Q: Why is the standard entropy of a substance in the gas state greater than its standard entropy in the…
A: The intermolecular force between gas molecules is less than the intermolecular force between liquid…
Q: A reaction has ΔH∘rxn= -134 kJ and ΔS∘rxn= 320 J/K. At what temperature is the change in entropy…
A: Given: -ΔH°rxn = -134 KJΔS°rxn = 320 J/KIf everything is carried out at a constant temperature and…
Q: Ca(OH)2(aq) + 2HCl(aq)CaCl2(s) + 2H2O(l) for which H° = -30.20 kJ and S° = 205.9 J/K at 298.15 K.…
A:
Q: methanol, CH3OH, vaporizes at 25°C is 35.4 kJ/mol. What is the entropy change (in J/K) when one mole…
A: Given, □Hvap = 35.4 kJ/mol T = 25 °c = 298 K □S = □H/T = 35.4 ×1000/298 = 118.79 J/K-mol
Q: Cyclopropane and propylene are isomers that both have the formula C3H6. Based on the molecular…
A:
Q: che standard entropy change to c
A:
Q: 8) For which of the following will the entropy of the system increase? A) reaction of magnesium with…
A:
Q: A particular reaction is spontaneous at 450 K. The enthalpy change for the reaction is +34.5 kJ.…
A: Given : Enthalpy change of reaction, ΔH = +34.5 KJ = 34500 J…
Q: Consider the reaction 6CO2(g) + 6H2O(l)C6H12O6 + 6O2(g) for which H° = 2.801×103 kJ and S° =…
A: The given reaction is, 6CO2(g) + 6H2O(l) → C6H12O6 + 6O2(g) Given that: H° = 2.801×103 kJ S° =…
Q: Explain how the entropy of a system is related to the number ofpossible microstates?
A: To find: The relation between the entropy and number of possible microstates
Q: Calculate the change in entropy that occurs in the system when 1.00 mole of isopropyl alcohol…
A: Given data Number of mole of isopropyl alcohol : n = 1.00 mole Boiling point of isopropyl alcohol :…
Q: 8. Predict the sign of the change in entropy for the condensation of steam to liquid water. 9.…
A: Entropy means randomness. The higher the randomness of the system, the higher will be its entropy.…
Q: 1. The heat capacity at 1 atm. pressure of solid magnesium in the temperature range from 0 °C to 560…
A:
Q: A reaction has ΔH °rxn = -112 kJ and ΔS °rxn = 354 J/K. At what temperature is the change in entropy…
A: The quantity of energy that is absorbed inside the system at a given temperature during a particular…
Q: What is the entropy when a system goes from 4 microstates to 5 microstates?
A:
Q: How can you calculate the standard entropy change for a reaction from tables of standard entropies?
A: The standard entropy change for a reaction from tables of standard entropies can be calculated by…
Q: The heat capacity at 1 atm. pressure of solid magnesium in the temperature range from 0°C to 560 °C,…
A: Since the units of T to be substituted in the Cp equation is not mentioned. Hence i'm assuming it to…
Q: 1A. The following 3 parts pertain to the entropy change for a sample of neon gas initially at 250 K…
A: 1B.Steps required to calculate the Entropy change: 1. First we have to put the entropy change…
Q: Under what circumstances does entropy of a system decrease for a spontaneous process
A: For any spontaneous process, the value of Gibbs free energy change i.e ΔG should be -ve And since…
Q: Consider the reaction 6CO2(g) + 6H2O(l)C6H12O6 + 6O2(g) for which H° = 2.801×103 kJ and S° =…
A:
Q: In freezing a mole of liquid water at the freezing point; the enthalpy of fusion is 6.0lkJ/mol…
A: The enthalpy of fusion is 6.01 KJ/mol To determine : entropy of system , surrounding and universe
Q: As shown here, one type of computer keyboard cleaner contains liquefied 1,1-difluoroethane 1C2H4F22,…
A: The degree of randomness is called entropy. Entropy in the high-pressure region is high due to the…
Q: The enthalpy of vaporization of methanol (CH3OH) is 35.3 kJ/mol at the boiling point of 64.2 °C.…
A: The vaporization can be defined as phase transition from liquid phase to vapor phase.
Q: Calculate the standard entropy change for the following reaction at 25°C. CH,(g) + 2 O2(g)→ CO2(g)…
A:
Q: How many microstates are present in a system with an entropy of zero?
A: The entropy (S) and microstates(W) are related by formula : S = kB ln(W) Here , kB = Boltzmann…
Q: If an ideal gas at 1.75 atm and 25 degrees Celsius is isothermally compressed from 800 mL to 150 mL,…
A: Initial volume of the gas, V1 =800 mL Final volume, V2 = 150 mL Calculation of initial moles of the…
Q: How does the standard entropy of a substance depend on its molar mass? On its molecular complexity?
A: Entropy is a thermodynamic property, which is the measure of randomness of a system. Entropy of the…
Q: The entropy of vaporization for a certain organic compound is 28.1 J/Kmol, and the boiling point is…
A: given data entropy of vaporization 28.1 J/kmol boiling T=51.7 ;c find out enthalpy of vaporization
Q: What happens to the entropy of a sample of the matter when it changes state from a solid to a…
A: Entropy of a system is defined as the measure of randmnoss,or disorder. Entropy is an extensive…
Q: How do you predict the entropy of a gas will change if a 2.0 L sample of that gas is compressed to a…
A: The entropy of a system is defined as the measure of randomness between the particles present in a…
Q: 5. The entropy change of the process H2O (s) H2O () is 22 J/K at 0 °C. Calculate the enthalpy of…
A: H2O(s)↔H2O(l)∆H=22J/KT=0∘C=0+273K=273K∆S=∆HT
Q: What is the entropy of the process when 0.6831 moles of N 2 gas are compressed from 54.42 L to 28.11…
A:
Q: 2.1. For the isothermal expansion of a gas into a vacuum, (a) Is this a spontaneous process? (b)…
A: a. During this constant temperature or isothermal, expansion into a vacuum, the gas does no work (w…
Q: Does the entropy of the surroundings increase for spontaneous processes?
A: The entropy is a measure of randomness of a system. In a spontaneous process, the entropy of the…
Q: How does the entropy of ice at −5°C diff er, if at all, from its entropy at −50°C?
A: Entropy- Entropy is a thermodynamic property. Entropy determines tendency of process. Tendency means…
Q: Explain why changes in both enthalpy and entropy determine the spontaneity of a process.
A:
Q: A hot rock ejected from a volcano’s lava fountain cools from 1100C to 40.0C, and its entropy…
A: Given: Initial temperature, T1=1100°C=1373.15 K Final temperature, T2=40°C=313.15 K Decrease in…
Q: Explain in molecular terms why the entropy of a gas increases (a) with volume, (b) with temperature.
A: (a) The dispersion of energy and matter in the system indicates the entropy. By increasing volume…
Q: The enthalpy for a certain process is 146.4 kJ/mol and the entropy for the process is 463.6 J/K. At…
A:
Q: Consider the reaction Ca(OH)2(aq) + 2HCl(aq)CaCl2(s) + 2H2O(l) for which H° = -30.20 kJ and S° =…
A: Given reaction is : Ca(OH)2(aq) + 2 HCl(aq)→CaCl2(s) + 2 H2O(l) H° = -30.20 kJ S° = 205.9 J/K…
Q: What is the entropy of a system that has only a single microstate?
A: Entropy is defined in the terms of randomness that a system possess or the disordered state. Change…
Q: In the reactions: 2SO3(g) → 2SO2(g) + O2(g) and CaO(s) + CO2(g) → CaCO3(g), the entropy of the…
A: Balanced Reaction 2SO3(g) → 2SO2(g) + O2(g) Number of gas molecules reacts is 2 Number of gas…
Q: Consider the reaction 6CO2(g) + 6H2O(1)–C¢H12O6 + 602(g) for which AH° = 2.801×10° kJ and AS° =…
A: The given reaction is : 6CO2(g) + 6H2O(l) → C6H12O6 + 6O2(g)∆H°=2.801×103kJ and ∆S°=-259.0 J/K…
Q: Does the entropy of the system increase, decrease, or stay the same when the volume of a gas…
A: Entropy is the thermal energy of a system per unit of temperature. This energy is not available for…
Q: 1. The heat capacity at 1 atm. pressure of solid magnesium in the temperature range from 0 °C to 560…
A: Since in the question its not mentioned that in what unit the T is being substituted in Cp equation.…
Q: The entropy of vaporization of water is 109.0 J/mol · K. What is the enthalpy of vaporization of…
A: Given: Entropy of vaporization of water = 109.0 J/mol.K.
Q: Consider the reactionCa(OH)2(aq) + 2HCl(aq)CaCl2(s) + 2H2O(l)for which H° = -30.20 kJ and S° = 205.9…
A:
Step by step
Solved in 2 steps with 2 images

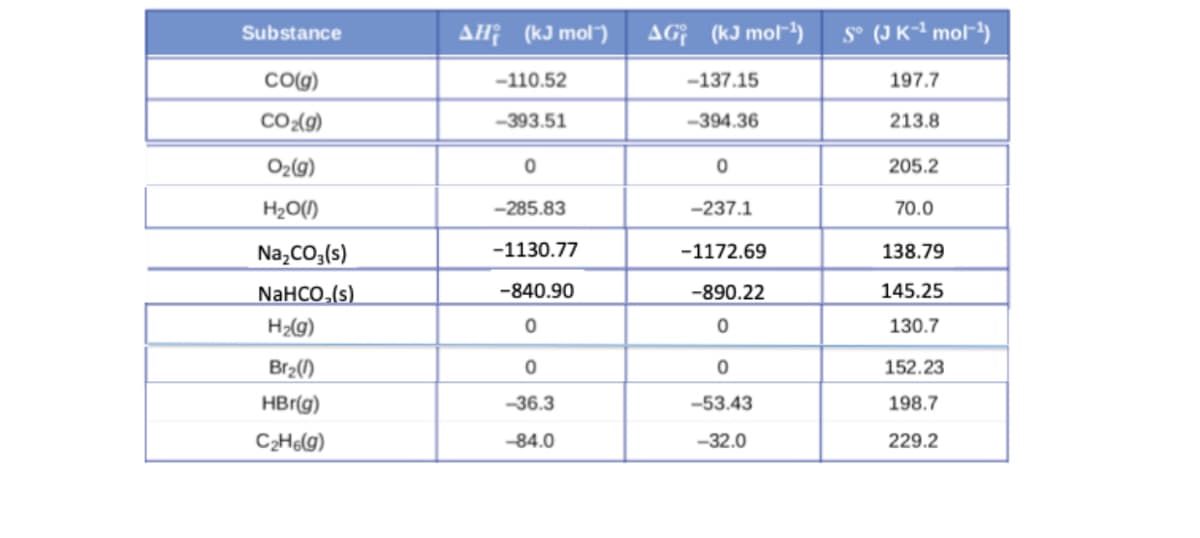
- Consider the reaction CO (g) + 2 H2 (g) = CH3OH (l)Thermodynamic Quantities for Substances at 25°C delta H°f (kJ/mol) delta G°f (kJ/mol) S°(J/mol*K) CO (g) -110.5 -137.2 197.5 H2 (g) 0 0 130.6 CH3OH -201.2 -161.9 238 17(a) Use the data above to calculate deltaH° and delta S° at 25°C. (17b) What is K at 25°C? (17c) What is delta G° at 1000°C?Thermodynamics Quantities for Selected Substances at 298.15 K (25⁰C) Substance ∆H⁰f (kJ/mol) ∆G⁰f (kJ/mol) S (J/K-mol) Carbon C2H2(g) 226.7 209.2 200.8 C2H4(g) 52.30 68.11 219.4 Hydrogen H2(g) 0 0 130.58 Oxygen O2(g) 0 0 205.0 H2O(l) -285.83 -237.13 69.91 What is the ∆S⁰ for the catalytic hydrogenation of acetylene to ethene, C2H2(g) + H2(g) → C2H4(g) inJ/K?Given below (c): Standard gibb's free energies (∆Gf0 kJ mol-1 ): UO2 = -962.7 UO22+ = -953.5 U4+ = -579.1 Fe2+ = -78.9 Fe(OH)3 ferrihydrite = -692.07 Mn2+ = -288.1 MnO2 pyrolusite = -465.1 HS- = 12.1 H+ = 0 H2O = -237.1 S0 = 0 Given: U(VI) as uraninite; UO2 (where Mn2+ = reductant; MnO2 pyrolusite = product): ∆ Gr0 = -21.3 KJ/mol E0 (emf) = 0.110 V n = 2 F = 96.42 QUESTION: Calculate Eh equation below to calculate at different pH: – Eh = E0 + (RT/nF) * lnK For U(VI) as uraninite; UO2 (where HS- = reductant; S0 = product): UO22+ + Hs- ---- > UO2 + S + H+ [A] pH 3 [B] pH 7
- Thermodynamics Quantities for Selected Substances at 298.15 K (25⁰C) Substance ∆H⁰f (kJ/mol) ∆G⁰f (kJ/mol) S (J/K-mol) Carbon C2H2(g) 226.7 209.2 200.8 CO2(g) -393.5 -394.4 213.6 Hydrogen H2(g) 0 0 130.58 Oxygen O2(g) 0 0 205.0 H2O(l) -285.83 -237.13 69.91 What is the ∆S⁰ in the combustion of ethane in the presence of excess oxygen yields carbon dioxide and water: 2C2H2(g) + 7O2(g) → 4CO2(g) + 6H2O(l) in J/K?Calculate the ΔS°rxn of the following reaction at 215°C and standard pressure. C2H4 (g)+302 (g)->2CO2 (g)+2H2 ΔHºf, kJ/mol Sº, J/mol•K ΔGºf, kJ/mol C2H4(g) 52.3 219.5 68.1 O2(g) 0 205.0 0 CO2(g) -393.5 213.6 -394.4 H2O(g) -241.8 188.7 -228.6 _____ J/mol*KCOMPOUND ∆Hf (kJ/mole) S° (J/K mole) MgCO3 –1111 65.86 BaCO3 –1213 112.1 MgO –601.6 27.0 BaO –548 72.1 CO2 –393.5 213.8 Calculate ∆G° for the reaction of MCO3 ---> MO + CO2 at 25°C for the alkaline earth compounds Mg and Ba
- Thermodynamics Quantities for Selected Substances at 298.15 K (25⁰C) Substance ∆H⁰f (kJ/mol) ∆G⁰f (kJ/mol) S (J/K-mol) Hydrogen H2(g) 0 0 130.58 Oxygen O2(g) 0 0 205.0 H2O(l) -285.83 -237.13 69.91 10. What is the ∆S⁰ in the combustion of hydrogen in the presence of excess oxygen yields water: 2H2(g) + O2(g) → 2H2O(l) in J/K?Assume no acid-base catalysis occurs and the following data: Under biological conditions (310 K) 1. dGb = -20.00 kJ/mol. 2. The dG of E +S binding to ES (dGs) is -3.00 kJ/mol. 3. dGd mismatch of energy in ES = +2.00 kJ/mol. What is the catalytic power (kcat/k_uncat) at 360 K? Use 1 decimal place.Use the free energies of the formation given below to calculate the equilibrium constant (Keq) for the following reaction at 298 K. 2 AXZ3 (aq) + XZ (g) → 3 XZ2 (g) + A2Z (l) Keq = ? Using the following Data: AXZ3 (aq) XZ (g) XZ2 (g) A2Z (l) ΔG°f (kJ/mol) = -110.9 87.6 51.3 -237.1
- Consider the following ∆H°f and S° at 300K: Chemical species ∆H°f S° (in J/mol. K) C2H6(g) -83.7 229.6 H2O(g) -241.82 188.83 CO2(g) -110.53 199.42 O2(g) 0 205.138 A) Determine the ∆H°rnx of the following reaction at 300K (in kJ/mol) C2H6(g) + 7/2 O2(g) → 3 H2O (g) + 2 CO2(g) B) Determine the ∆S°rnx of the following reaction at 300K (in kJ/mol) C2H6(g) + 7/2 O2(g) → 3 H2O (g) + 2 CO2(g) C) Determine the ∆G°rnx of the following reaction at 300K (in kJ/mol) C2H6(g) + 7/2 O2(g) → 3 H2O (g) + 2 CO2(g) Show the process and calculationsDifferentiate between circumstances for the use of nRln(V2/V1) ??? Cvln(T2/T1) in calculating change in SThermodynamics Quantities for Selected Substances at 298.15 K (25⁰C) Substance ∆H⁰f (kJ/mol) ∆G⁰f (kJ/mol) S (J/K-mol) Carbon C(s, diamond) 1.88 2.84 2.43 CO(g) -110.5 -137.2 197.9 Hydrogen H2(g) 0 0 130.58 Oxygen O2(g) 0 0 205.0 H2O(l) -285.83 -237.13 69.91 What is the ∆S⁰ for the reaction: 2C(s, diamond) + O2(g) → 2CO(g) in J/K?



