4. In EACH PAIR of drug forms below: (a) Circle which form is the most soluble in water and... (b) Explain your choice using the strongest attractive force between the drug form(s) and water. NH H S CH3 -N- CH3 HO. Na* Penicillin Salt form of penicillin CH, | CH, Methamphetamine (Free Base) Crystal Methamphetamine (salt) H. CH3 CH, Cr O N 0-CH, C-0-CH, Salt Form of Cocaine Free Base Form of Cocaine ("Crack") 22
4. In EACH PAIR of drug forms below: (a) Circle which form is the most soluble in water and... (b) Explain your choice using the strongest attractive force between the drug form(s) and water. NH H S CH3 -N- CH3 HO. Na* Penicillin Salt form of penicillin CH, | CH, Methamphetamine (Free Base) Crystal Methamphetamine (salt) H. CH3 CH, Cr O N 0-CH, C-0-CH, Salt Form of Cocaine Free Base Form of Cocaine ("Crack") 22
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 24QAP
Related questions
Question
100%
In EACH PAIR of drug forms below:
(a) Circle which form is the most soluble in water and . . .
(b) Explain your choice using the strongest attractive force between the drug form(s) and water.
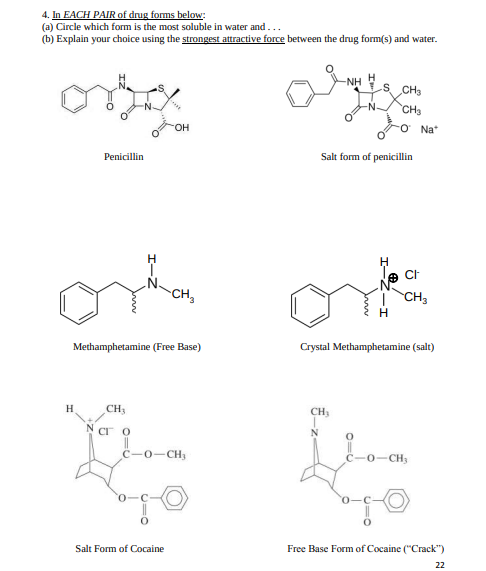
Transcribed Image Text:4. In EACH PAIR of drug forms below:
(a) Circle which form is the most soluble in water and...
(b) Explain your choice using the strongest attractive force between the drug form(s) and water.
H
s CH3
-NH
CH3
он
Na*
Penicillin
Salt form of penicillin
CH,
| CH,
Methamphetamine (Free Base)
Crystal Methamphetamine (salt)
H.
CH
CH
CI O
N
C-0-CH,
0-CH
Salt Form of Cocaine
Free Base Form of Cocaine ("Crack")
22
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning


World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:
9781305577190
Author:
Kenneth L. Williamson, Katherine M. Masters
Publisher:
Brooks Cole