4.) Consider the reaction below. Reaction: AH (kJ/mole) 2 ICI (g) 17.78 1 (g) Ca (g) 247.551 116.135 223.066 Umole K) Which is the equilibrium concentration of ICI when 0.45 mol l2, 0.45 mol C2, and 2.42 mol ICl are added to a 2.0L flask at 25°C? A. 0.076 M B. 1.36 M C. 1.14 M D. 0.151 M Keywords: reaction quotient, equilibrium constant, initial concentration(s), final concentration(s), RICE table, equilibrium expression, x-variable atration Concepts: Determination of equilibrium conce
4.) Consider the reaction below. Reaction: AH (kJ/mole) 2 ICI (g) 17.78 1 (g) Ca (g) 247.551 116.135 223.066 Umole K) Which is the equilibrium concentration of ICI when 0.45 mol l2, 0.45 mol C2, and 2.42 mol ICl are added to a 2.0L flask at 25°C? A. 0.076 M B. 1.36 M C. 1.14 M D. 0.151 M Keywords: reaction quotient, equilibrium constant, initial concentration(s), final concentration(s), RICE table, equilibrium expression, x-variable atration Concepts: Determination of equilibrium conce
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter15: Principles Of Chemical Reactivity: Equilibria
Section15.3: Determining An Equilibrium Constant
Problem 15.3CYU: A solution is prepared by dissolving 0.050 mol of diiodocyclohexane, C5H10I2, in the solvent...
Related questions
Question
100%
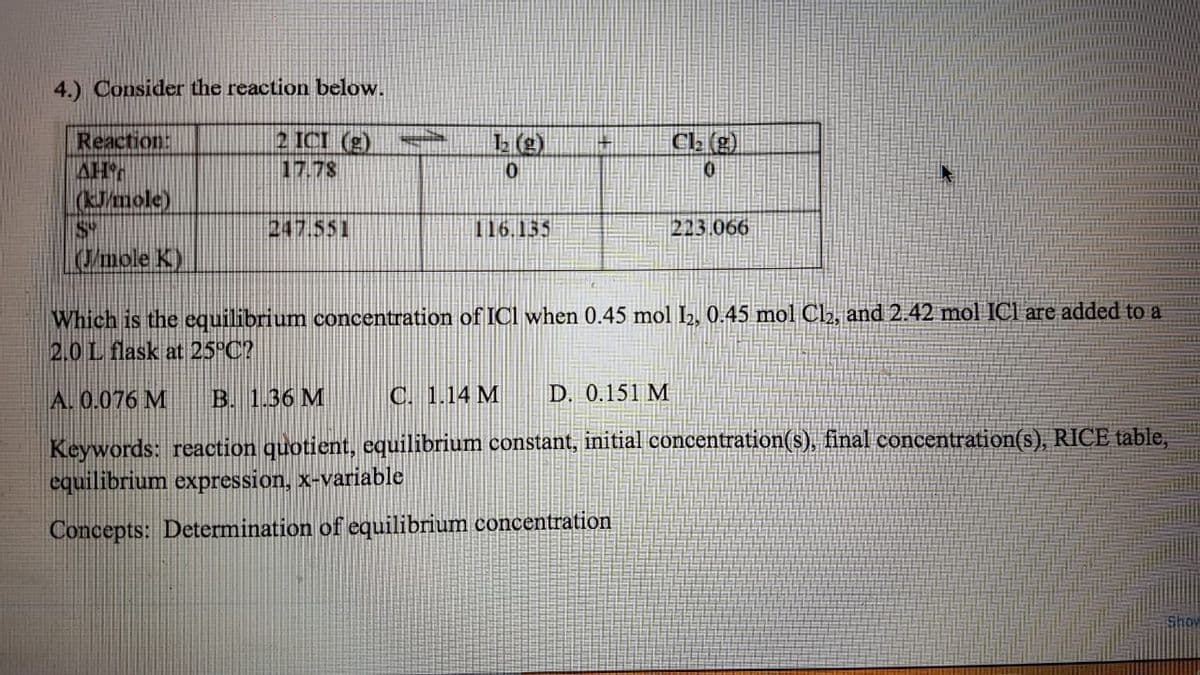
Transcribed Image Text:4.) Consider the reaction below.
2 ICI (g)
17.78
Reaction:
AH
(Jmole)
S
/mole K)
CL (g)
247.551
116.155
213.066
Which is the equilibrium concentration of ICI when 0.45 mol 2, 0.45 mol Cl2, and 2.42 mol ICI are added to a
2.0L flask at 25°C?
A. 0.076 M
B. 1.36 M
C. 1.14 M
D. 0.151 M
Keywords: reaction quotient, cquilibrium constant, initial concentration(s), final concentration(s), RICE table,
equilibrium expression, x-variable
Concepts: Determination of equilibrium concentration
Show
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning