44,000 - 56,000 56,000 -68,000 68,000 -80,000 0.28 0.18 Clear musheiss 0.10 0.29 0.03 0.23 80,000-92,000 If the weight average DP for this sample is found to be DP=536, which of the following common polymers is this unknown sample? ** 0.16 0.05 O a. Polycarbonate with a monomer molecular weight of 61.0168 g/mol b. Polystyrene with a monomer molecular weight of 104.15 g/mol Oc. Polypropylene with a monomer molecular weight of 42.08 g/mol O d. Polyvinyl chloride with a monomer molecular weight of 62.498 g/mol Oe. Polymethylmethacrylate with a monomer molecular weight of 100.121 g/mol
44,000 - 56,000 56,000 -68,000 68,000 -80,000 0.28 0.18 Clear musheiss 0.10 0.29 0.03 0.23 80,000-92,000 If the weight average DP for this sample is found to be DP=536, which of the following common polymers is this unknown sample? ** 0.16 0.05 O a. Polycarbonate with a monomer molecular weight of 61.0168 g/mol b. Polystyrene with a monomer molecular weight of 104.15 g/mol Oc. Polypropylene with a monomer molecular weight of 42.08 g/mol O d. Polyvinyl chloride with a monomer molecular weight of 62.498 g/mol Oe. Polymethylmethacrylate with a monomer molecular weight of 100.121 g/mol
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter23: Carbon: Not Just Another Element
Section: Chapter Questions
Problem 120SCQ
Related questions
Question
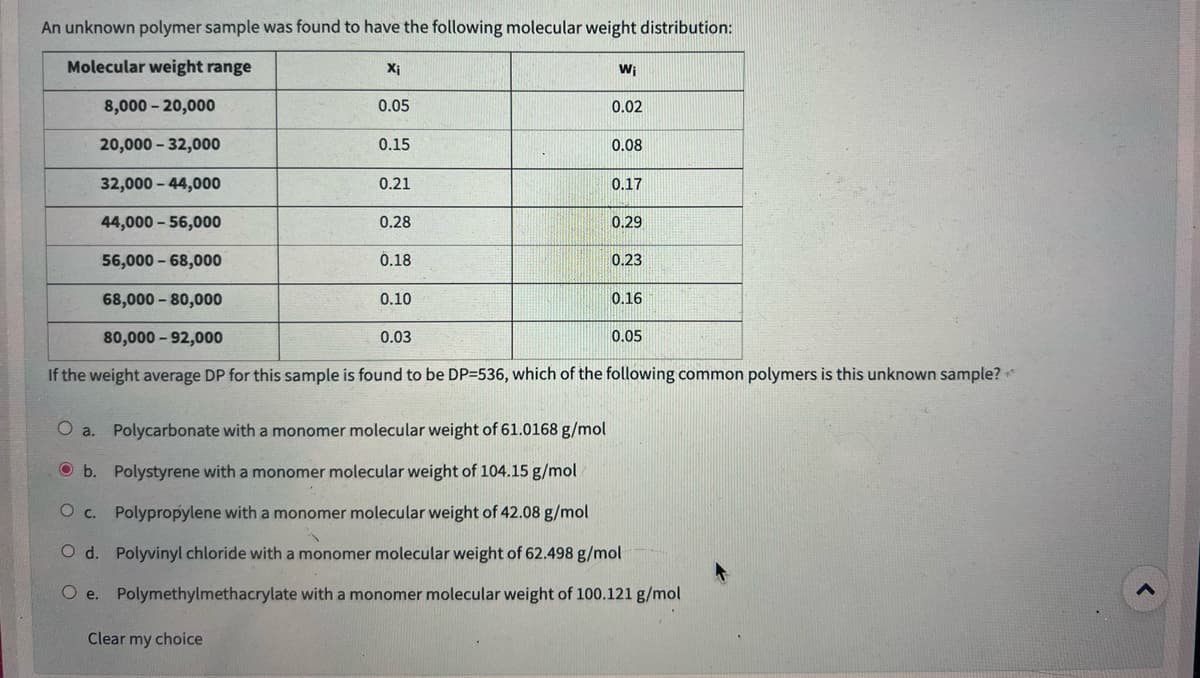
Transcribed Image Text:An unknown polymer sample was found to have the following molecular weight distribution:
Molecular weight range
8,000-20,000
20,000 -32,000
32,000 - 44,000
44,000 - 56,000
56,000 - 68,000
68,000 -80,000
X₁
0.05
Clear my choice
0.15
0.21
0.28
0.18
0.10
0.03
Wi
0.02
0.08
0.17
0.29
0.23
0.16
80,000-92,000
If the weight average DP for this sample is found to be DP=536, which of the following common polymers is this unknown sample? **
0.05
O a. Polycarbonate with a monomer molecular weight of 61.0168 g/mol
Ob. Polystyrene with a monomer molecular weight of 104.15 g/mol
O c. Polypropylene with a monomer molecular weight of 42.08 g/mol
Od. Polyvinyl chloride with a monomer molecular weight of 62.498 g/mol
Oe. Polymethylmethacrylate with a monomer molecular weight of 100.121 g/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning