485 J of work must be done to compress a gas to half its initial volume at constant temperature. Part A How much work must be done to compress the gas by a factor of 10.0, starting from its initial volume? Express your answer with the appropriate units. µA ? Value Units
485 J of work must be done to compress a gas to half its initial volume at constant temperature. Part A How much work must be done to compress the gas by a factor of 10.0, starting from its initial volume? Express your answer with the appropriate units. µA ? Value Units
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 81AP: One process for decaffeinating coffee uses carbon dioxide ( M=44.0 g/mol) at a molar density of...
Related questions
Question
19.8 Please help me answer this physics question
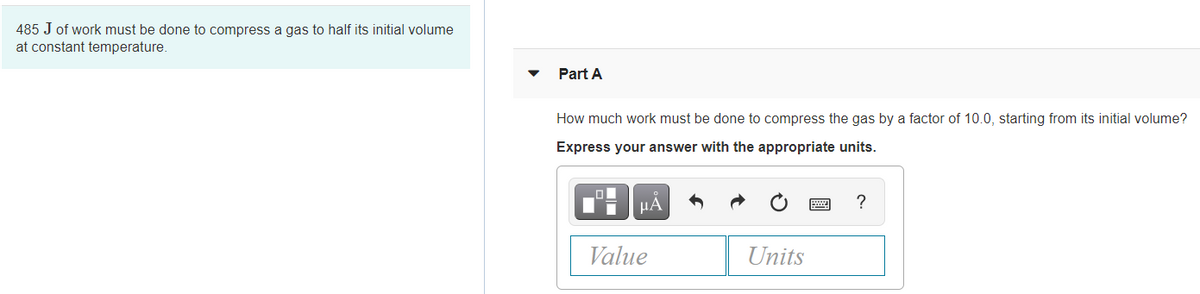
Transcribed Image Text:485 J of work must be done to compress a gas to half its initial volume
at constant temperature.
Part A
How much work must be done to compress the gas by a factor of 10.0, starting from its initial volume?
Express your answer with the appropriate units.
HÁ
?
Value
Units
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


University Physics Volume 1
Physics
ISBN:
9781938168277
Author:
William Moebs, Samuel J. Ling, Jeff Sanny
Publisher:
OpenStax - Rice University

College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College

An Introduction to Physical Science
Physics
ISBN:
9781305079137
Author:
James Shipman, Jerry D. Wilson, Charles A. Higgins, Omar Torres
Publisher:
Cengage Learning