(4) For the reaction below, the reaction is said to be: A +B →C AH: = +30 kJ, AS = +50 J/K spontaneous at all temperatures nonspontaneous at all temperatures spontaneous at higher temperature spontaneous at lower temperature Clear selection (5) Which statement is incorrect? At constant pressure, deltaH = deltaE + P(deltaV) For an endothermic process, deltaH is negative. O Gibbs free energy is a state function. When the work done by the system is greater than the heat absorbed by the system, deltaE is negative
(4) For the reaction below, the reaction is said to be: A +B →C AH: = +30 kJ, AS = +50 J/K spontaneous at all temperatures nonspontaneous at all temperatures spontaneous at higher temperature spontaneous at lower temperature Clear selection (5) Which statement is incorrect? At constant pressure, deltaH = deltaE + P(deltaV) For an endothermic process, deltaH is negative. O Gibbs free energy is a state function. When the work done by the system is greater than the heat absorbed by the system, deltaE is negative
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter16: Thermodynamics: Directionality Of Chemical Reactions
Section: Chapter Questions
Problem 89QRT: Determine the standard Gibbs free energy change, rG, for the reactions of liquid methanol, of CO(g),...
Related questions
Question
100%
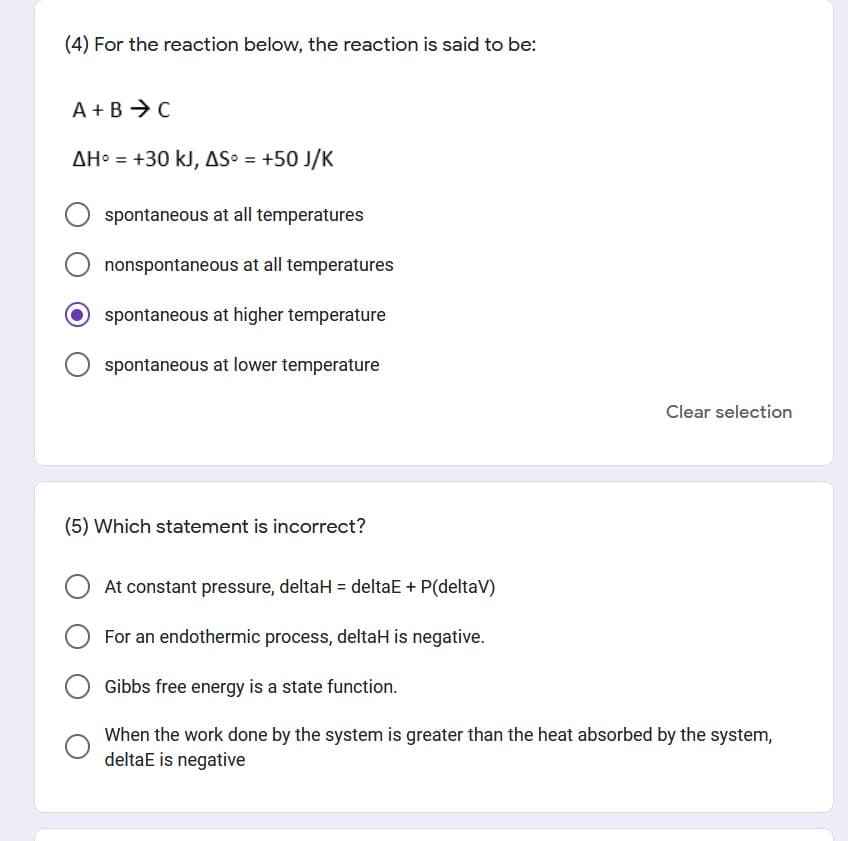
Transcribed Image Text:(4) For the reaction below, the reaction is said to be:
A +B → C
AH = +30 kJ, AS = +50 J/K
spontaneous at all temperatures
nonspontaneous at all temperatures
O spontaneous at higher temperature
spontaneous at lower temperature
Clear selection
(5) Which statement is incorrect?
At constant pressure, deltaH = deltaE + P(deltaV)
For an endothermic process, deltaH is negative.
Gibbs free energy is a state function.
When the work done by the system is greater than the heat absorbed by the system,
deltaE is negative
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning