4NH3(g) + 3O2(g) ⇌ 2N2(g) + 6H2O(g) If a chemist mixed 2.930 moles of ammonia (NH3) gas with 1.978 moles of oxygen (O2) gas in a 7.95 Liter airtight metal flask at 127°C and after the pressures stabilize at equilibrium and then draws an air sample and found 0.683 moles of nitrogen (N2) gas then how many moles of ammonia (NH3) is present at equilibrium?
4NH3(g) + 3O2(g) ⇌ 2N2(g) + 6H2O(g) If a chemist mixed 2.930 moles of ammonia (NH3) gas with 1.978 moles of oxygen (O2) gas in a 7.95 Liter airtight metal flask at 127°C and after the pressures stabilize at equilibrium and then draws an air sample and found 0.683 moles of nitrogen (N2) gas then how many moles of ammonia (NH3) is present at equilibrium?
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter13: Solutions And Their Behavior
Section13.5: Colloids
Problem 2.4ACP
Related questions
Question
4NH3(g) + 3O2(g) ⇌ 2N2(g) + 6H2O(g)
If a chemist mixed 2.930 moles of ammonia (NH3) gas with 1.978 moles of oxygen (O2) gas in a 7.95 Liter airtight metal flask at 127°C and after the pressures stabilize at equilibrium and then draws an air sample and found 0.683 moles of nitrogen (N2) gas then how many moles of ammonia (NH3) is present at equilibrium?
Transcribed Image Text:hitrogeni riz is sclubie in bl
N2, s Soluble in.
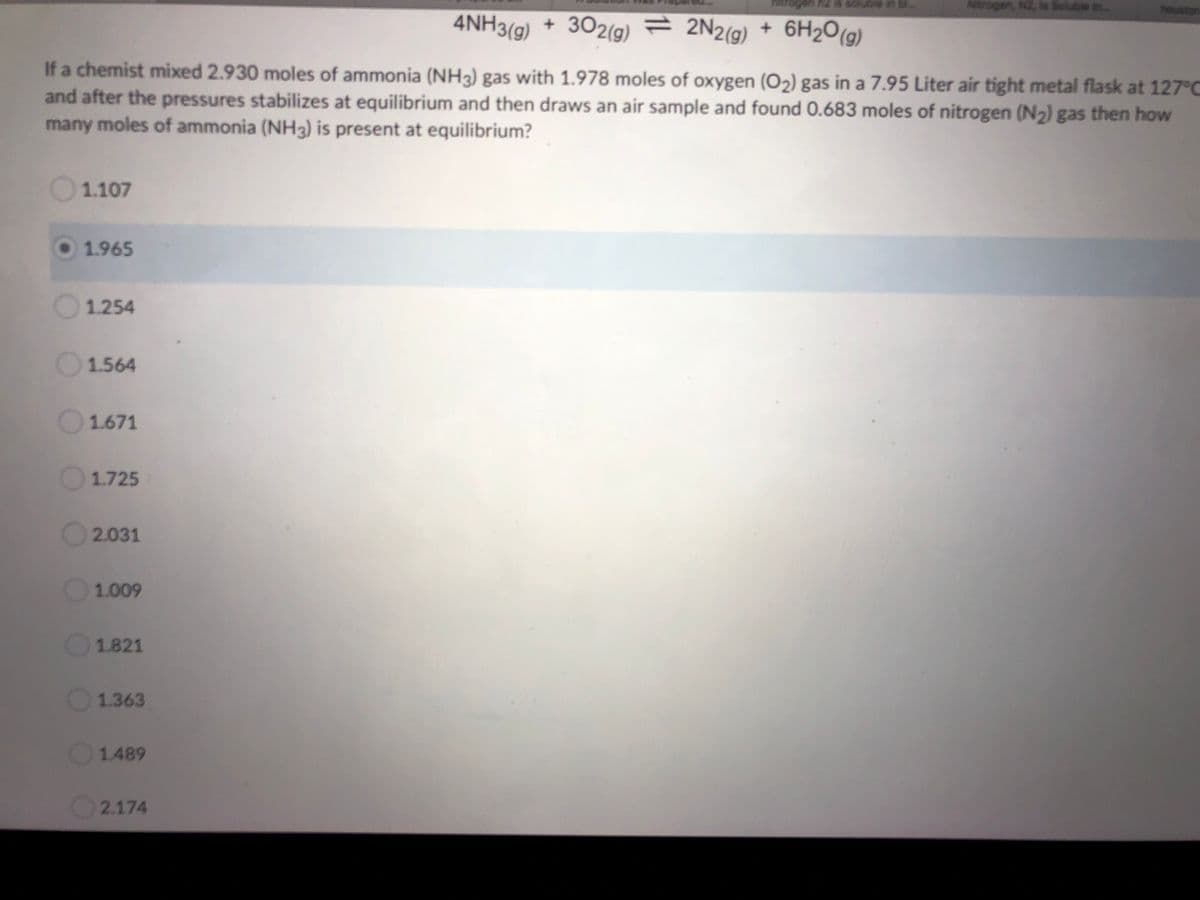
4NH3(g) + 302(g) = 2N2(g) + 6H2O(g)
If a chemist mixed 2.930 moles of ammonia (NH3) gas with 1.978 moles of oxygen (O2) gas in a 7.95 Liter air tight metal flask at 127°C
and after the pressures stabilizes at equilibrium and then draws an air sample and found 0.683 moles of nitrogen (N2) gas then how
many moles of ammonia (NH3) is present at equilibrium?
1.107
1.965
1.254
1.564
1.671
1.725
2.031
1.009
1.821
1.363
1.489
2.174
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning