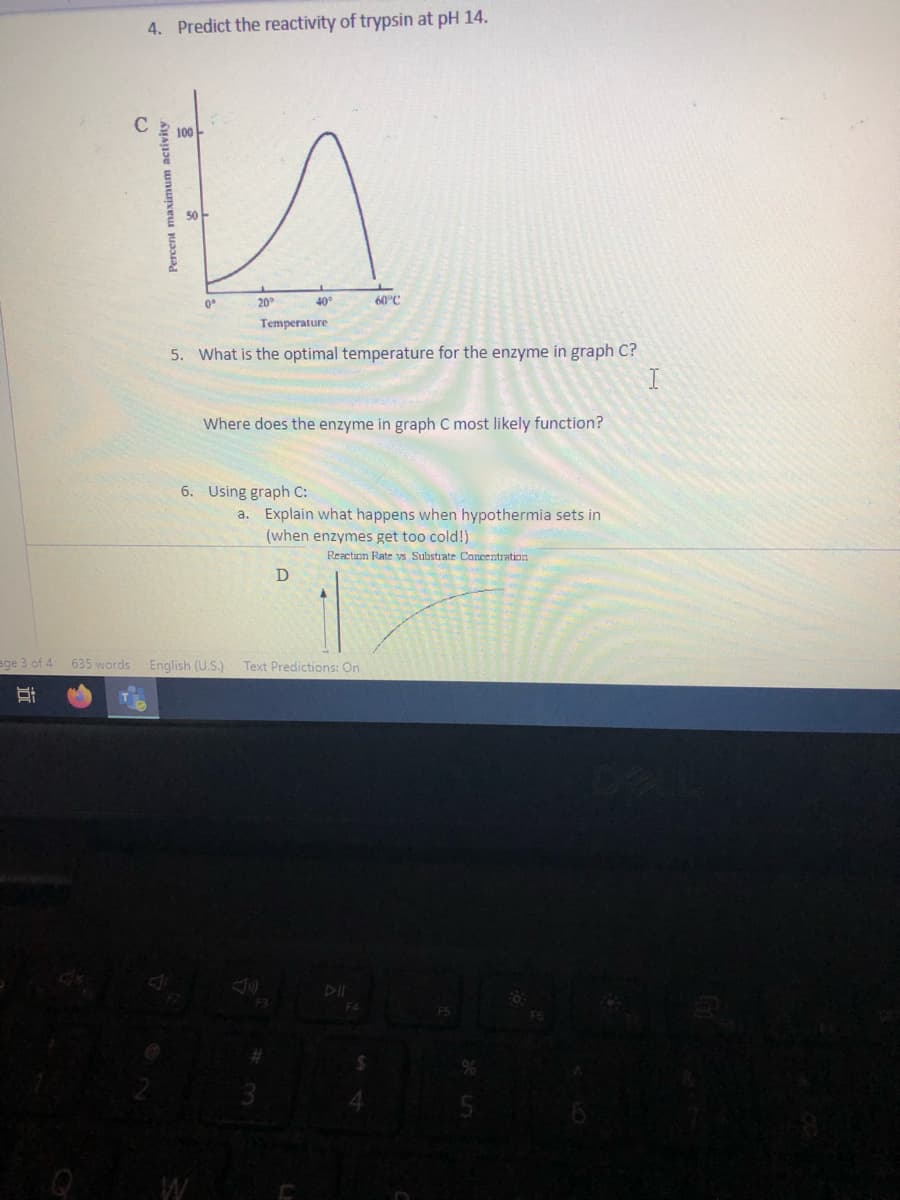
4. Predict the reactivity of trypsin at pH 14.
Q: 6) What is the cognate a-keto acid of alanine? You may have to look at alanine.
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: 4. Draw the formula of Ala-Cys at pH = pl a) b) pH > pl c) pH < pl (Ala: pK.(COOH)= 2,34, pK.(NH3")…
A: Ala-Cys is a dipeptide. Alaninne has R= CH3 but cysteine has R= CH2-SH.
Q: 8) Define the following terms: a. Hydrophobic b. Hydrophillic
A: Introduction The cell membrane is a thin semi-permeable membrane that surrounds the cytoplasm of a…
Q: 1.Discuss the Biochemical Tests for Lipids.
A: Lipids are biomolecules composed of fatty acids that are esterified to an alcohol. The biochemical…
Q: 3. Describe the function of each of the following molecules/cofactors:
A: a. ATP is made up of adenosine and three phosphate. b. NAD+ is nicotinamide adenine dinucleotide. It…
Q: 11. Choose the polyunsaturated triglyceride from the compounds below. A) HO, B) H,C-0-C E) HĊ-0-C C)…
A: Triglycerides are glycerol molecules esterified with three molecules of fatty acids. It is a kind of…
Q: 6) The order is for amoxicillin 500 mg PO. You have on hand a 150-mL bottle of dry powder. The…
A: In this question we will ist calculate the concentration of Amoxicillin in 1 ml of the solution and…
Q: 5. At pH 7, the net charge of tyrosine is? А. -2 В. -1 С. 0 D. +1 E. +2
A: PH is defined as the negative logarithm of H+ ion concentration. The amount of H+ ions in a solution…
Q: 3. Based on the name of the following hypothetical drug salts, which of the following statements is…
A: The given options of hypothetical drug can be described as below in terms of acid and base:…
Q: 4. Match the terms on the left with those on the right: AUG dihydrouridine Esite
A: Above given in the match the following are the terms involved in the translation process studied in…
Q: 3. Identify the amino acids contained in each of the following tripeptides. он он он || |…
A: Amino acids are the monomers of polypeptides that are linked by peptide bonds. A peptide bond is…
Q: 4. Which of the following biopolymers is correctly paired with the bond that forms between the…
A: The body exhibits different biological molecules, which are formed from the monomers.
Q: 2. How many calories we need for excretion toxic nitrogenous waste due to 200g Protein?
A: Urea is primarily produced by mammals, including humans. They are known as ureotelic animals because…
Q: 4. Which of the following is an example for group specificity? a) Trypsin hydrolyzing peptide…
A: Group specificity - It implies that the enzyme will catalyze a particular reaction on the function…
Q: 3. Why do some people overdose with folic acid?
A: Folic acid : Synthetic form of folate.
Q: 2. Classify the following alkaloid according to ring system, draw the structure, then give the…
A:
Q: 6.) Predict the product of the following chemical reactions and state where the product (waste)…
A: Introduction (d) 3‐pentanol→PCC 3‐pentanone Ignitability characteristic: hazardous waste number…
Q: 5. What is the reaction of phenolphthalein and methyl orange in acid-base solution? Acid Base…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: (9 Why does DNA contain thymine instead of uracil? Justify.
A: Given: DNA contain Thymine instead of Uracil. DNA is a long polymer of polynucleotide chain. It…
Q: 4. Lipids can also be detected by a preliminary translucent spot test, also called the paper bag…
A: A translucent spot test is a qualitative assay for the determination of fats and oils. When the…
Q: 1. The use of natural proteins as drugs requires compliance with certain storage and use conditions,…
A: Protein: Polymer of amino acid joined together through peptide bond removing water in a process…
Q: 2. Which of the following forms of lysine would you expect to predominate at low pH, neutral pH, and…
A: Lysine is a positively charged polar amino acid with ε-amino group as the functional group. Amino…
Q: 3. What are the functions of lysine in human health?
A:
Q: 1. Discuss the difference between an ⍺-D-glucose and a β-D-glucose. How can the structure of a…
A: Monosaccharides having 5 or 6 carbons in the chain gives cyclic structure in aqueous solution via…
Q: 6. Which of the following acts as lipid-soluble radical trapping antioxidants? A. Ascorbate B. Uric…
A: There are two types of radical trapping antioxidants:- Water soluble lipid soluble
Q: 3. What would be the optimum pH for pepsin, an enzyme that breaks down protein in your stomach? Why?
A: ANSWER) The optimum pH for enzyme Pepsin is 2. At 2 pH Pepsin exhibits maximal activity.
Q: 12. An increase in the concentration of a substance can increase a reaction rate because it effects
A: Factors that affect the reaction rate are - 1. Concentration of reactant 2.Physical state of…
Q: 4. What is the total charge of lysine in the strong acidic medium? A) +2 B) -2 C) +1 D) 0 E) -1
A: Proteins are known as the muscle-building substances of the body. They are very complex…
Q: 3) The following is the amino acid sequence of bradykinin, a peptide released by certain organisms…
A: P
Q: your hospital pharmacy makes a fortified tobramycin eyedrops that is 13.6% in a 10 ml bottle. How…
A: The ratio of mass of solute present in a solution relative to the volume of the solution represents…
Q: 2. How do oils and fats differ? Explain for 6 sentences
A: Organic chemicals are important in the diet and valuable in a variety of industries. Oils and fats…
Q: 7. For those drugs that need to be applied with dissolution sustained system, what are their…
A: Note: Only the 8th and 10th questions can be answered under nursing. Angles at which needles are…
Q: 2. Draw the structure of pentapeptide Trp-Asp-Cys-Lys-Gln that would predominate in aqueous solution…
A: .
Q: 1. Write the reversible reactions with its corresponding enzymes of Dehydration via Hydrolysis…
A: Biomolecules are the biological molecules that are present inside the living organisms. These…
Q: 4 Identify the following glycerophospholipid, which helps con- duct nerve impulses in the body, as a…
A: Lipids are a chemically diverse group of organic compounds which are insoluble or only poorly…
Q: 4. Predict the products (draw their structures) of the fatty acid with the reagents below. (a) H* +…
A: The reaction that is depicted in a, is using H+ as catalyst. So it is acid catalysis reaction. Acid…
Q: 1. Draw the phospholipid formed from palmitate, oleate, glycerol, phosphate and serine.
A: Phospholipids are types of lipids that contain two hydrophobic tails made up of fatty acids and one…
Q: 1. What are the effects CCl 4 solvents when mixed with lipids? 2. Explain enzyme hydrolysis.
A: Carbon tetrachloride (CCl4) binds to triacylglycerols and phospholipids throughout subcellular…
Q: 1. Write the reversible reactions with its corresponding enzymes of Dehydration via Hydrolysis…
A: Biomolecules are the biological molecules that are present inside the living organisms. These…
Q: 3. Fill in the missing molecules or required cofactors. PLP b) @NH₂ OH
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have…
Q: 1. Which of the following is a weak base? A. CH,COOH B. CH;CH;NHCH, C. HOCN D. NAOH E. None of…
A: Base and acid both interact with each other and form salt and water. They both are used in everyday…
Q: Explain how water activity and pH can affect the susceptibility of proteins to denaturation by heat.
A: Proteins are the ultimate products of the genes. Peptide bond is bond which is formed between two…
Q: 9. Which of the following changes would not affect a peptide GENSHIN? A. Heat B. Inorganic Salts C.…
A: Proteins are unbranched polymers constructed from 20 standard α-amino acids. They have four levels…
Q: 1. What is the advantage of using an iodophor (betadine) rather than iodine or an iodine tincture?
A: As per the guidelines, first is answered. Disinfectants are chemical agents that are built on inert…
Q: 3. Which of the following does not apply to steroids? A. Some are hormones B. Composed of fatty…
A: Introduction A steroid is an organic molecule having four rings organised in a certain molecular…
Q: What feature of the structure of verbascose makes it indigestible
A: Verbascose is a pentasaccharide composed of one galactopyranose, two glactose monomers, one glucose,…
Q: Why would you not expect sucrose (disaccharide) to test positive in Benedict’s test?
A: 4. Carbohydrates are the energy providing macromolecules which comprises of sugars and starch, whole…
Q: draw an isomer of the triglyceride in sunflower seed oil. Show the reaction and write out the…
A: Hydrogenation is the process of chemical modification that is performed by addition of hydrogen to…
Q: 10. Why would it be inaccurate to say that solution that maintains a constant pH? KU a buffer is a
A: The pH of blood is approximately 7.4, which is somewhat basic. This pH fluctuates by less than a…
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

- In an enzymatic reaction: a. the enzyme leaves the reaction chemically unchanged. b. if the enzyme molecules approach maximal rate, and the substrate is continually increased, the rate of the reaction does not reach saturation. c. in the stomach, enzymes would have an optimal activity at a neutral pH. d. increasing temperature above the optimal value slows the reaction rate. e. the least important level of organization for an enzyme is its tertiary structure.Which of the following statements about the allosteric site is true? a. The allosteric site is a second active site on a substrate in a metabolic pathway. b. The allosteric site on an enzyme can allow the product of a metabolic pathway to inhibit that enzyme and stop the pathway. c. When the allosteric site of an enzyme is occupied, the reaction is irreversible and the enzyme cannot react again. d. An allosteric activator prevents binding at the active site. e. An enzyme that possesses allosteric sites does not possess an active site.Which of the following methods is not used by enzymes to increase the rate of reactions? a. covalent bonding with the substrate at their active site b. bringing reacting molecules into close prosimity c. orienting reactants into positions to favor transition states d. changing charges on reactants to hasten their reactivity e. increasing fit of enzyme and substrate that reduces the energy of activation
- In the following graph: A represents the product. B represents the energy of activation when enzymes are present. C is the free energy difference between A and D. C is the energy of activation without enzymes. E is the difference in free energy between the reactant and the products.When two glucose molecules react to form maltose: a. the reaction represents a negative G. b. free energy had to be available to allow the reaction to proceed. c. the reaction is exothermic. d. it supports the second law of thermodynamics, which states there is tendency of the universe toward disorder. e. the resulting product has less potential energy than the reactants.1. Using the following data, graph percent enzyme activity on the y-axis and pH on the x-axis 2.Do these enzymes have different optimum pHs? Explain why or why not. 3. Research what specific human enzymes would best match the data.
- 1. Make a Lineweaver-Burk plot and use the plot to complete the information in the table and the following questions. a. Is it possible for the enzyme to overcome the effect of the inhibitor in question from the chart. Explain. b. What prevents this enzyme from being an even more catalytically efficient enzyme? c. What do single molecule data indicate about the validity of ensemble data?d. What is the reason that humans are insensitive to sulfa drugs?1) what is the Vmax of the enzyme WITHOUT inhibitor 2) What is the Km of the enzyme WITHOUT the inhibitor 3) The specificity constant for enzyme X is (8*10^7) /(M*seconds); what is the kcat of the enzyme WITHOUT the inhibitor? 4) what was the concentration of the enzyme used for measuring the kinetics of enzyme X without inhibitor? 5) the dashed line represents enzyme with inhibitor. The concentration of the inhibitor is 5 micromolar. Calculate the equilibrium constant for the inhibitor Please show work and units1. Consider the following parameters related to an enzyme that follows Michaelis-Menten kinetics for the reaction: k(1) k(2) S ----> ES ----> P k(-1)
- An enzyme is found that catalyzes the reaction X ⇌ Y. Researchers find that the Km for the substrate X is 4 μM, and the kcat is 20 min−1.(a) In an experiment, [X] = 6 mM, and V0 = 480 nM min−1. What was the [Et] used in the experiment?(b) In another experiment, [Et] = 0.5 μM, and the measured V0 = 5 μM min−1. What was the [X] used in the experiment?(c) The compound Z is found to be a very strong competitive inhibitor of the enzyme, with an α of 10. In an experiment with the same [Et] as in (a), but a different [X], an amount of Z is added that reduces V0 to 240 nM min−1. What is the [X] in this experiment?(d) Based on the kinetic parameters given above, has this enzyme evolved to achieve catalytic perfection? Explain your answer briefly, using the kinetic parameter(s) that define catalytic perfection.1 the concentration of the enzyme competition inhibitor was 1×10-3M. If 1 µmol of the inhibitor is present inthe 1 mL reaction mixture, Indicate how much the initial degree of hydrolysis decreases as a proportion(in thepresence of inhibitors/in the absence of inhibitors) with respect to the absence of inhibitors.1 what is does the induced -fit model account for? 2 why are most enzyme inactive at higher temperature ( greater than 37degree Celsius) 3 Does this happen to all enzymes in all species? 4 What are the formulas for the reduced form of the coenzymes?

