Q: Label the following pairs of compounds as: a. identical b. constitutional isomers c. neither
A: If the compounds have the same molecular formula but have a different bonding arrangement then they…
Q: Consider the following molecule `H. a) Draw a different staggered conformation of the same molecule…
A: There are various methods of representation of a molecule. It can be: Newman projection Saw-horse…
Q: A chiral alkyne A with molecular formula C3H10 is reduced with H2 and Lindlar catalyst to B having…
A: Lindlar Catalyst is the catalyst (H2 / Pd / CaCO3) which is use to reduce the triple bonds between…
Q: Draw the two chair conformations of menthol, and identify the more stable conformation. Explain…
A: Menthol is an example of substituted cyclohexene. In the chair conformation of cyclohexane, 6 of the…
Q: Q2: Draw all possible stereoisomers of 1,2-cyclobutanedicarboxylic acid, and indicate the…
A:
Q: Draw the eight constitutional isomers having molecular formula C3H;,CI. Label any stereogenic…
A:
Q: give the most stable chair conformation given bleow structures
A: The structure of the cyclohexane molecule can be expressed in two conformations: chair and boat. The…
Q: a) How many degrees of unsaturation (double bond equivalents) are represented by the molecular…
A:
Q: How are the compounds in each pair related? Are they identical molecules or enantiomers?
A: Enantiomer: Two molecules having non-superimposable mirror image are called as enantiomer.…
Q: Drawn are four isomeric dimethylcyclopropanes. a.How are the compounds in each pair related…
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: Rank the alkenes shown in the ball-and-stick models (A–C) in order of increasing stability.
A: Stabolity is decided by two factors a. at the double bond it should be more alkylated then it will…
Q: For the molecule shown below, draw both chair conformations and circle which is the lower energy…
A: The given molecule is (1S,2S,4S)-1,2,4-trifluorocyclohexane. It contains three fluorine group…
Q: Rank the following compounds in order from smallest heat of combustion to largest heat of…
A: Heat of combustion, ∆Hc is the energy released (exothermic) when 1 mole of substance is burned to…
Q: Draw the topological structure of the following molecule, clearly illustrating the stereochemistry.…
A:
Q: Considering rotation around the bond highlighted in red in each compound, draw Newman projections…
A:
Q: to a b. Identify each of the above structures as either achiral, chiral, or a meso compound
A: Chiral molecules Molecules from whom the plane of symmetry does not pass are called chiral…
Q: Explain the stereochemistry between Molecule A and B
A: The stereoisomers that have a mirror-image relationship with each other is termed as the…
Q: Select all the structural (constitutional) isomers for Y from the set of compounds A-E. (Note that…
A: Structural isomer are the compound having same chemical formula but structure are different.
Q: Label the following pairs of compounds as: a. identical b. constitutional isomers c. neither Place…
A:
Q: A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having…
A: The molecular formula of the compound is C6H10. The compound contains 6 carbon atoms and 10 hydrogen…
Q: (a) Are compounds B–D identical to or an isomer of A? (b) Give the IUPAC name for A.
A: The structure of chemical compound can be represented as structural formula. A structural formula…
Q: (A) Which is the anti-conformer of hexane in Newman projection? (B) Which is the equivalent…
A: Anti conformation: The most stable conformation in which the alkyl groups are at a dihedral angle of…
Q: ) BH,- THF B (i) HO, NAOH A
A: The Newmann Projection of the product can be drawn as follows:
Q: Labeling Stereogenic Centers with R or S ? Define ?
A: To explain the labeling R and S on the stereogenic Centers.
Q: 3.1 Draw both chair conformations of compound M on the templates (X and Y) provided. Indicate which…
A: The solution is as follows:
Q: Complete the following for the compound shown below. a) Draw the line structure using the wedge/dash…
A: We only answer up to 3 sub-parts. Please resubmit the question with other sub-parts (up to 3) to be…
Q: (a) Are compounds B–D identical to or an isomer of A? (b) Give theIUPAC name for A.
A: IUPAC nomenclature is the systematic method of naming organic compounds following simple rules. The…
Q: Bongkrekic acid is a toxic compound producted by Pseudomonas cocovenenans, and isolated from a mold…
A: If the higher priority group at one end of the double bond and the higher priority group at the…
Q: Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A
A: Stereoisomers–these isomers have same molecular formula and same sequence of bonds but differ in…
Q: Drawn are four isomeric dimethylcyclopropanes. A B C a. How are the compounds in each pair related…
A: We are given four isomeric structure of 1,2-dimethylcyclopropane. We have to find the relation…
Q: What structure has the lowest energy?
A: For trans-1,3-substituted cyclohexane, the substituents should be in (a,e) or (e,a) positions to…
Q: Label compounds B–D as stereoisomers, conformations, or constitutional isomers of A.
A: Stereoisomers–these isomers have same molecular formula and same sequence of bonds but differ in…
Q: A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having…
A: The molecular formula of the compound is C6H10. The compound contains 6 carbon atoms and 10 hydrogen…
Q: (e)
A: Complete solution is given below in next step with these conformation.
Q: 2. Draw the flipped chair conformation of a. Which of the two conformations is more stable? Justify…
A:
Q: a) State two (2) types of stereoisomer. b) Explain briefly, which compound hexane or…
A: a. Stereoisomerism is the arrangement of atoms in molecules whose connectivity remains the same but…
Q: Consider the following compound represented in Newman projection: a) Draw this compound as shown in…
A: We can assign the solid and dashed line to the different substituents by observing the overall…
Q: Draw in a plane of symmetry for each molecule. а. b. с. d. CI CI
A:
Q: Take a look at the butane conformers below. Identify: (a)Which is an anti conformation in Newman?…
A: In anti conformation the two methyl groups lie as far apart from each other as possible with a…
Q: Identify A,B,C, and D
A: An aldose chain's C1–C2 bond is broken by the Wohl degradation, which also shortens the chain by one…
Q: B) Write a structural formula that corresponds to the molecular formula CH,O and is a. acyclic b.…
A: In organic chemistry, one type of Molecular formula used to draw more than one structural formulas ,…
Q: Drawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related…
A: A pair of compound is said to be constitutional isomers if it has the same molecular formula with…
Q: Add wedges and dashed wedges for all stereogenic centers with the following information: the…
A: The skeletal structure of the anabolic steroid 4-androstene-3,17-dione can be drawn by adding…
Q: Label each attached compound as cis or trans. Then draw the second chair conformation.
A: The axial and equatorial positions in chair conformation of cyclohexane are as, In case of cis…
Q: Which of compounds A-C would you expect to have the greatest heat of combustion? The smallest?…
A: Heat of combustion of the substance can be defined as the energy value or calorific value is the…
Q: Answer the following question about compounds A–D (See in attachment) Label each compound as a cis…
A: Cis isomers are those where the substituents are on the same side and Trans isomers are those where…
Q: Which isomer, A or B, would you expect to have the greater heat of combustion? Explain. A B
A:
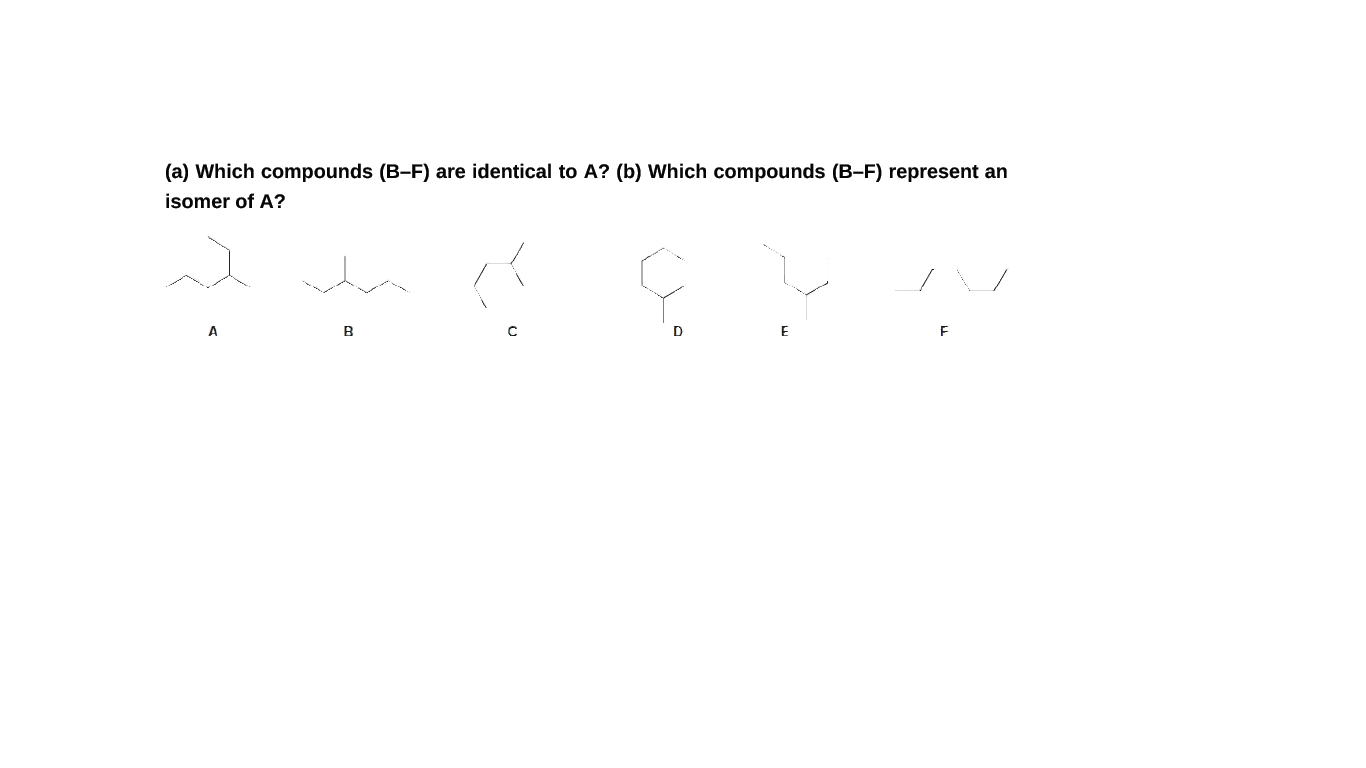
Which compounds (B–F) are identical to A? (b) Which compounds (B–F) represent an isomer of A?
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Answer the following question about compounds A–D (See in attachment) How are the compounds in each pair related? Choose fromconstitutional isomers, stereoisomers, or identical molecules: A and B; A and C; B and D.Classify attached compound as identical to A or its enantiomer.Drawn are four isomeric dimethylcyclopropane. a. How are the compounds in each pair related (enantiomers, diastereomers,constitutional isomers): A and B; A and C; B and C; C and D?b. Label each compound as chiral or achiral.c. Which compounds, alone, would be optically active?d. Which compounds have a plane of symmetry?e. Which of the compounds are meso compounds?f. Would an equal mixture of compounds C and D be optically active? Whatabout an equal mixture of B and C?g. How many stereogenic centers are there for each compound?
- Explain why compound A has two stereoisomers but compounds B and C exist as single compounds.Having difficulty knowing difference between enantiomer and identical isomerHow are the compounds in attached pair related to each other? Are they identical, enantiomers, diastereomers, constitutional isomers, or not isomers of each other??
- Classify each compound as identical to A or its enantiomer.Part D. Do the two structures A and B of each pair drawn below represent the same molecule, constitutional isomers, or stereoisomers? If A and B are stereoisomers, further classify them as enantiomers or diastereomers.How is each compound (B–D) related to A? Choose from enantiomers, diastereomers, identical molecules, constitutional isomers, or not isomers of each other.
- Locate the stereogenic centers in attached compound. A molecule may have one or more stereogenic centers. Gabapentin enacarbil [part (d)] is used to treat seizures and certain types of chronic pain.Consider the following six compounds (A–F). How are the two compounds in each pair related? Choose from enantiomers, epimers, diastereomers but not epimers, constitutional isomers, and identical compounds.a. A and Bb. A and Cc. B and Cd. A and De. E and FDraw both enantiomers for the attached biologically active compound.

