3.1. Give the products that would be expected to be formed under the specified reaction conditions. Be sure to specify all aspects of the stereochemistry. (a) CH₂CH₂ HCI CH₂OH C,H₁₂O₂Cl (b) CH₂(CH₂) CH₂OH + CICH₂OCH, EtN(/Pr)₂ CH₂Cl₂ C₂H₁80₂
3.1. Give the products that would be expected to be formed under the specified reaction conditions. Be sure to specify all aspects of the stereochemistry. (a) CH₂CH₂ HCI CH₂OH C,H₁₂O₂Cl (b) CH₂(CH₂) CH₂OH + CICH₂OCH, EtN(/Pr)₂ CH₂Cl₂ C₂H₁80₂
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter15: Radical Reactions
Section: Chapter Questions
Problem 11E
Related questions
Question
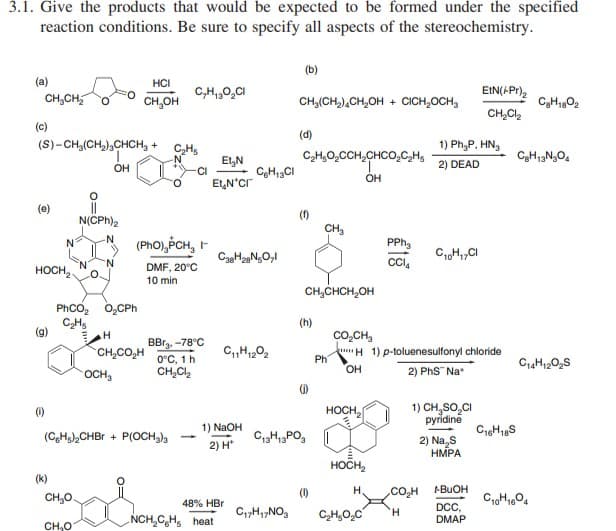
Transcribed Image Text:3.1. Give the products that would be expected to be formed under the specified
reaction conditions. Be sure to specify all aspects of the stereochemistry.
(a)
CH₂CH₂
(c)
HOCH₂
(1)
(S)-CH₂(CH₂)₂CHCH₂ + C₂H₁
(k)
CH₂O
OH
11
N(CPH)2
PhCO, O,CPh
C₂Hs
CH₂O
(CH)₂CHBr +
OCH₂
H
CH,CO,H
HCI
CH₂OH
-
(PhO),PCH, F
DMF, 20°C
10 min
C₂H₁₂O₂Cl
BB₂, -78°C
0°C, 1 h
CH₂Cl₂
(OCH3)3
Et₂N
EN*Cr
C38H28N5O,1
1) NaOH
2) H*
NCH₂CH heat
CH₁3Cl
C₁1H₁202
48% HBr
(b)
C₁7H₁7NO₂
CHJ(CH,),CH,OH + CICH,OCH,
(d)
C₂H₂O₂CCH₂CHCO₂C₂H₁
OH
(1)
CH₂CHCH₂OH
(h)
(₁)
C₁3H₁3PO3
CH₂
(1)
Ph
CO, CH
HOCH₂
HOCH₂
H
PPh₂
CC14
C₂H₂O₂C
H1) p-toluenesulfonyl chloride
OH
2) PhS™ Na*
1) Ph₂P, HN₂
2) DEAD
C10H₁7CI
CO₂H
'H
1) CH₂SO₂CI
pyridine
EIN(+Pr)₂
CH₂Cl₂
2) Na S
HMPA
+- BuOH
DCC,
DMAP
C₂H₁3N₂O4
C16H18S
C₂H1802
C14H12O₂S
C10H1604
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning