Q: Write the balanced net ionic equation for the reaction that occurs in the following case:…
A:
Q: (Q70) Using the tabulated values from the text (located in Appendix G, beginning at p. 1211)…
A: In the given question we have to calculate the standard gibs free energy (∆G°rxn) of the reaction.…
Q: Answer all questions 1. It has been announced on radio that a toxic gas is spreading towards UENR…
A: The solution is given below -
Q: Select all answers that show the atoms or ions that are isoelectronic with each other Ge, Br3+ He,…
A: Isoelectronic species: Isoelectronic species can be define as the atoms or ions having the same…
Q: a. Provide the structural formula of each product for the reaction shown below: Et N H₂C OH b.…
A: To give: a) structural formula of product b) curved arrow mechanism
Q: Explain why p-nitrophenol (the left molecule) is more acidic than p-methylphenol (the right…
A: Why p-nitrophenol is more acidic than p-methylphenol ?
Q: What is the mechanism of the phosphate buffer action? Write down the equations of these reactions.
A:
Q: Tube No. 1 2 3 4 5 Substance Glycerol Cottonseed c Olive oil Walnut lip Water
A: Fats are solid at Room Temperature but fats are liquid at room temperature.
Q: 1. H₂CRO4 2.NH, CH3 1. Swern 2. 2 mol EtOH OH 1. H₂CRO4 2. CH₂CH₂CH₂Br. PPh3, KOA 1. KMnO4 2.NH, CH3
A: Given are organic reactions
Q: Gas Theory Master Select all of the following that are true: At constant pressure, the volume of a…
A: At constant pressure volume of a gas is directly proportional to the temperature of the gas. Gas…
Q: A chemist fills a reaction vessel with 0.999 g calcium phosphate (Ca3(PO4)2) solid, 0.689 M calcium…
A:
Q: г? НО
A:
Q: What is the cell potential for the following cell at 25°C? Al ∣ Al3+(0.00212 M) Sn2+(0.435 M) ∣ Sn…
A:
Q: Given the following balanced equation, if the rate of Cl2 disappearance is 4.84 × 10-2 M/s, what is…
A: Given : 2 NO(g) + Cl2(g) ---> 2 NOCl(g) rate of Cl2 disappearance = 4.84 × 10-2 M/s To…
Q: Arrange the following elements in order of increasing atomic radius. Al, S, Mg, Si, Na (a) Mg<S<Si…
A:
Q: H₂C Which structures below represent the diastereomers of the compound on the picture? CH₁ B H₂C H₂C…
A: In stereochemistry, diastereomers are a type of stereoisomer. Diastereomers are defined as…
Q: Give the IUPAC name for each compound.
A:
Q: (Q66). What is the change in entropy (in J/K) when 14.1 g of liquid hexane boils at its normal…
A: ∆Suniverse = ∆Ssystem + ∆Ssurrounding . And ∆Ssurrounding = - ∆Hreaction / T.
Q: change 18h Dry nswer contains a unit symbol. Round your answer to 3 significant digits. μ ロ・ロ X 010…
A: Given, □Hfus = 4.39 kJ= 4390 J T = -112 K = 161 K Mass of CS2 = 400 g Molar mass = 76 g/mol Mole…
Q: 10.00 mL of 0.45 M NaOH is added to 9.00 mL of 1.00 M HCOOH. What is the molar concentration of…
A:
Q: student is asked to prepare methyl salicylate from salicylic acid and methanol. How would the…
A: In this question, we have to answer how would the student will prepare methyl salicylate from…
Q: This is an impossible formula. If it were real, determine the molar mass of:
A: To find Molecular mass for a hypothetical compound.
Q: Write the appropriate reagents, conditions and products for the following electrophilic aromatic…
A: Write the appropriate reagents, conditions and products for the following electrophilic aromatic…
Q: Molecules Nitrogen trifluoride Carbon diselenide Aluminum triiodide Lewis structure Dots Lone pair…
A: The Shape is determined by the central atom and its valence electron.
Q: Free Response Questions in this section are not weighted equally. Allocate your not (11) gepo to…
A:
Q: Provide the major product(s) for each of the following reactions. a. NH₂ pyridine b. 1. excess CH3l…
A: These Reactions are categorized as Nucleophilic substitution Reaction , reduction reactions .
Q: An experiment was conducted to determine the over-all order of the reaction: YOI → MIYA At the start…
A: Given: Run Initial concentration [YOI]o M Final concentration [YOI]o M Time (seconds) 1 5.142…
Q: The following compound may undergo the Dieckmann condensation reaction (an intramolecular Claisen…
A:
Q: A galvanic cell is composed of these two half-cells, with the standard reduction potentials shown:…
A:
Q: The equilibrium constant, Kc, was found to be 4.1 x108 at 25°C for the following reaction, 2X(s) +…
A: At equilibrium Ecell = 0. And Q = Kc . We can calculate the E°cell using the nearst equation.
Q: b) For two of the carboxylic acids, do an ARIO analysis to explain their relative acidities.…
A:
Q: Which of the Newman projections below would correspond to the following molecule from the…
A: In this question we have to tell the Newman projection of the given molecule.
Q: two functional group isomers having the molecular formula: C4H8O
A: Functional group isomers have same molecular mass but different molecular formula.
Q: What would be the pH of 0.00551 M H2SO4 solution?
A:
Q: Devise a synthesis for the following transformation. SM = starting material. TM = target molecule Br…
A: Here we have to synthesize the following target material from the given starting material from…
Q: How much heat is needed to convert 36.5g of ice at 0°C to water at 66°C? ΔHfus of ice = 6.01 kJ/mol…
A: given, mass of Ice = 36.5 g Initial temperature (Ti) = 0°C Final temperature (Tf) = 66°C Note: molar…
Q: A sample of methane gas collected at STP occupies a volume of 16.0 L. How many moles of gas does the…
A:
Q: Calculate the vapor pressure of a solution prepared by dissolving 90.0 g of naphthalene, C10H8 (a…
A:
Q: To do: Identify the oxidizing agent and reducing agent in the reactions. 1. 2Na2S2O3+/2→…
A: Here we have to predict the oxidising agent and reducing agent in the following redox reactions.
Q: Which neutral atom is isoelectronic with each of the following ions: F, Zr, Mn, I, Pb²+? Express…
A: Which neutral atom is isoelectronic with the given ion---
Q: Click in the answer box to activate the palette. Balance the following equation for the reaction in…
A: Given that, Fe2+ + MnO4- ----> Fe3+ + Mn2+ Balanced the reaction under acidic condition ?
Q: #2
A: You isolated 4.39 grams of aspirin analogue. What was your percent yield. Show ALL calculations ...
Q: 5.7 mL of 0.1 mol/L HCI solution was added to 10 mL of buffer solution at pH 4.0. Calculate the…
A:
Q: The Robinson annulation reaction involves Michael addition and aldol condensation and it is a ring…
A:
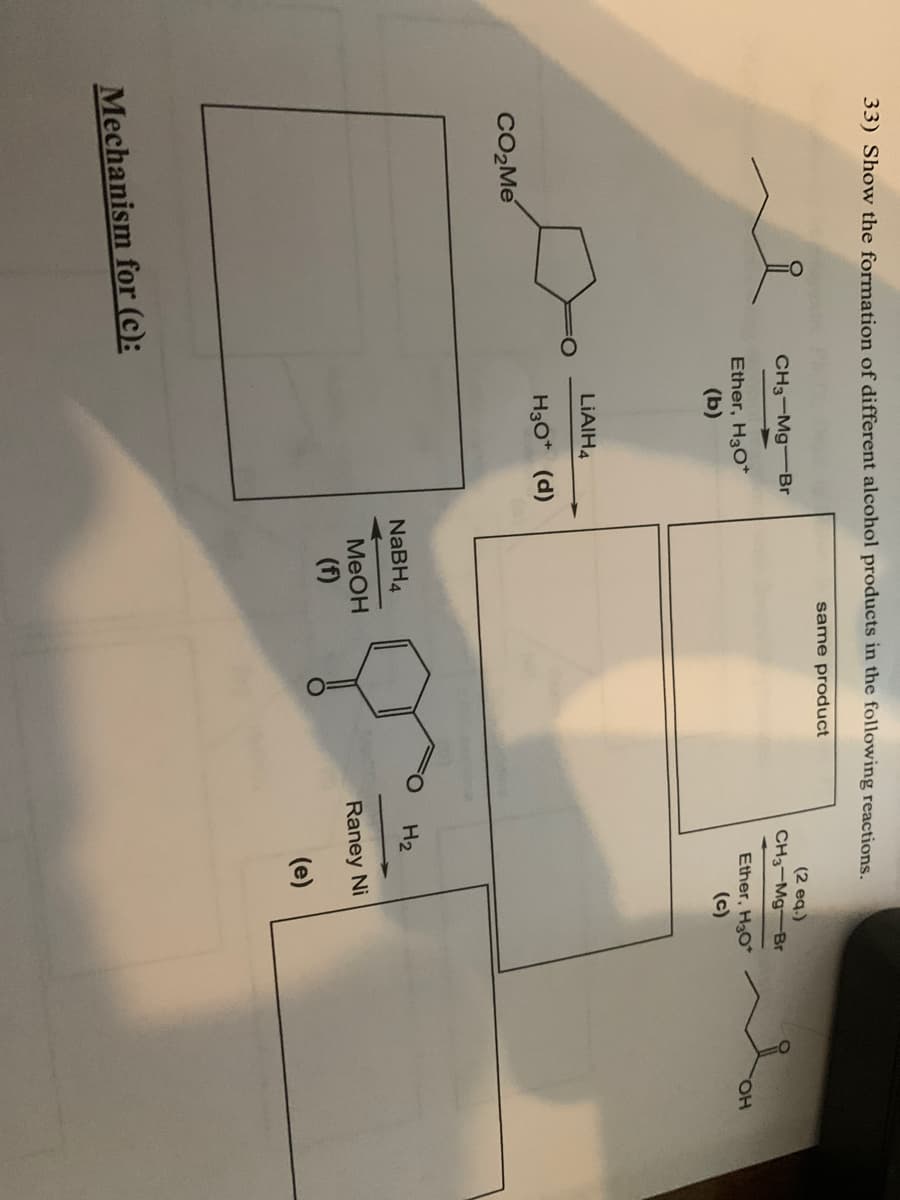
Q: ve the major products for each of the following reactions OH PCC CH₂Cl2 CrO3, H₂SO4 1. LiAlH4, THF…
A:
Q: Propose a synthesis for the peptide leu-lys-cys
A: Detail synthetic route is given below
Q: Calculate the potential (E) in V for the following reaction at 25oC: 2X(s) + 3Z2+(aq, 2.24M) ⟶…
A:
Q: If ΔH° = 280 kJ/mol and ΔS° = 258 J/mol · K, above (>) or below (' (greater than) or '67)
A:
Q: Give the major elimination or substitution products and indicate the reaction mechanisms (SN1, SN2,…
A: We can decide the outcome and the mechanistic pathways for the given reactions based on the nature…
Q: The decomposition of N₂O5 proceeds according to the following equation: 2N2O5 (g) →→ 4NO2(g) + O₂(g)…
A:
Step by step
Solved in 2 steps with 1 images

- The Favorskiireaction involves treatment of an a-bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclohexanone with aqueous NaOH yields cyclopentanecarboxylic acid.what is the mechanism of the reaction.Provide a mechanism for the following isomerization in aqueous acid. Thank you in advance!Provide a stepwise synthesis for benzyl chloride to benzyl alcohol with reagents Na2CO3 and H2O (including arrow-pushing mechanisms) Kindly show all the arrow push
- Devise a synthesis of the ketone hexan-3-one, CH3CH2COCH2CH2CH3, from CH3CH2Br as the only organic starting material; that is, all the carbon atoms in hexan-3-one must come from CH3CH2Br. You may use any other neededreagents.a) Write out the first 3 steps only(to the tetrahedral intermediate shown) in the 6-step arrow pushing mechanism showing how ethyl propanoate is hydrolyzed in acid to form propanoic acidand ethanol. b) NaOH/H2O also serveto hydrolyze an ester; briefly explain why NaOH/H2Ois generally preferable.Compounds A, B, and C have the same molecular formula C4H8. They all react wotu H2/PtO2 to give the same compound. The reaction of A or B with H2O/H2SO4 or with BH3-THF, followed by treatment with a basic solution of hydrogen peroxide, gives the same compound, namely D. The reaction of C with H2O/H2SO4 also gives D. However, the reaction of C with BH3-THF, followed by HO-, H2O2 gives a new compound, E. Provide the identity of A, B, C, D, and E along with explanations of reactivity.
- Provide a stepwise synthesis for benzyl alcohol to sodium benzoate with reagents KMnO4 and NaOH (including arrow-pushing mechanisms) Please show all the arrow push with the given reagents above.Capsaicin, the spicy component of hot peppers, can be prepared from amine X and acid chloride Y. Devise a synthesis of Y from (E)-6-methylhept-4-en-1-ol [(CH3)2CHCH=CH(CH2)3OH], CH2(CO2Et)2, and any required inorganic reagents.The analgesic acetaminophen is synthesized by treating 4-aminophenol with one equivalent of acetic anhydride. Complete the equation for the formation of acetaminophen. a. Acetaminophen: b. Additional Product pls add the name for both, thanks!
- Sharpless epoxidation of allylic alcohol X forms compound Y. Treatmentof Y with NaOH and C6H5SH in an alcohol–water mixture forms Z. Identifythe structure of Y and draw a mechanism for the conversion of Y to Z.Account for the stereochemistry of the stereogenic centers in Z. Z hasbeen used as an intermediate in the synthesis of chiral carbohydrates.Compound A (C9H12) absorbed 3 equivalents of H2 on catalytic reduction over apalladium catalyst to give B (C9H18). On ozonolysis, compound A produced 3 products, C, Dand E which was a ketone identified as cyclohexanone. On treatment with NaNH2 in NH3,followed by addition of iodomethane, compound A gave new hydrocarbon, F (C10H14). Drawstructures of A, B, C, D and F.An optically active alkyne A (C10H14) can be catalytically hydrogenated to butylcyclohexane. Treatment of A with EtMgBr liberates no gas. Catalytic hydrogenation of A over Pd/C in the presence of quinoline poison and treatment of the product with O3 and then H2O2 gives an optically active tricarboxylic acid C8H12O6 (A tricarboxylic acids is a compound with three - CO2H groups.). Give the structure of A, and account for all observations.



