5 6 C 10.81 12.01 13 14 Al Si 26.98 28.09 22 23 24 25 26 27 28 29 30 31 32 Ti Cr Mn Fe Zn Ga Ge 96 47.90 50.94 52.00 54.94 55.85 58.93 58.70 63.55 65.38 69.72 72.59 41 42 43 44 45 46 47 48 50 Nb Mo 95.94 197.91] 101.07 102.91 Ru Rh d Ag Cd Sn 91 91.22 92.91 106.4 107.87 112.41 114.82 118.69 71 72 73 74 75 76 77 78 79 80 81 82 Au Hg Pb 178.49 180.95 183.85 186.21 190.2 192.22 195.05 196.97 200.59 204.37 207.2 03 104 105 106 107 108 109 110 111 112 113 114 Db Sg Hs Rg Cn Nh FI 1265.121 1268.131 1271.131 (270) 1277.15| (276.15 281.161 1280.161|1285.17l 1284.181|1289.19 58 59 60 61 62 63 64 65 66 67 68 Pr Nd Pm Sm Eu Gd Tb Dy Ho Er 151.96 157.25 158.93 Ce 140.12 140.91 144.24 [145] 150.4 164.93 167.26 90 91 92 93 94 95 96 97 98 99 100
States of Matter
The substance that constitutes everything in the universe is known as matter. Matter comprises atoms which in turn are composed of electrons, protons, and neutrons. Different atoms combine together to give rise to molecules that act as a foundation for all kinds of substances. There are five states of matter based on their energies of attraction, namely solid, liquid, gases, plasma, and BEC (Bose-Einstein condensates).
Chemical Reactions and Equations
When a chemical species is transformed into another chemical species it is said to have undergone a chemical reaction. It consists of breaking existing bonds and forming new bonds by changing the position of electrons. These reactions are best explained using a chemical equation.
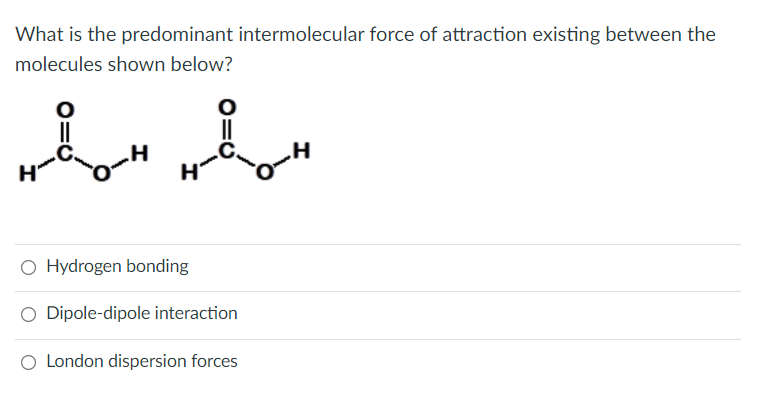
(3.) This is urgent. Please answer ASAP, thanks. I will live thumbs up afterwards
![1
2
H
Не
1.008
4.00
3
5
6.
10
Ве
В
Ne
6.94
9.01
10.81
12.01
14.00
16.00
19.00
20.18
11
12
13
14
15
16
17
18
Na Mg
Al
Ar
22.99
24.31
26.98
28.09
30.97
32.06
35.45
39.95
19
20
21
22
23
24
25
26
27
28
29
30
31
32
34
35
36
Mn
Ga | Ge
Kr
39.10
40.10
44.96
47.90
50.94
52.00
54.94
55.85
58.93
58.70
63.55
65.38
69.72
72.59
74.92
78.96
79.90
83.80
37
38
39
41
42
45
46
49
52
53
54
Rb
Nb Mo
Pd Ag
Хе
85.47
87.62
88.91
91.22
92.91.
95.94
[97.91) 101.07
102.91
106.4
107.87
112.41
114.82
118.69
121.75
127.60
126.90
131.30
55
56
57-71
72
73
74
75
76
77
78
79
80
81
82
83
84
85
86
Cs
Та
Rn
132.91
137.33
178.49
180.95
183.85
186.21
190.2
192.22
195.05
196.97
200.59
204.37
207.2
208.98
[209]
[210]
[222]
87
88
89-103
104
105
106
107
108
109
110
112
113
114
115
116
117
118
Fr
Ra
1223.02) (226.03]
Db Sg
Rg | Cn Nh
Og
[294]
Mt
FI
[265.12 1268.131 [271.131 (270) 1277.15|| [276.15) (281.16)] 280.16] (285.17) (284.18]|1289.19)|1288.191 [293]
[294]
57
58
59
60
61
62
63
64
65
66
67
68
69
70
71
Lanthanides La
Pr
Nd Pm Sm Eu
173.04 174.97
138.91
89
140.12
140.91
144.24
[145]
150.4
151.96
157.25
158.93
162.50
164.93
167.26
168.93
91
92
93
94
95
96
97
99
100
101
102
Pa
231.04 238.029 [237.05] [244.06] [243.06] [247.07] [247.07] [251.08] [252.08] [257.10] [258.10] [259.10] [262.11]
Np Pu Am Cm
Fm M
Actinides
[277.03] 232.04](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Ffafbb92d-a229-4527-8115-8040b9e90ba1%2Feb34a46c-2618-4e0a-a44e-5828b9b5d474%2Fgiykeyi_processed.jpeg&w=3840&q=75)
Step by step
Solved in 2 steps









