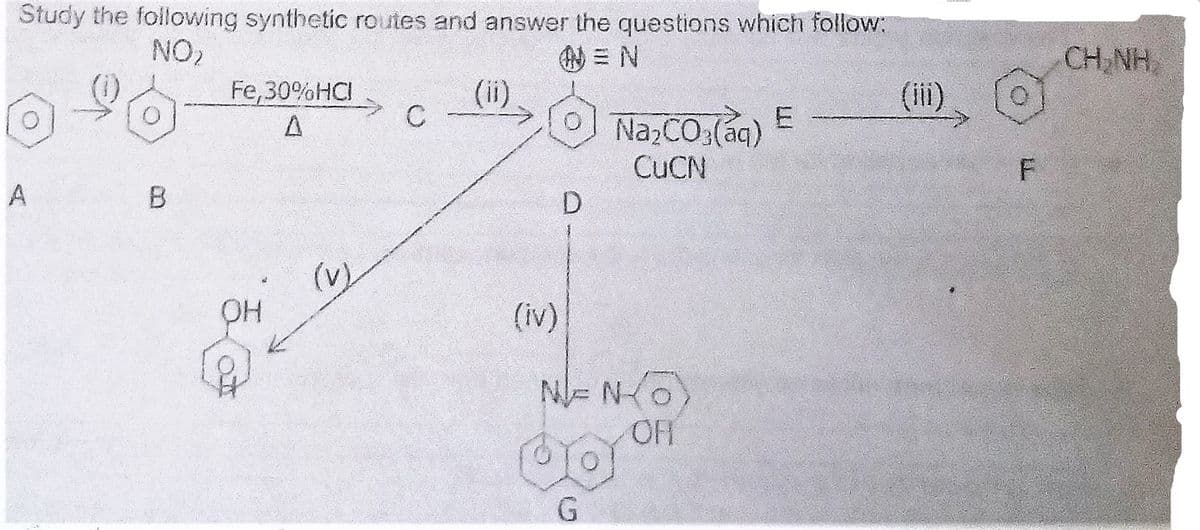
What are the names of the compound A, B, C, D, E, F, G & H. Hence suggest structures for compound C and E. What are the reagents and reaction conditions used for the following conversions? (i), (ii), (iii), (iv) & (v)
Q: Starting with benzene, give a sequence of laboratory reactions which could be used to prepare…
A:
Q: Using benzene and any necessary organic or inorganic reagents, suggest efficient syntheses of.
A:
Q: Write suitable detailed mechanisms to account for the formation of the products in the following…
A:
Q: When pent-2-yne reacts with mercuric sulfate in dilute sulfuric acid, the product is amixture of two…
A:
Q: Give the reagents and conditions necessary for the following conversion. A to B, B to D, B to C, B…
A: Compound (A) is ethene, which gives Bromo Ethane ,(B) with Hydrogen Bromide as - CH2=CH2 + HBr…
Q: Outline a synthesis for m-dichlorobenzene from benzene using diazonium salts as intermediates.
A:
Q: Show by writing a suitable series of steps of equations how you would prepare 1-Hexene from 1-butene…
A: The structure for 1-butene and acetylene is represented as follows:
Q: An unknown hydrocarbon G, whose formula is C16H26 contains two triple bonds. Ozonolysis followed by…
A: Ozonolysis: Ozonolysis is an organic reaction where the unsaturated bonds of alkenes, alkynes,…
Q: (c) Write the products and comment on the natures of the following reactions :
A: organometallics reaction
Q: Your research project involves the synthesis of a family of organometallic reagents so you can…
A: The compound mentioned is a grignard reagent. To synthesize a grignard reagent we have to be very…
Q: Sodium nitrite (NaNO2) reacted with 2-iodooctane to give a mixture of two constitutionally isomeric…
A: Given that, 2-iodooctane reacts with sodium nitrite (NaNO2) to give a mixture of two…
Q: 1. NaNH2/sivi NH3/-78°C 1. NaNH2/sivi NH3/-78°C A( C9H16) B(C19H36) H-C=C-H 2. 1-Bromo-5-metil…
A:
Q: Propose, how to obtain the following alkene in the Wittig reactions, starting from alkyl halide and…
A: The Wittig reaction, also known as Wittig olefination, is a chemical reaction that occurs when an…
Q: Outline the synthesis of the following compound using only acetylene and/or simple alcohols.…
A:
Q: Represent the reaction equation of the alkene supplied to you with propanol, in acidic medium,…
A: In this question, we will draw all the products with stereochemistry when this above alkene reacting…
Q: To meet the ever increasing demand of global population, the demand for textile products and…
A: When a color-producing substance is involved in bonding with a material on which it is applied and…
Q: Name, draw and describe the organic product of the reaction between 2-methylbut-1-ene and H2O in the…
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Write suitable detailed mechanisms to account for the formation of the products in
A: AIBN (Azobis isobutyro nitrile) act as initiator Tri n butyl Tin hydride = Bu3S n H Free radical…
Q: The German chemist Wilhelm Kӧrner (1839-1925) observed in 1974 that each of the three isomers of…
A: The three isomers of dibromobenzene can be drawn as follows-
Q: In the reactions given below, write to which organic molecule Y, Z, T, A ‘belongs, and give…
A: Description: Concept introduction: a) Williamson ether synthesis: asymmetric ether can be prepared…
Q: Outline a mechanism for the dehydration of 2-methylcyclohexanol catalyzed by phosphoric acid.…
A: Mechanism : Dehydration of alcohol under acidic condition follows E1 reaction mechanism. It is…
Q: A task is assigned to an undergraduate student to test two samples (known as compounds K and L) in…
A: When treated with 2,4-dinitrophenylhydrazine, all of these compounds produce a brightly coloured…
Q: Suggest short, efficient reaction sequences suitable to produce the compound from the given starting…
A: Benzyl methyl ether can be prepared by the reaction of benzaldehyde dimethyl acetal and…
Q: Give a proposal on the synthesis methods of CoSO4.7H2O
A: Cobalt (II) sulphate can be synthesized by the reaction of cobalt metal with aqueous solution of…
Q: Deduce the identity of the compound ? Explain
A: Permanganate solution is used as oxidation agent in chemistry.
Q: . The reaction of benzene with produces principally which of these?| in the presence of anhydrous…
A: We have to predict the product from the reaction with anhy. AlCl3.
Q: (a) (b) (b) The following reactions as written will NOT give the indicated product. For each of the…
A: a. (i) and (iii)Reaction does not give the given product if the given reagents are used as per…
Q: (a) suggest the suitable mechanism and illustrate the stepwise of the (heptyloxy)cyclopentane…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Outline the synthesis of racemic 3-methyl-3-heptanol, Et(Me)C(*)OHn-Bu, starting from alcohols of…
A: Using a 4 or less membered alcohol, we have to form 3-methyl 3-heptanol.
Q: Predict the products of reactions of carboxylic acids with alcohols, amines, diazomethane,reducing…
A: Since you have posted multiple questions and have not specified which part you want us to solve, we…
Q: Predict the epoxidation products of the rigid cyclic alkenes (I, II) and examine the reasons for the…
A: The reaction in which alkene is converted into epoxide in presence of oxidizing agent is known as…
Q: OH then
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out…
Q: Suggest a method of preparing ethylbenzene, starting with benzene and ethylene as the only organic…
A:
Q: To
A: Introduction: In a given conversion reaction, firstly bromination is done of pentanol at second…
Q: The relative reactivity of the 1°: 2°: 3° hydrogens of (CH3)3CCH2CH3 in free radical chlorination is…
A: The reaction for the above process is given below :
Q: 9. The reaction of benzene with principally which of these? in the presence of anhydrous aluminum…
A: The given reaction is an example of Friedel-crafts reaction in which AlCl3 is used as a strong Lewis…
Q: Give the structure of the product and/or intermediates of the following reactions. Indicate, where…
A: Ozonolysis of alkene: One of the important reactions of alkene is ozonolysis. In the ozonolysis…
Q: Outline the step-by-step method (initiation, propagation(s), and one termination step) for…
A:
Q: Predict the coupling products of organometallic substitutions, and use them in syntheses.
A: The chemical compounds that has one or more bond between carbon and a metal element which is…
Q: Assuming you are a chemist and you need to extract ethanolic acid in a benzene solution. Propose and…
A: A salt of carboxylic acid is more soluble in water. So, a wash with NaOH converts the ethanoic acid…
Q: 4) (a) Provide a series of synthetic steps to synthesize 2-bromo-3-nitrobenzoic acid from toluene.…
A: Aromatic compounds undergo electrophilic substitution reaction. Incase benzene has electron donating…
Q: 3. Outline a synthesis for m-dichlorobenzene from benzene using diazonium salts as intermediates.
A: We have to synthesise m- dichlorobenzene from benzene
Q: The relative reactivity of the 1°: 2°: 3° hydrogens of (CH3)3CCH2CH3 in free radical chlorination is…
A:
Q: A compound with the molecular formula, C,,H0 was treated with Wolff-Kishner reagent and the product,…
A:
Q: In two parts, outline the Electrophilic aromatic substitution for the nitration of benzene using…
A: Nitration of benzene involves following three steps 1. Generation of Electrophile (Nitronium ion) 2.…
Q: Give a clear handwritten answer..and give all possible products
A: Claisen-Schmidt condensation: Aromatic aldehyde/ketone without alpha hydrogens are treated with…
- What are the names of the compound A, B, C, D, E, F, G & H. Hence suggest structures for compound C and E.
- What are the reagents and reaction conditions used for the following conversions? (i), (ii), (iii), (iv) & (v)
Step by step
Solved in 2 steps with 1 images

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?.771g of acetominophen reacts with 1.0 eqivalent of 1.5M KOH and 1.2 equivalents of pure liquid n-butyl bromide in ethanol solvent to give .901g of the willimason ether product a) calculate the volume of the base and the volume of the SN2 electrophile needed for the reaction needed for the reaction using the equivalents given above b) caclulate the percent yield of the reaction
- What happens when (write reactions involved) iv and vWhy cis-Ru(II)Cl2(DMSO)4 reacts with pyridine, et cetera, to give substitution of the DMSO but not the chloride ligands, but trans-Ru(II)Cl2(py)4 react with suitable Na+ and K+ salts in aqueous pyridine to afford chloride-substituted derivatives. write the reactions equations.Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…
- Rank the species below in order of increasing nucleophilicity in protic solvent. I. H2O II. CH3S— III. CH3COO— IV. t-BuO— I, II, IV, III I, III, II, IV I, III, IV, II I, II, III, IVThe sex attractant of the common housefly is a hydrocarbon named muscalure, C23H46. On treatment of the muscalure with aqueous acidic KMnO4, two products are obtained, CH3(CH2)12CO2H and CH3(CH2)7CO2H. Propose a structure for the muscalure. Please provide a full explanation with steps when providing the structure as a response, thank you!What are the volume (uL), moles, and/or weight (mg) of each reagent and solvent in:50 mg of the “Bromo” starting material (1 equivalent), acid catalyst (if any, 0.1 eq) and in 0.25 M of solvent?
- I need help to find the mechansim for Preparation of Cyclohexanone by Hypochlorite Oxidation and explain the mechanisms step by step so I can understand it pleaseWhen pyrrole is added to a dilute solution of D2SO4 in D2O, 2-deuteriopyrrole is formed. Propose a mecha-nism to account for the formation of this compound.Pls solve this question correctly in 5 min i will give u like for sure Balance the chemical equation of a reaction formed by the bromination of trans-cinnamic acid if we started with 0.3088 grams of trans cinnamic acid and 1mL of bromine. The product of this reaction is 2,3 - dibromo - 3- phenylpropanoic acid

