Q: Choose the formation reaction that represents for liquid methanol (CH3OH): a. CH3OH(l) →...
A:
Q: The phosphodiesterase that catalyzes hydrolysis of cyclic AMP is inhibited by caffeine. What overall...
A: The phosphodiesterase that catalyzes hydrolysis of cyclic AMP is inhibited by caffeine. What overall...
Q: 3. Given the following set of fluorescence data for Fluorescein, solve for the following: A. LLOD (i...
A: To Calculate: A. LLOD B. LLOQ C. Linear range of quantification
Q: What is the mass % of ethylene glycol in a 1.89 M solution of ethylene glycol (MM = 62.07 g/mol) in ...
A:
Q: How does aspirin lower the risk of heart attack? O Aspirin stimulates the conversion of prothrombin ...
A:
Q: (4) For a rotational-vibrational spectrum of H"Br (B = 8.46 cm) taken at 520 K, determine the R-bran...
A:
Q: Benzillic Acid Lab Q3. Cold water and ethanol was used to wash/rinse the collected solid during vacu...
A: The rinse is to wash off impurities that adhere to the surface of the tetraphenylcyclopentadienone c...
Q: : Determine the pH of a .25 M CH3NH3Cl solution (Ka for CH3NH3+ = 2.3*10^-11)(Show the dissociation ...
A: To Calculate pH , we would need to draw an ICE table and calculate concentration of H3O+. From this ...
Q: A 0.651 L solution was made by mixing 186. mL of hexane, which has a density of 0.66 g/mL, and 0.44 ...
A:
Q: Calculate the molar solubility of barium fluoride in: (a.) pure water (b.)0.10 M Ba(NO3)2 (c.)0.15 ...
A: Here We have to calculate the molar Solubility of barium fluoride in different solvents given follow...
Q: 0- + CH;CH2NH2 a. Draw the structure of the tetrahedral intermediate INITIALLY-FORMED in the reactio...
A:
Q: For each of the following reactions, (i) write a complete, balanced equation that includes appropria...
A: In balanced chemical equation number of atoms of each species are equal on both reactant and product...
Q: Assume that in a certain cell, the ratio of products/reactants or Keq = 614.9 (Keq is dimensionless)...
A: Given that, an equilibrium reaction is Glucose + ATP ↔Glucose-6-P + ADP. The equilibrium constant is...
Q: The time of outflow of a gas through a small opening is 43.3 mins, while that of an equal mass of hy...
A:
Q: Assume that in a certain cell, the ratio of products/reactants or Keq = 894.2 (Keq is dimensionless)...
A: For the reaction., Glucose + ATP ----> Glucose-6-P + ADP
Q: help use significant figures please
A: Given, the balanced Chemical equation is - Ag2O(s) + 2 HNO3(l) ----> 2 AgNO3(s) + H2O(l) Temper...
Q: - You have been given an order to prepare 14 progesterone capsules for a patient in your pharmacy. T...
A: No of capsules to be made = 14 Total weight of capsules = 4.9 g
Q: will act as buffer solutions? 50 ml 0.1M H2CO3 and 100 ml 0.1M NaOH 50 ml 0.1M H2CO3 and 50 ml 0.1M...
A: Buffer solution is solution of weak acid or weak base with it's conjugate base or conjugate acid res...
Q: What is the concentration in %m/v of a 0.729 M aqueous solution of ethanol (MM = 46.07 g/mol)? %3D
A: % m/v mean grams of solute per 100 mls of solution.
Q: Fusion describes the process of * a. two small atoms joining together to produce one larger atom. ...
A: Answer Fusion is the process that, It is the reaction in which two ato...
Q: Suppose that 6.56 moles of A at 4.10 atm and 298.15 K and 5.35 moles of B at 4.82 atm and 298.15 K a...
A: Given: For gas A Moles (nA) = 6.56 mol Pressure (PA) = 4.10 atm Temperature = 298.15 K For gas B Mol...
Q: At equilibrium, the concentration of the reactants change until the concentration of the products is...
A:
Q: ROH .R Pyridine H3C
A: Here we have to write the mechanism of reaction in which ethanoyl chloride CH3COCl reacts with alcoh...
Q: (38) (a) For the Xee) atoms at 250K, considered to be an ideal gas, indicate in the distribution fun...
A:
Q: QUESTION 11 In order to convert between moles of one compound and moles of another compound, where d...
A: Question 11 The coefficients in a balanced chemical equation stands for the moles of reactants and ...
Q: Draw chiral molecules that meet the following descriptions:
A: Chiral molecule : A moleucle that contains at least one chiral carbon are called chiral molecule C...
Q: An increase of greenhouse gasses would have what effect on the planet? * a. Decrease the Temperatu...
A: Effect of greenhouse gases — Greenhouse gases plays important role in environment of planet.
Q: Practical use of chemistry is all around us. It improves our daily life in a way you may not be awar...
A:
Q: Industrially, methanol is normally produced by reacting hydrogen and carbon monoxide gasses together...
A: The ideal gas law is given by PV=nRT equation, where P, V, n are the pressure, volume, and number of...
Q: QUESTION 19 What is the exchange energy (in T e) of a d complex in the low spin state ? 17 O A. 2 O ...
A:
Q: ne neutral organic product forme
A:
Q: What is the compounded chemical symbol for nitrous oxide? * NOa aNO A. B. NaOa Na0 D.
A: Nitrous oxide is also known as laughing gas...
Q: Prelaboratory Assignment: Qualitative Analysis Group II Cations 1. Prepare a complete flow chart for...
A: Please find the below attachment.
Q: A1 liter solution is made by adding 0.8603 moles NaH2PO4 and 0.4973 moles Na2HPO4. What will be the ...
A: Given, NaH2PO4 = 0.8603 mol Na2HPO4 = 0.4973 mol [HCl] = 6.0287 M VHCl = 1.5857 mL = 0.0015857 L
Q: HBr (1 equiv) -80°C
A: Low temperature favour Kinetically control reaction that is 1,2 addition take place
Q: 5. Which solution has the lowest pH, a buffer solution made with 0.10 M acetic acid and 0.01 M sodiu...
A: We need to determine, which buffer solution has lower pH
Q: Describe the temperature-composition diagram. What did you observe with the temperature of the vapor...
A: Type of chart used to show conditions at which thermodynamically distinct phases occur and coexist a...
Q: Prelaboratory Assignment: Qualitative Analysis Group II Cations 1. Prepare a complete flow chart for...
A: The group II cations are ||Bi3+, Sn4 +, Sb3+ and Cu²+Follow the steps to determine the presence of c...
Q: MgBr |. 2.
A:
Q: There are three primary subatomic particles that comprise an atom. (i) Name the three subatomic part...
A:
Q: Please help use proper significant figure
A: Entropy of vaporization = 87.30 J/Kmole Enthalpy of vaporization = 33.30 KJ/mole
Q: hich of the properties below apply to the polymer that forms when the fo H H + H- OH ÓH hoose all th...
A:
Q: 2. The bromination of 2-butene is an exergonic reaction. Br H3C. Br2 CH3 CH3 Br a. Draw the mechanis...
A: a ) The given reaction is an example of addition of Br to an alkene. It is a two step reaction . b ...
Q: . Which is the stronger acid: a) NH4* or H2O b) BrCH2- О or FCH2—ОН c) H2SO4 or H2SO3
A:
Q: A gas originally occupying 10.1L at 0.925 ATM and 25 degrees is changed to 13.3 L at 625 torr. What ...
A:
Q: propane (C3H8) undergoes complete combustion in oxygen, it produces carbon dioxide and water, as sho...
A:
Q: What is the pH of a 0.215 M solution of HIO (hipoiodous acid)? (Use your workbook to find Ka) Upload...
A: [H+] = (Ka × C )1/2
Q: A. The hydrogen ion concentration of a solution is 6.40x10-12 M. What is the pOH of the solution? Ex...
A: For part A, given the concentration of the hydrogen ion = 6.40x10-12 M. We have to calculate the pOH...
Q: Calculate the initial concentration of Fe ion in the solution below. Fe3+ (aq) + SCN– (aq) ⇌ ...
A:
Q: Indicate for each species below whether it is a strong acid, a strong nucleophile, both, or neither:...
A: Strong acid means ,the species which can donate H+ very easily or indicate the availability of H+. S...
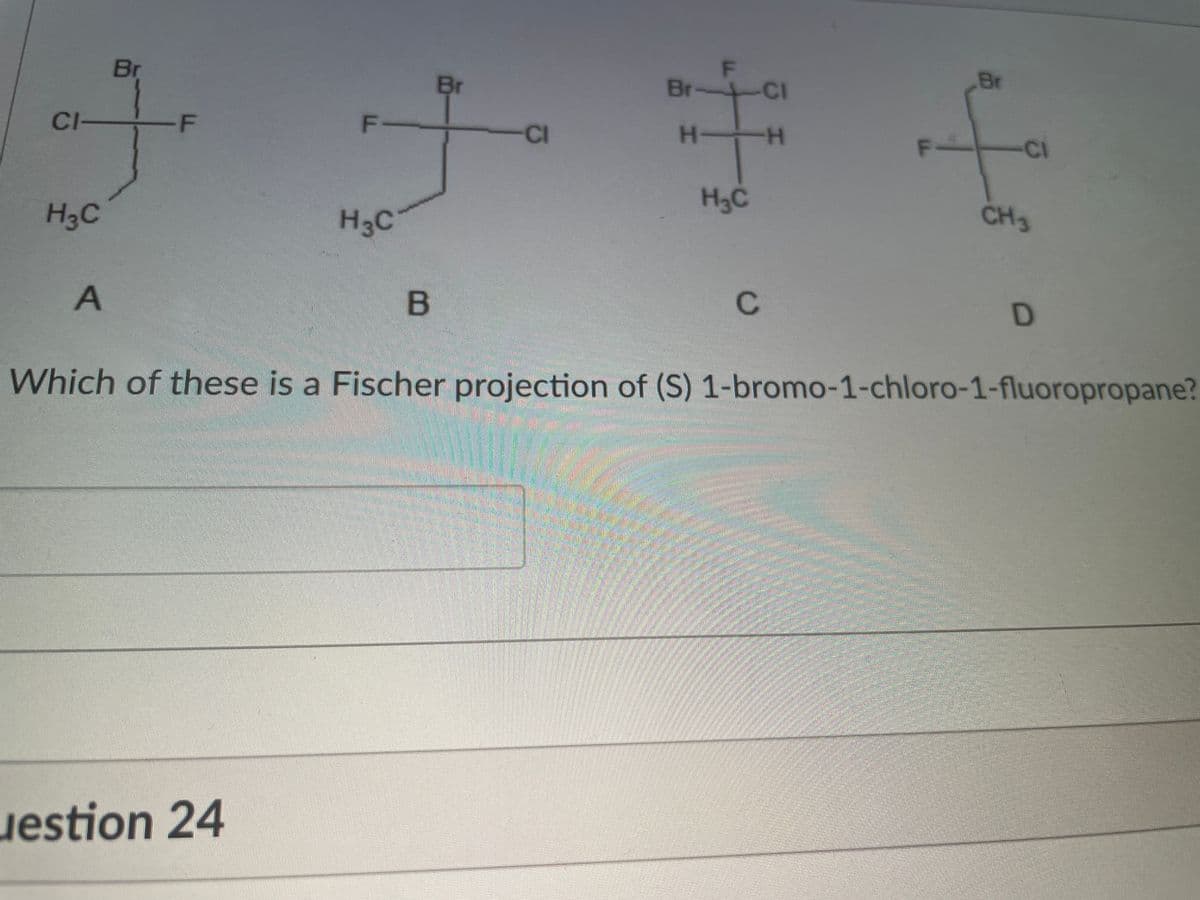
Step by step
Solved in 2 steps with 1 images

- Draw the structure of Ethyl 2-deoxoy-2-fluoro-a-D-galactopyranoside in: i) cyclized Fischer projection ii) in its most stable chair configuration iii) in its least stable chair confirmationConvert the following Fischer projections into tetrahedral representations, and assign R or S stereochemistry to the molecules on part a and b in the pictureAllenes are compounds with adjacent carbon-carbon double bonds. Many allenes are chiral, even though they don’t contain chirality centers. Mycomycin, for example, a naturally occurring antibiotic isolated from the bacterium Nocardia acidophilus, is chiral and has [α]D = -130. Explain why mycomycin is chiral.
- Org. Chem. 1. EA26. Can you please draw out all four , and explain what a stereoisomer is in relation ? ThanksRank the following groups in order of decreasing priority. −C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.
- Draw the Fischer projection for the final product when trans-2-butene reacts with mCPBA/H2O.a. Draw a three-dimensional structure for the following steroid. b. What is the structure of the single stereoisomer formed by reduction of this ketone wrth H2, Pd-C? Explain why only one stereoisomer is formed.(a) (S)-2-chlorobutane, draw any diastereomers.

