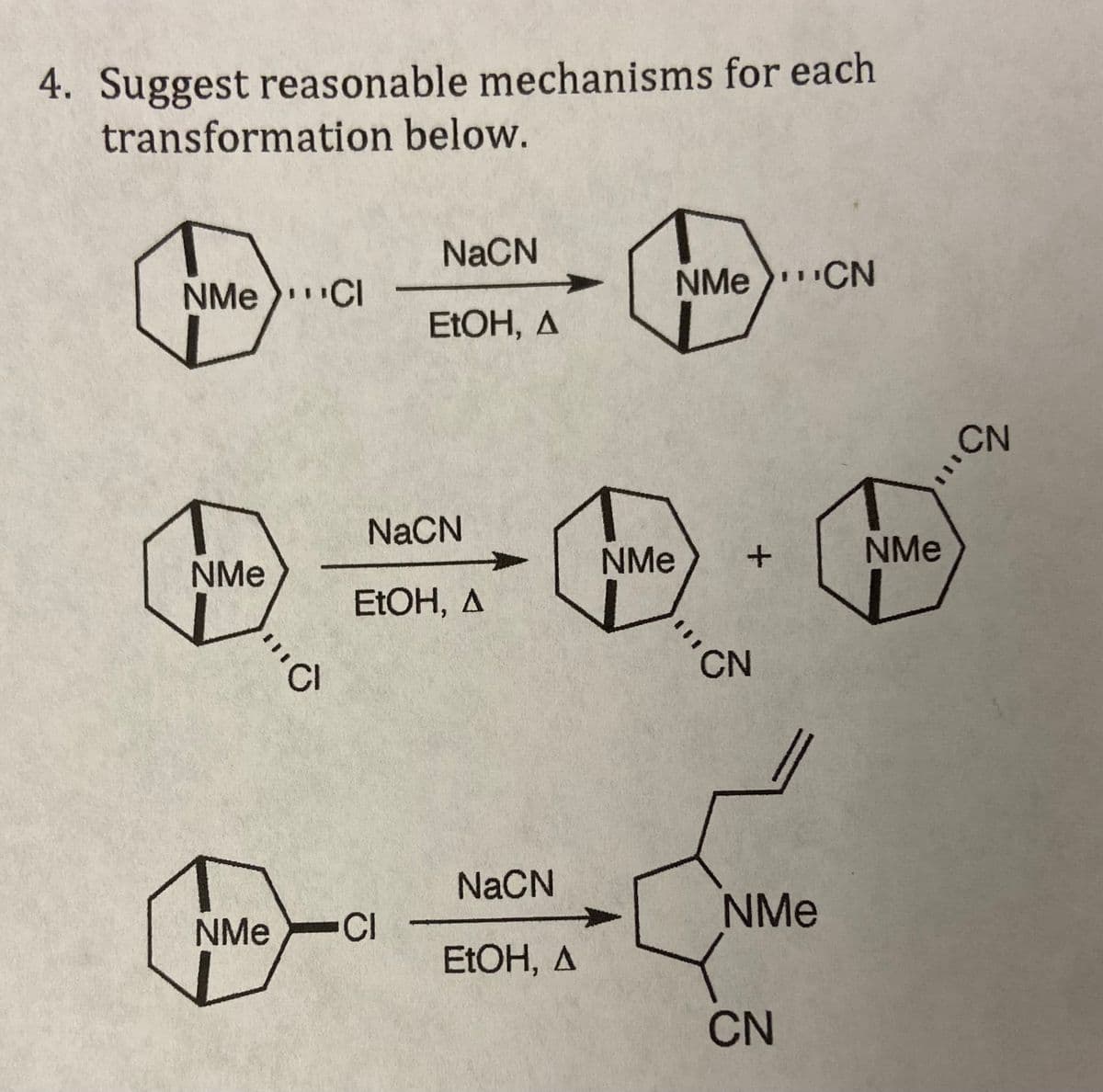
4. Suggest reasonable mechanisms for each transformation below. NME CI NMe "C 'Cl NaCN EtOH, A NaCN EtOH, A NMe CI NaCN EtOH, A NMe NMe CN + CN NMe CN NMe CN
Q: The following transformation can be accomplished by using the provided below (table) complete the…
A:
Q: What is the correct IUPAC name for the following: Br 5 3 a. (S)-3-bromohexane b. (R)-3-Bromohexane…
A:
Q: Using the VSEPR theory, predict that the shape of ammonia, NH3. bent linear tetrahedral triangular…
A: We know for geometry we calculate both lone pair and bond pair where as for shape we consider only…
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A:
Q: 58 AM Wed Jul 5 < Question 14 of 15 96% 1 The pH of a solution of HBr is 2.88 at 25 °C. What is the…
A:
Q: What part does adsorption play in froth floatation for the concentration of sulphide ores and…
A: In Froth Flotation: Froth flotation is a widely used method for concentrating sulphide ores, such as…
Q: Which of the following liquids do you expect to have a visible meniscus in a glass buret? Select all…
A:
Q: what is the formal charge of each atom in the structure, and choose the best structure. mal charge…
A:
Q: Name the following molecules, including all relevant stereochemical descriptors. 3 1 2 a "OH
A: A question based on nomenclature of organic compounds. The organic compounds given are to be named…
Q: Draw each step of the mechanism separately. Use proper electron pushing to show where the electrons…
A: The given reaction involves a mechanism of 3 steps:Step 1: Protonation of oxygen of epoxide takes…
Q: Please explain the meanufacturing of Ethanol. Full process diagram with material balance as well as…
A:
Q: The major product(s) of this reaction would be (R) only יון H Br CH₂OH ?
A:
Q: A relativistic electron has a de Broglie wavelength of 1.25 pm1.25 pm (1 pm=10−12 m).(1 pm=10−12 m).…
A: To determine the velocity of a relativistic electron, we can use the de Broglie wavelength and the…
Q: 14. Which one of the following compounds is consistent with the following IR spectrum? NH₂ I CH3…
A: Find the correct structure based on IR Spectrum
Q: What is equilibrium constant K for the following reaction at 25°C with a AG* of -16.4 kJ/mol? 3H₂(g)…
A: ΔG = -2.303 RT logKWhere, R = 8.314 T is in Kelvin
Q: What is the percent-by-mass concentration of NaCl in a solution that is prepared by adding 13.0 g of…
A: The percent by mass concentration is defined as the mass of solute dissolved in total mass of…
Q: A sample of oxygen gas collected at a pressure of 0.591 atm and a temperature of 278 K is found to…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: What is the pOH of a 0.123 M solution of Na₂CO3 and 0.0243 M solution of NaHCO3 if the pK, is 3.68?…
A: pH of the solution is to be calculated.Concentration of Na2CO3 = 0.123 MConcentration of NaHCO3 =…
Q: What is the major organic product of the following reaction? 1 Equiv. HBr (ignore stereochemistry)
A:
Q: What is the boiling point of a 1.2 m Ca(NO3)2 solution?
A: Given,molality of Ca(NO3)2 = 1.2 m
Q: Draw the major product of this elimination. Consider regiochemistry and stereochemistry. Ignore…
A: Strong base favors E2 reactionWeak base favors E1 reactionDuring elimination greater substituted…
Q: Your turn: Calculate pH, pOH, [H3O+], [OH-] for HNO3 solution, 1.14 mol/L ● A base solution,…
A: pH = -log[H3O+] pH + pOH = 14 [H3O+][OH-] = 1 x 10-14
Q: Complete the table below for calculating the formula weight of the ionic compound chromium(II)…
A: Formula weight is the sum of the atomic weights of all atoms appearing in a given chemical…
Q: d) Fill in the missing compounds in the reactions below. Be sure to show stereochemistry whe c) b)…
A: Draw the missing parts in the given questions.
Q: Provide a synthesis for each of the following reactions using reagents containing no more than 4…
A: The given question on synthesis can be approached with the following important points:Starting…
Q: Consider: 2KCIO3 (s) = 2KCI (s) + 30₂ (g) a) What is the effect of decreasing the volume? b) What is…
A: we have to determine the change in equilibrium on decreasing volumeadding an inert gasfor the given…
Q: 650.0−g sample of river water taken near an industrial plant contains 27 mg of chromium. What is the…
A: Weight of sample or solution = 650.0 gm Weight of chromium = 27 mg = 0.027 gm (1 gm = 1000 mg)
Q: Aqueous hydrochloric acid (HC1) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A: We have to determine the mass of HCl left in the reaction
Q: he reaction between zinc and chromium III ions has a E° cell of 0.356 V. But when test out the…
A: The reaction between Zn and Cr ions is,Reduction happens at the cathode and oxidation happens at the…
Q: 1. Hg(OAd)z, но но 2. Na BH
A: In this reaction, an alkene is treated with mercury(II) acetate, Hg(OAc)2, and the product formed is…
Q: 13. Which of the following Lewis structures are valid? A) i only .. BE i. Í :Z: B) ii only : **…
A: Lewis structures are indeed commonly used for predicting the number and types of bonds formed by…
Q: Determine whether each of the IR spectra given is consistent with the structure of an 00 2 A.Alcohol…
A: Infrared (IR) spectroscopy- IR spectroscopic analysis helps to determine the chemical functional…
Q: What does "shape-selective catalysis" mean to you? Zeolites make effective shape-selective…
A: The word shape selective catalysis implies a special type of catalysis depending on shape of…
Q: The vapor pressure of ethanol, CH3CH2OHCH3CH2OH, at 30.0 °C30.0 °C is 10.46 kPa10.46 kPa. If 2.21…
A: Data
Q: 7 Draw the major product for the following Diels-Alder reaction. Clearly show any stereochemistry…
A: The given reaction is Diels elder and it is also called as a 4+2 cycloaddition reaction.In this…
Q: 0. Which solution would you choose to suppress the dissolution of CaCO3? a. 0.200 M MaCl₂ b. 0.200…
A: Which solution would you choose to suppress the dissolution of CaCO3?option :a. 0.200 M MaCl2 b.…
Q: A non-reactive substrate analogue has been found to inhibit a target enzyme. Modifying the inhibitor…
A: An enzyme inhibitor is a molecule that binds to an enzyme and blocks it's activity.Here in question…
Q: Given the specific heat capacity of Cu is 0.385 j/g Celsius, what is the final temperature of a 10.0…
A:
Q: Question 36 Complete the statement below by predicting the major product. Use the pool of possible…
A: Given reaction :
Q: What is the Eocell (in V) for a voltaic cell using the Pb2+/Pb and Mg2+/Mg half-reactions?
A: In voltaic cell, a spontaneous reaction occur.Greater the value of standard reduction potential of a…
Q: 323. Which curve shows the solubility of amino acids in water? a. b. C. セルト [AA] [AA] [AA] 17 14 PH…
A: The question is based on the concept of solubility. solubility follows a basic principle "like…
Q: For the gas-phase equilibrium A(g) + 2 B(g) = C(g) the initial partial pressures of A, B, and C are…
A: Equilibrium reaction:Initial pressures of A, B and C are 0.300 atmThe equilibrium pressure of C =…
Q: b. Propose a product and draw a full arrow-pushing mechanism for the following epoxide opening. 8 1)…
A: In this question, we will draw the structure of the final organic product and we will draw a full…
Q: 9. Consider the following grayscale electrostatic potential map. The regions are labeled as to…
A: We have to find the atom which is most electron poor.
Q: Choose the best reagents to complete the reaction shown below. A Br2 FeBr3 SOCI2 pyridine CI
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: what is the electronegativity difference of HCN?
A: We have to find the Electronegativity difference of HCN .
Q: Write the balanced NET ionic equation for the reaction when AgC₂H3O2 and Ni(CIO4)2 are mixed in…
A: When siver acetate , AgC2H3O2 and nickel perchlorate , Ni(ClO4)2 are mixed in aqueous…
Q: At 80.2°C and 781 torr, calculate the number of moles and mass of 1.31 L of CH4 gas
A: To calculate the number of moles and mass of CH4 gas at a given temperature and pressure, we…
Q: Based on periodic trends and the data in the table, which of the following are the most probable…
A: We have to determine the atomic radius and first ionisation energy of potassium .
Q: Question I. Consider the reaction of 2-bromo-2-methylpropane with water, shown below, to answer the…
A: Energy diagram of the reaction is to be drawn.
Step by step
Solved in 3 steps with 1 images

- identify and interpret the peaks and their characteristics (C=O, 4H, doublet) product name: dulcin (4-ethoxyphenylurea) (C9H12N2O2) images include CNMR, HNMR, and IR spectroscopyA stock solution of alcohol dehydrogenase (ADH) has been diluted 1 in 10 and the absorbance reading at 280 nm of this diluted solution is 0.04. Given that the absorption coefficient for ADH at 280 nm is e mg/ml = 1.6 and that you require to do 30 assays in triplicate, each assay using 2 ug of enzyme, how many ul of the stock ADH do you need to do the experiment?creat a separation flowchart of Al3+, Sb3+ and Zn2+ without using sulfate or thioacetamine
- Identify the Ergosterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.Which of the following structures has the highest resolution? a. An X-ray structure with 3.0 Ǻ resolution b. An X-ray structure with 4.0 Ǻ resolution c. An X-ray structure with 1.5 Ǻ resolution d. An X-ray structure with 1.0 Ǻ resolution e. An NMR structure with 10 NOE’sThe following data was collected for this experiment: A sample of 0.8281 g of phenylsuccinic acid was dissolved in 10 mL of acetone. This sample gave a reading, aobs, of +10.278 deg on the polarimeter. A tube measuring 1 dm was used for the sample. What is the major enantiomer present in this sample? Calculate the concentration of the sample used in g/mL. Calculate the observed specific rotation [a]obs of the sample. Give your answer to 4 significant figures.
- Provide a multistep synthesis.Interpret peaks on this c13 NMR of Ethanoic Acid with cooresponding structureIdentify the 24-Methylenecholesterol peak of the GC-MS of Burn Morel mushrooms (Tomentosa) and the M (+/-) peaks that are present. Include the mass and signature intensities of the peaks main product.
- Why is it better to acquire the 1H-NMR spectrum in DMSO-d6 and not in CDCl3?1. The conversion of the concentrations into the required expression is as follows: Concentration given Expression required 0.5% NaCl molarity 1 mM of KBr mg/ml 5 mg/100 ml paracetamol Molarity 0.15 N H2SO4 g/litre 0.5% ethanol/water (w/v) ppm 2. 0.641 g of a semi-synthetic alkaloid was dissolved in 25 ml of 1% w/v acetic acid and was analysed directly by HPLC. The solution was found to contain 1.42 mg/100 ml of an impurity. What is the level of impurity in % w/w and ppm? 3. Calculate the pH of a buffer system made by dissolving 1.2 g of acetic acid and 0.82 g of sodium acetate in 500 ml of distilled water (pKa of acetic acid = 4.7) 0.641 g of a semi-synthetic alkaloid was dissolved in 25 ml of 1% w/v acetic acid and was analysed directly by HPLC. The solution was found to contain 1.42 mg/100 ml of an impurity. What is the level of impurity in % w/w and ppm?post lab 4 Why does the p-bromoacetanilide migrate more than the p-bromoaniline on the TLC plate?

