The statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG° stands for the standare Gibbs free energy of reaction and K stands for the equilibrium constant. In each table, there is one statement that is false because it contradicts the other three statements. When you find a false statement, check the box next to it. statement false? statement false? AG°<0 AG°>0 K<1 K>1 AH°>TAS° In K<0 In K>0 AH°
The statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG° stands for the standare Gibbs free energy of reaction and K stands for the equilibrium constant. In each table, there is one statement that is false because it contradicts the other three statements. When you find a false statement, check the box next to it. statement false? statement false? AG°<0 AG°>0 K<1 K>1 AH°>TAS° In K<0 In K>0 AH°
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter16: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 5ALQ
Related questions
Question
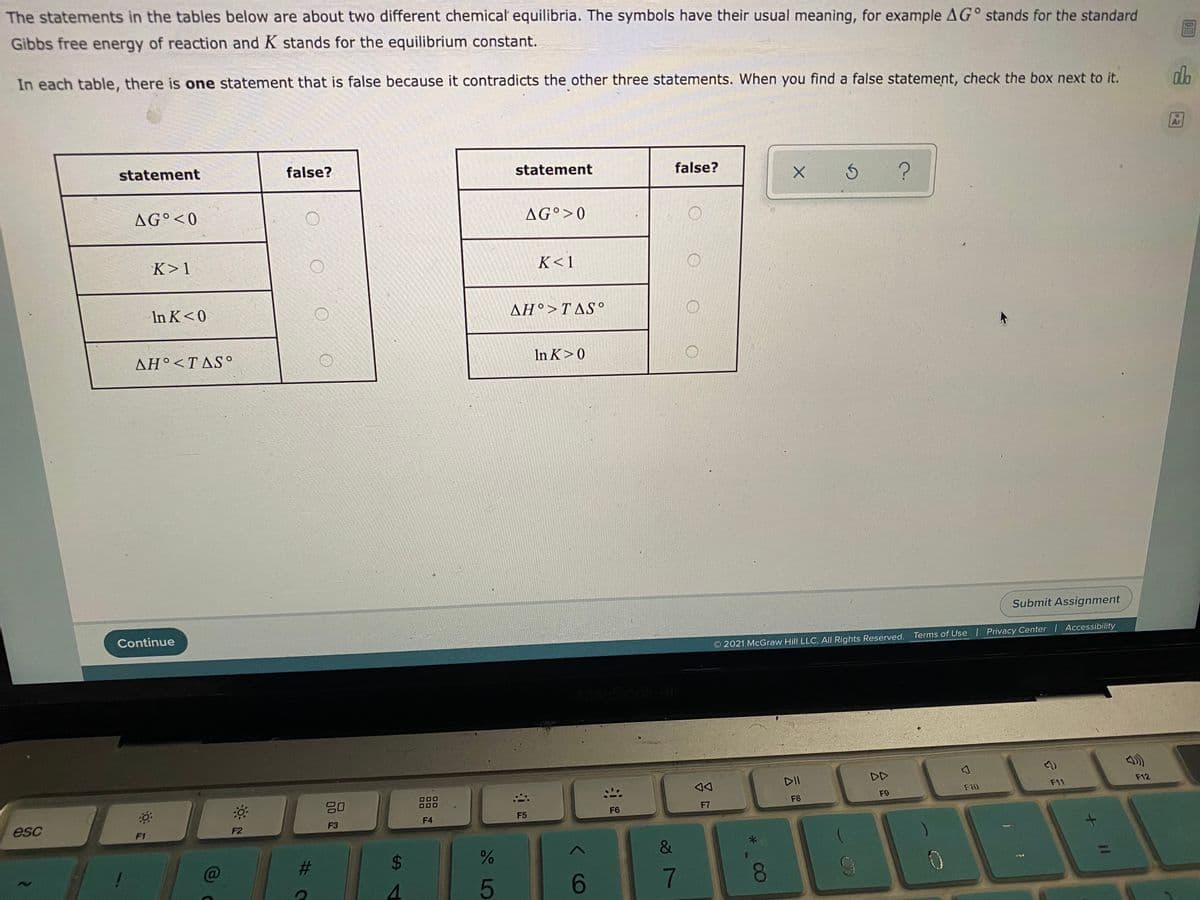
Transcribed Image Text:The statements in the tables below are about two different chemical equilibria. The symbols have their usual meaning, for example AG° stands for the standard
Gibbs free energy of reaction and K stands for the equilibrium constant.
In each table, there is one statement that is false because it contradicts the other three statements. When you find a false statement, check the box next to it.
do
Ar
statement
false?
statement
false?
AG°<0
AG°>0
K>1
K< 1
In K<0
ΔΗ> ΤAS.
AH°<TAS°
In K>0
Submit Assignment
Continue
Accessibility
O 2021 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center
MacBooki
DII
DD
F11
F12
O00
80
F9
F10
F8
F6
F7
esc
F3
F4
F5
F1
F2
&
C@
#3
5
6
7.
1O国
CO
%24
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning