Q: 5- Predict the products of the following reactions NH₂ a- + HCI b- CH3COOH + NHẠCH, + HCI - ин сиз…
A: Aniline react with HCl to form anilinium Chloride. Pyridine reacts with HCl to form pyridinium…
Q: Given the galvanic cell below, which letter represents the electrode that is positive? с D Zn(s)→…
A: In the electrochemical cell, the left side electrode is anode and the right side electrode is the…
Q: What is the water table in terms of groundwater and what role does groundwater play in karst…
A: Introduction: The water table represents the boundary between saturated and unsaturated zones in an…
Q: Define the point group for the following molecules. Draw out and label the operations ON the…
A: A Point Group describes all the symmetry operations that can be performed on a molecule that result…
Q: Gas Stoichiometry Scenario #3 What mass (in grams) of sodium chloride (NaCl) will be produced when…
A: The given unbalanced reaction is as follows: Na+Cl2 → NaCl The balanced equation is as follows:…
Q: 2. Given: 2. MOLAR MASS NUMBER OF MOLES MASS PRODUCTS C₂H4 5g + Which is the limiting reactant?…
A: As per our company guidelines we are supposed to answer only first three sub-parts. Kindly re-post…
Q: Aromatic compounds generally undergo substitution reactions. Substitution of a halogen, such as…
A:
Q: O Macmillan Learning Determine the hybridization and geometry around the indicated carbon atoms. A B…
A: Hybridization is defined as the intermixing of atomic orbitals with the same energy levels to give…
Q: Complete and balance the chemical equation
A: Balanced chemical equation can be define as the reaction in which number of atoms of all the…
Q: If a student decreased the temperature for this reaction: C₂H4(g) + H₂(g) - heat C₂H6(g) what would…
A: This principle is used to predict the effect of a change in conditions on a chemical equilibrium.…
Q: Use the molecular orbital diagram shown to determine which of the following is most stable.
A: Bond Order of a species is determined by the following formula : BO = (No. of bonding electrons -…
Q: You will find it useful to keep in mind that HF is a weak acid. 0.52 mol of KOH is added to 1.0 L of…
A: Given,moles of KOH =0.52 molMolarity of HF = 1.3 MVolume of solution = 1.0 Lmoles of HBr =0.31…
Q: Which of the following is the most acidic?(assume that Ha is the hydrogen that will be released)…
A: In a cyclic conjugated system, the hydrogen atom attached to sp3 hybridized carbon will be more…
Q: (SHOW CALCULATIONS) The mass of a sample is 8.2 x 10-2 milligrams. Express that mass in kilograms…
A:
Q: The density of helium at standard temperature and pressure is 0.166 g/L. What volume in liters would…
A: Density = 0.166 g/L Weight of He = 3100 mg = 3.100 g (1g = 1000mg)
Q: -- atm 13, At 25 °C and 1 atm of CO2 pressure, the solubility of CO2 (g) is 149 mg CO2/100 g water.…
A: Given,
Q: How does the molecular structure of a compound affect its chemical reactivity and reaction rate?
A: The molecular structure of a compound is a key determinant of its chemical reactivity and reaction…
Q: The weak acid HZ has a Ka of 2.55x10-4. (a) Calculate the pH of 0.075 M HZ. (b) Calculate the pOH of…
A: Since you have asked multiple questions, we will answer only the first question for you. In order to…
Q: Match each of the following chemical equations to the type of chemical reaction it describes:…
A:
Q: Nr Table 1 shows the relative viscosity, nr for a polyisobutylene with the molecular weight of…
A: To calculate the intrinsic viscosity of a polymer sample having a molecular weight of 1500000 g/mol…
Q: Determine the number of bonding electrons and the number of nonbonding electrons in the structure…
A: Lewis Structure represent how the electrons are arranged around the atoms in a molecule.
Q: An aqueous solution of a nonionic (that is, nonelectrolyte) solute has a freezing point of -0.332…
A: Given,Mass of solute = 1.79 gMass of water = 64.3 gfreezing point of the solution = -0.332°C
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A: Given,The aqueous solutions:1. 0.137 m Mg(CH3COO)22. 0.117 m Mn(NO3)2 3. 0.190 m CoSO44 0.320 m…
Q: Complete the biosynthetic pathway for the synthesis of Cadinene. Hint: Cyclize to form a ten…
A: Cadinene (C15H24) are bicyclic sesquiterpenoids that are the main component of oils found in cubeb…
Q: A sample of zirconium experiences a temperature change of +790.5 K while absorbing 150820 joules of…
A: The change in temperature (∆T) = + 790.5 K The heat (q) = 150820 J The specific heat capacity (c) =…
Q: a) This alkene can be prepared via Wittig reaction. Draw structural formulas for the aldehyde and…
A: A question based on reactions of aldehyde. Structure of a product alkene is given whose aldehyde…
Q: Compare the 13C NMR spectra of isoborneol and camphor. Explain which peaks can be used to determine…
A: 13 C NMR spectroscopy is useful to determine types of carbons, their signals, splitting etc hence we…
Q: Name the given lipid molecule below:
A: Lipids are biomolecules that are insoluble in water and are comparably nonpolar. These are…
Q: You are asked to make 43.0 grams of iron (Fe) from iron III oxide (Fe-O:) and carbon monoxide (CO)…
A: A question based on mole concept. A balanced equation is given based on which mass of Iron is to be…
Q: 1. A hydrogen bond is a special case of which intermolecular force? Provide some examples of…
A: To provide the type of interaction of which the hydrogen bonding is a special case and also provide…
Q: Consider the following acid-catalyzed hydration reaction: Which of the following ions are…
A:
Q: In the acid-catalyzed dehydration of 2-methyl-1-propanol, what carbocation would be formed if a…
A: Acid catalyzed dehydration of 2 -methyl -1 propanol
Q: What orbital is described by the following quantum numbers? n = 4 1 = 3 m₁ = -4
A: n is principal quantum number which represents the main energy level occupied by the electron. The…
Q: 3. Chloroacetic acid, CICH₂COOH, has a pKa of 2.87. What are [H3O+], pH, [CICH₂COO-], and…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: A 150. g sample of KCl is dissolved in enough water to make 1.00 liter of solution. How many…
A: Mass of KCl = 150 g Molar mass of KCl = 74.55 g/mol Moles of KCl = mass / molar mass…
Q: Which of the following structures will give a base peak of 43 in mass spectrometry? CH3 оснас O…
A:
Q: If deuterated borane were used instead of borane in the hydroboration oxidation of the alkene shown…
A: A question based on reactions of alkenes. An alkene reactant is given product of deuterated…
Q: See Periodic Table See Hint A weather balloon with a volume of 151 L is launched at 20.0°C at sea…
A:
Q: When classifying chemical reactions, one can use a couple of key features for the reaction types.…
A: Chemical reactions can be classified into 5 types: Combination reaction- reaction in which 2 or…
Q: Draw the structure of the major organic product(s) of the reaction. OH + • You do not have to…
A: Anhydrides react with alcohols to form esters and Carboxylic acids.
Q: Flowchart the separation of hexane and butyric acid.
A: This flowchart outlines the basic steps involved in separating hexane and butyric acid. The…
Q: 3. The graph demonstrates the viscosity or thickness of a liquid and how it is affected by…
A:
Q: 30. The electrical potential for the * reaction between nitric acid and copper, under standard…
A: The electrical potential for the reaction between nitric acid and copper, under standard conditions,…
Q: Consider the following elementary reaction: N₂O(g) →N₂(g) + O(g) Suppose we let k₁ stand for the…
A: The rate constant of the reaction = k1 The rate constant of the reverse reaction = k-1 We have to…
Q: 3. An unknown compound 'A' with the formula C4H6 reacts with excess Br2 at room temperature to give…
A: The question is based on the concept of organic reactions. we need to identify the compounds.
Q: #p+ #e- #p+ #e- Fe (s) + Cu²+ (aq) Fe²+ (aq) + Cu (s) -> Fe² (?) + --> Fe? Fe? (?) + Cu² (?) Cu? (?)…
A: A question based on atoms. Two electrochemical reactions are given the details of number of protons…
Q: How does the coordination geometry of a metal center in a bioinorganic system affect its reactivity…
A: The coordination geometry of a metal center in a bioinorganic system plays a crucial role in…
Q: Consider the species present in a 1.0×10 M solution of HCIO(aq). Place the species in order of their…
A: Concentration of HCIO =1.0×10-4 M Ka(for HCIO]=2.9×10-8
Q: Which metal ion (Cu²+, Ni2+, Fe³+) had the largest surface area? Suggest a reason why?
A: A question based on atoms. 3 ions are given whose surface area are to be compared and explained with…
Q: How does the nature of the ligand binding to the iron ion in heme complexes affect the oxygen…
A: Introduction: The relationship between ligand binding and oxygen affinity in bioinorganic chemistry…
Step by step
Solved in 4 steps with 2 images

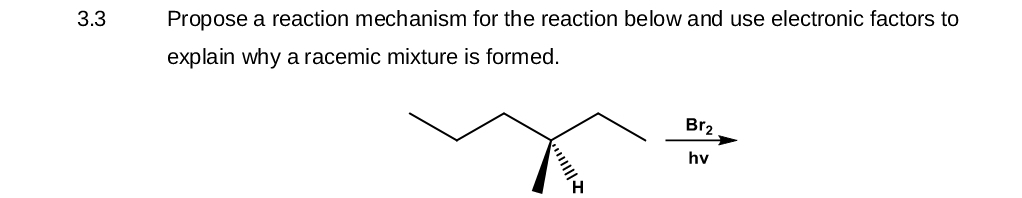
- Please show the entire mechanism with curved arrows for the reaction. Why is the reaction considered unfavorable in reference to the stability ladder? Why does the reaction favor the product formation? *please show the resonance structures in the reaction.*what is the rate at which Br- (aq) disppears in reaction below if the rate of disappearence of BrO3- (aq) is 0.022 M/swhat is a possible mechanism for the following reaction ??
- Step 4c: Draw the resulting structure or structures indicated by the curved‑arrow mechanism shown. Include formal charges, but not lone pairs.Following is a balanced equation for bromination of toluene.(a) Using the values for bond dissociation enthalpies given in Appendix 3,calculate ∆H0for this reaction.(b) Propose a pair of chain propagation steps and show that they add up to theobserved reaction.(c) Calculate ∆H0for each chain propagation step.(d) Which propagation step is rate-determininRank the reactivity of these below: most reactive (1) and least reactive (3)
- Arrange the following according to INCREASING reactivity towards E2: 1st ( least reactive)? 2nd? 3rd? 4th (most reactive)?Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2OCan you do 3 and 4 and explain it? And how yo identify it. State the mechanism in which through reaction proceeds sn2 or sn1
- What carbon radical is formed by homolysis of the C–Ha bond inpropylbenzene? Draw all reasonable resonance structures for thisradical.i) Ph2PH ii) Nal iii) PhCH2OH iv) CH3SH v) Pr2CuLi At what letter in the picture will each above subtrate react on? (A/B/C)Which reaction is faster, one with Eact = +41.8 kJ/mol or one with Eact = +20.9 kJ>mol? Explain.

