reagent may be used more than once.) Hint: step 1 is conducted at –78 °C.3. Prepare each compound from cyclopentanol. More than one step may be needed.llustrate the mechanism of the reaction. Include the intermediate product and by-product formed in the reaction. (see attached photo)Can you please provide the reaction mechanisms (arrow pushing mechanism)? You can use I2 as a reagent and do not need to show how it is generated from KI and Oxone.Thank you.
- V. Come up with a reasonable mechanism to explain the following reaction.The replacement of CH3OH to dimethyl sulfoxide, DMSO (CH3)2S=O) as a solvent in the substitution reaction below results in what?(b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane(CH3CH2CH2Br) with sodium hydroxide (NaOH). Two reactions are shown below:
- Tunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):N(CH2CH3)3 + HNO3 --------> a.) rewrite the reaction using bond-line structure of reagents and products of the reaction b.) supply the curved arrows explaining the mechanism of the reaction2. In not more than three (3) sentences, explain why terminal alkynes are acidic.3. What impurities are removed when acetylene gas is made to pass through an acidifiedsolution of CuSO4?4. Explain the difference in the rate of free radical bromination reactions of toluene and cyclohexane.

