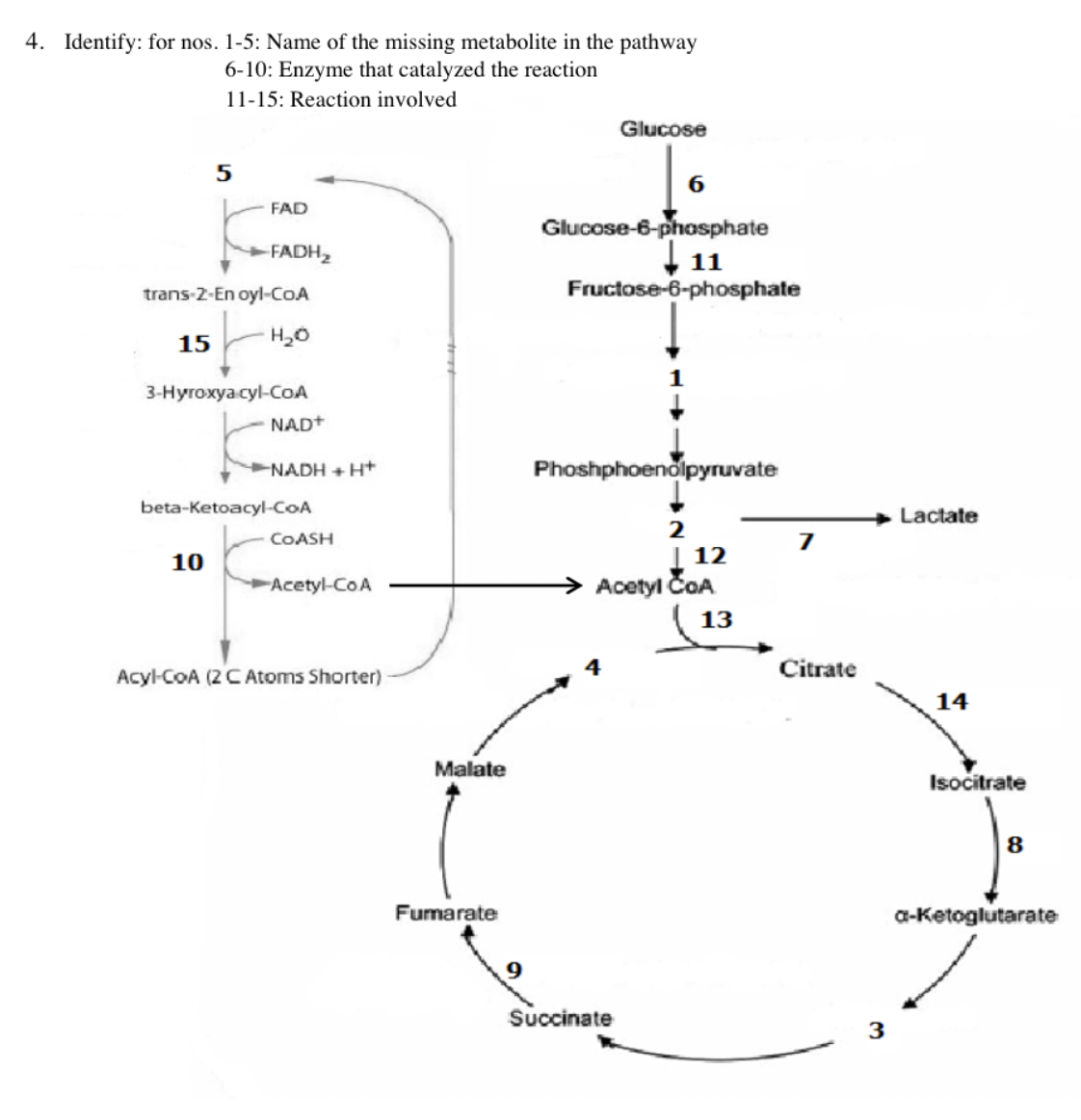
4. Identify: for nos. 1-5: Name of the missing metabolite in the pathway 6-10: Enzyme that catalyzed the reaction 11-15: Reaction involved 5 FAD -FADH₂ trans-2-En oyl-CoA 15 H₂O 10 3-Hyroxya.cyl-CoA NAD+ NADH + H+ beta-Ketoacyl-CoA COASH ►Acetyl-CoA Acyl-CoA (2 C Atoms Shorter) Malate Fumarate 9 Glucose Glucose-6-phosphate 11 Fructose-6-phosphate Phoshphoenolpyruvate Succinate 2 | 12 Acetyl ČOA 13 Citrate Lactate 14 Isocitrate 8 a-ketoglutarate
Q: a) What would be the effect on glycogen degradation if a mutation prevents the subunits of protein…
A: After a meal, there is a surplus supply of glucose in the blood. this causes the pancreas to secrete…
Q: Triosephosphate isomerase catalyzes the conversion of glyceraldehyde 3-phosphate to dihydroxyacetone…
A: In a general reaction such as: aA + bB⇌ cC + dD At equilibrium, the concentration of reactants and…
Q: How many hydrogen bonds exist between this DNA strand and its complementary strand? 5'-GCATAAT-3'
A: Nucleic acids are biomolecules that are essential for all life forms. They are polymers of…
Q: 1. Carbohydrate digestion: gastrointestinal enzymes and the optimal pH environment for their…
A: Carbohydrates are key energy source in our body and basic subunit of carbohydrate is…
Q: Proteins destined for the peroxisomes can be translocated in their functional shape. True False
A: Protein targeting is a biological process by which the cells localize the proteins in different…
Q: Indicate whether each of the following descriptions concerning the molecule sphingosine is true or…
A: Phospholipids are compound lipids composed of fatty acids, alcohol, phosphate group, and nitrogenous…
Q: Question: 1. Give the Haworth and chair forms for the a and ß anomers of: D-galactose b. D-mannose…
A: Haworth projection is a cyclic representation of monosacharide sugars in the form of pyranoses and…
Q: Which of the following statements is/are FALSE about Bradford assay? 1. Upon addition of the…
A: Bradford Assay is an accurate spectroscopic analytical method to measure the concentration of total…
Q: Describe various detection methods used in High-Performance Liquid Chromatography (HPLC) and types…
A: High-performance liquid chromatography or high-pressure liquid chromatography, is an analytical…
Q: When k2 >> k-1, KM approximates the affinity of the enzyme•substrate complex. The (circle one)…
A: Km is also known as Michaelis constant. It is named after German biochemist Leonor Michaelis. It is…
Q: Briefly explain chymotrypsin with reference to the following: a) Catalytic triad (not the mechanism…
A: DISCLAIMER FOR MULTIPART Since you have posted a question with multiple sub-parts, we will solve…
Q: If your body sense that is very low on the substrates necessary to produce ATP homeostatic control…
A: Deamination of amino acids - it is the process of breakdown of amino acids which occurs in excess…
Q: A certain amount of white solid was dissolved in enough water and gives a colorless solution. A…
A: Carbohydrate: Also known as saccharides are often composed of monosaccharides, which are simple…
Q: A PCR reaction was performed to amplify the CHE2 gene. The PCR primers used were designed to amplify…
A: Polymerase chain reaction is a molecular biology technique that is sued to synthesize multiple…
Q: Provide the correct three-letter abbreviation for the following amino acid: H3N-CH-C- I CH3
A: The proteins are constituted of twenty naturally occurring amino acid that are connected by peptide…
Q: multimeric protein contains 20 or greater different amino acid residues At least 2 or more…
A: Introduction Proteins are the most abundant macromolecules in our body. Proteins are made up of…
Q: 1 2 3 4 5 6 N Sample Lysate Name volume 8 Tilapia Cod Sole Perch Tilapia Cod Sole 2 μl Perch 10 με 2…
A: Protein sample: A non-branching sequence of amino acids joined together by a single peptide bond…
Q: 1. Consider the reaction: succinyl-CoA succinate H3C H3C C H₂ a. What kind of reaction is being…
A: Acetyl CoA produced from fatty acid oxidation in the hepatic cells has two possible fates: enter…
Q: For class I MHC, which of the following statements are correct? P. They are expressed on all…
A: MHC are proteins encoded by a huge complex of genes called the major histocompatibility complex…
Q: 1. What is the possible identity of the amino acid? [ Select] V 2. What is the isoelectric point of…
A: Amino acids are biomolecules that have an amino group, a carboxyl group and a side group that is…
Q: 1. Propose a mechanism by which HIV protease hydrolytically cleaves the peptide bond of the…
A: Proteins can be degraded by breaking the peptide bonds that hold amino acid residues together.…
Q: The structure shown below iS: H HO CH₂OH -0 H OH H -H OH HOH₂C H 1 0 A) Sucrose 2ÍCH HỌ OB)…
A: The four types of biological macromolecules are nucleic acid, proteins, lipids and carbohydrates.…
Q: Isozymes: d) One way of regulating metabolic pathways is through isozymes. What is an isozyme? e)…
A: Introduction: Every living system requires various forms of specific enzymes in order to increase…
Q: Increased ROS levels are a hallmark for Alzheimer's Disease. This is accurately described by all but…
A: Within a cell, reactive oxygen species are produced as a byproduct of oxygen metabolism and the cell…
Q: Which of the following types of bonds are not involved in maintaining the tertiary structure of…
A:
Q: Draw the catalytic triad of a serine protease at the first tetrahedryl intermediate stage. Your…
A: Serine proteases cleaves peptide bonds of protein substrates. They are called serine proteases…
Q: Sketch 1/v v.s. 1/[s] for enzyme 1 and enzyme 2 where enzyme 1 has same Vmax as enzyme 2 but has…
A: Enzymes are bio-catalyst that participate in biochemical process and they are highly specific in…
Q: Riboflavin is an important nutrient required for a number of biochemical processes that produce…
A: Riboflavin is commonly known as vitamin B2. Riboflavin is a type of Vitamin B. It is soluble in…
Q: Think of an additional application of the Disk diffusion method based on your experience and…
A: Disk diffusion method is a culture based microbiological assay technique in which bacteria is grown…
Q: The naturally occurring opioid met-enkephalin is a peptide with the structure Tyr- Gly-Gly-Phe-Met.…
A: Amino acids are zwitterions having both positive and negative charges at neutral pH. The net charge…
Q: From a kinetics experiment, Kcat was determined to be 55sec^-1. For the kinetic assay, 0.05mL of a…
A: The concentration of enzyme stock solution was 0.05mg/ml. i.e. 1ml contain 0.05mg of enzyme. We took…
Q: In the final step of the citric acid cycle, malate is oxidized to regenerate the oxaloacetate…
A: In a general reaction such as: aA + bB ⇌ cC + dD At equilibrium (steady state), the concentration of…
Q: What is the biochemical cause and effect of anemia in sickle cell disease?
A: Sickle cell anemia is a genetic disease (it is an autosomal recessive disorder). This affects the…
Q: 8. Features of anaerobic oxidation of glucose in erythrocytes. Pyruvate kinase, its biological…
A: Anaerobic oxidation (or Glycolysis): Anaerobic glycolysis is the conversion of glucose to lactate…
Q: Structural and functional features of the lysosomes include I. internal pH is higher than the…
A: Lysosomes are cell organelles that help in cellular digestion of foreign particles, food particles…
Q: in triacylglycerol mobilization, triacylglycerol molecules is activated by: phosphorylation…
A: Triacylglycerol are esters of fatty acid and glycerol. Triacylglycerol, as the name indicates,…
Q: The highest level of structural complexity completely retained by the molten globule state of a…
A: The term "molten globule state of a protein "refers to the diverse types of partially-folded protein…
Q: The dissociation of lactic acid to lactate is shown in the reaction. Lactic acid has a pKa of 3.86.…
A: Dissociation of a weak acid is mathematically described by the Henderson-Hasselbalch equation: pH =…
Q: The beta-pleated sheets are stabilized by hydrogen bonds among adjacent regions of the peptide…
A: Introduction Protein is the most abundant macromolecule in our body. proteins are made up of carbon,…
Q: The structure shown is an example of a type of macromolecule (carbohydrate, lipid, protein, or…
A: The biological macromolecules that constitute a cell are proteins, nucleic acids, lipids and…
Q: Amino acid analysis of a HEPTAPEPTIDE reveals the following information below: (NOTE: when the…
A: The proteins are composed of sequence of amino acids connected via peptide bonds. The sequence of…
Q: Which type(s) of chromosomal aberrations is/are likely to cause semisterility? Select all correct…
A: Introduction Any change in chromosome number and chromosome structure can cause various genetic…
Q: Identify the biochemical role that each plays within a biochemical transformation:…
A: Coenzymes and cofactors are non protein components that are essential for a protein function. Most…
Q: G Protein-Coupled Receptor (GPCR) Signaling Q.7.3: Glucagon and epinephrine both signal stress,…
A: Glucagon is a peptide hormone secreted from the alpha cells of the pancreatic islets of Langerhans.…
Q: Define what a substrate cycle is and how it might be beneficial to an organism.
A: Introduction All living organisms need energy for their survival. We take food to get energy. The…
Q: At a substrate concentration that is much greater than the KM for the reaction, which statement is…
A: The velocity of an enzyme catalyzed reaction is given by: V=Vmax×SKM+SWhere,V= velocity of the…
Q: Amino Acid Derivatives Q7.3: What explains the observation that individuals with phenylketonuria…
A: Phenylketonuria is an inborn error of metabolism. This disease is caused by mutations in the gene…
Q: Match the following experimental methods with their specialty procedures and objectives.…
A: There are experimental methods that are used in biological processes. Autoradiography, the use of…
Q: Does the presence of a competitive inhibitor increase / decrease the apparent affinity of the…
A: There are three types of enzyme inhibitors. They are competitive, non-competitive and mixed…
Q: Draw the following Lipids in: A) Condensed (detailed) Structure, B) its Block Diagram. C)…
A: Lipids are a chemically diverse group of biomolecules that have two things in common: low…
Step by step
Solved in 5 steps

- Extending the Mechanism of Methylmalonyl-CoA Mutase to Similar Reactions Based on the mechanism for the methylmalonyl-CoA mutase (see problem 14), write reasonable mechanisms for the following reactions shown.CHOOSE THE CORRECT LETTER 1.What are the B-oxidation products of lauric acid (C12H2402), a major component of coconut oil?A.6 acetyl-CoA, 6 NADH, 6 FADH2B. 6 acetyl-CoA, 5 NADH, 5 FADH2C. 5 acetyl-CoA, 6 NADH, 6 FADH2D. 5 acetyl-CoA, 5 NADH, 5 FADH2 2.What is the mechanism of ATP synthesis in glycolysis?A. substrate level phosphorylationB. reductionC. oxidationD. oxidative phosphorylation 3.Which of the following enzymes need ATP as a substrate in the reaction?A.phosphoglycerate kinaseB. phosphofructokinaseC. pyruvate kinaseD.glyceraldehyde-3-phosphate dehydrogenaseComplete the table below. Consider docosanoic acid (C21H43CO2H) Questions Solutions Answers a. Label the alpha and beta carbons. b. Draw the acetyl CoA derived from this fatty acid. c. How many acetyl CoA molecules are formed by complete B-oxidation? d. How many cycles of B-oxidation are needed for complete oxidation? e. How many molecules of ATP are formed from the complete catabolism of this fatty acid?
- Complete the following paragraph describing the fate of the second 14CO-acetyl-CoA molecule incorporated into the glyoxylate cycle. The glyoxylate produced in the isocitrate reaction combines with a (A.) 2nd molecule of oxoaloacetate, B.) 2nd molecule of succinate, C.)2nd molecule of acetyl coa) in the malate synthase reaction. Malate is (A.Chiral, B.diasteromeoic C.)Achiral) ; therefore, the oxaloacetate produced from glyoxylate will be labeled at (A.Either of carboxylate carbon B. specifically at methylene carbon C. Specifically at carbonyl carbon D. Specifically at carboxylate carbon bonded to methylene).succinyl-COA synthetase will do which of the following: Select one: a. Fumarate is combined with water to become Malate b. alpha-ketoglutarate is oxidized and decarboxylated to produce Succinyl-CoA, Carbon dioxide and NADH c. Succinate is oxidized to become fumarate forming FADH2 d. Citrate is rearranged to become Isocitrate e. Malate is oxidized to become oxaloacetate forming NADH f. Pyruvate is decarboxylated to become acetyl-CoA producing NADH and Carbon dioxide g. Succinyl-CoA becomes Succinate and forms one ATP molecule and Coenzyme A-SH h. Oxaloacetate combines with the acetyl from acetyl-CoA to produce Citric acid(citrate) i. Isocitrate and then decarboxylated and oxidized to produce alpha-ketoglutarate, Carbon dioxide and NADHCHOOSE THE CORRECT LETTER 1.Which of the following substrates is used in the first oxidation step of the Kreb's cycle to produce NAH and CO2?A. isocitrateB.a-ketoglutarateC. citrateD. succinyl-CoA 2.What is the reason for isomerization of citrate to isocitrate?A.The reaction converts a tertiary alcohol, which cannot easily be oxidized, to a secondary alcohol that can be oxidizedB. Isocitrate protects cells from the toxic effects of arsenite ion.C. The reaction is one major regulatory step for the citric acid cycle because it functions as a rate limiting step.D. Isocitrate is a substrate for a reaction that occurs spontaneously without enzymatic catalysis
- Which of the following biochemical conversions can be carried out by the least number of proteins? Assume proper metabolic conditions, abundance of cofactors, necessary accessory molecules, etc. 1 propionyl-CoA → 1 succinyl-CoA 1 palmitate (16:0) → 8 acetyl-CoA 1 acetyl-CoA + 7 malonyl-CoA → 1 palmitate (16:0) 1 palmitate (16:0) → 1 acetyl-CoA + 1 myristic acid (14:0) 1 acetyl-CoA → 1 succinate Please answer very soon will give rating surelyMany components of the glycolytic pathway and the citricacid cycle are direct exit or entry points to metabolic pathways ofother substances. Indicate another pathway available to the following compounds:(a) Fructose-6-phosphate(b) Oxaloacetate(c) Glucose-6-phosphate(d) Acetyl-CoA(e) Glyceraldehyde-3-phosphate(f) -Ketoglutarate(g) Dihydroxyacetone phosphate(h) Succinyl-CoA(i) 3-Phosphoglycerate(j) Fumarate(k) Phosphoenolpyruvate(l) Citrate(m) PyruvateFor myristic acid, C 13H 27CO 2H: (a) How many molecules of acetyl CoA are formed from complete β-oxidation? (b) How many cycles of β-oxidation are needed for complete oxidation?
- Which of the following is most helpful in ensuring that wasteful (unproductive) hydrolysis of acetyl CoA does not occur in the reaction catalyzed by citrate synthase? Group of answer choices Citrate synthase is a tetramer Induced fit ensures oxaloacetate binds before acetyl CoA 3 molecules of oxaloacetate bind for every 1 molecule of acetyl CoA Citrate inhibits the reaction Which of the following catalyzes a step that does NOT produce CO2? Group of answer choices alpha-ketoglutarate dehydrogenase complex malate dehydrogenase pyruvate dehydrogenase complex isocitrate dehydrogenase Citrate synthase is regulated by... Group of answer choices Concentrations of substrate and products Fructose-2,6-Bisphosphate Covalent modification Allosteric regulation by the levels of NADDHComplete the table below. Consider docosanoic acid (C21H43CO2H) Questions Show complete solutions Answers a. How many acetyl CoA molecules are formed by complete B-oxidation? b. How many cycles of B-oxidation are needed for complete oxidation? c. How many molecules of ATP are formed from the complete catabolism of this fatty acid?Consider oleic acid (18:1D9): How many rounds of beta oxidation will omit the enzyme acyl-CoA DH?

