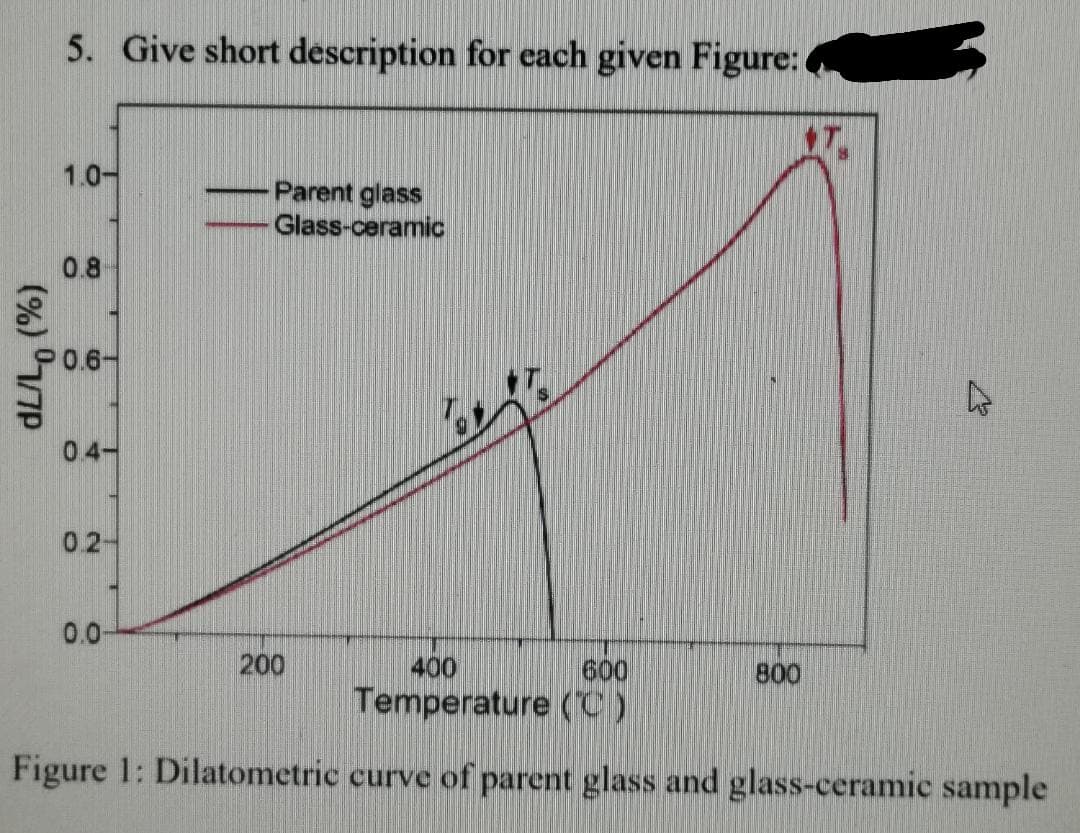
5. Give short description for each given Figure: 1.0- Parent glass Glass-ceramic 0.8 0.6- 04- 0.2- 0.0- 200 400 600 800 Temperature (C) Figure 1: Dilatometric curve of parent glass and glass-ceramic sample (%) 7P
Q: What is the pH of a solution made by mixing 25.00mL of .1500M NaOH with 25.00mL of .2500M HClO2?
A: HClO2 is Chlorous acid which is a weak acid. Weak acid dissociate weakly in aqueous solutions.…
Q: 4.. After mixing 47 grams of glucose in 5 litres of 1X PBS, the final solution is %. A. 0.0094 B.…
A: Given Values: Weight of glucose = 47 grams The volume of the solution in which 47 grams of glucose…
Q: 1. Based on Figure 2, at 210 sec, the thermometer reading in the 30°C air was most likely closest to…
A: Answer:- Option D seems to be the correct choice. The temperature at 200 seconds is as marked in the…
Q: 2. Which of the following are physical properties of covalent network solids? Choose ALL that apply.…
A: Covalent network solids are a special group of molecules bonded b covalent bonds. The bonds from 2D…
Q: 8 7- 6- 2- 1 405 180 225 270 315 [Substrate] (nM) 45 90 135 360 450 495 540 58! What are the units…
A: Solution Michaelis menten kinetics is one of the best models of enzyme kinetics. Value of…
Q: 8. When evaporation process is done, what happens to the liquid substance in a mixture? * O A. It…
A: 8. Evaporation is the process of separating dissolved solid in liquid. the liquid gets evaporated to…
Q: 2. What concentration of H (aq) corresponds to each of the following? pH = 10.92 РОН %3D4.32 РОН %3D…
A: pH is the measure of the strength of H+ ion or Hydronium ions in solution. pH at which 50%…
Q: Alanine has pKa values of 2.4 and 9.9. Its isoelectric point, pI, is equal to____. A. 6.15 B. 23.76…
A: Amino acids contain an alpha-carboxylic acid group and an alpha-amino group. Every amino acid has…
Q: 1. If I make a solution by adding 83 grams of sodium hydroxide (NaOH) to water to give 750 mL of…
A: Number of moles (mol) = Mass (g) / Molar Mass (g/mol)Concentration (g/L) = Mass (g) / Volume…
Q: 2. Describe how you would prepare the following solutions: d) 250 mL of 0.10 M NH3 from a 14.8 M…
A: To prepare 250ml of 0.10M solution we need to dilute the 14.8M stock solution. The dilution is…
Q: 10. A neutral aqueous solution has a 7.0 M H3O+ concentration b. contains neither hydronium ions nor…
A: The acidity of an aqueous solution is determined by the concentration of protons or hydronium ions…
Q: 1. Choose the one answer that fits best. One mole is a. A baker's dozen b. One molecule of a given…
A: "Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 1) A solution is made by combining 128 mL of ethanol and 0.452 L of water. In this solution, what is…
A: Hi! Thanks for your question. But as you have posted multiple questions, I am answering only the…
Q: Potatoes contain carbohydrates. Which form of carbohydrate do they contain? Part Stomach secretes a…
A: since you have posted multiple questions we'll help you out with the first two. potatoes are…
Q: The molecular weight of ethanol (CH3CH2OH) is46 and its density is 0.789 g/cm3.A. What is the…
A: Ans: Molarity: The moles of solute per liter of the solution is defined as Molarity. All…
Q: 6. CONSTANT VOLUME CALORIMETRY Using a bomb calorimeter, 1.0 g of benzene (CeHs), a component of…
A: A bomb calorimeter is used to measure the heat released during the combustion of a sample. The basic…
Q: 1.What is the molar concentration of a 15% glycerol solution?…
A: For the first question, 15% glycerol solution represents that 15g of glycerol is dissolved in 100mL…
Q: 1. How much NaOH(in grams) is required to prepare 0.2 M of 250 m! NaOH?(Naoh Ma= 40g/mo)
A: Please note that keeping with the policy of the site and in order to provide accurate and quality…
Q: 4) A doctor ordered Mesalamine oral suspension 300 mg every 2 hours; direction for mixing: A 125 mL…
A: Question is related to drug calculation. Doctor ordered mesalamine oral suspension 300 mg every 2…
Q: 2. How many cm are in 562 um? 210.0562 sa lo00 3. How many milligrams is 71 grams? 71grams x…
A: Dilution and Conversions in biology specially in laboratory experiments are really important because…
Q: 1. Particles are in close contact with one another leading to strong forces of attraction and…
A: There are four different states of matter. Solid,liquid,Gas,Plasma When small molecules are packed…
Q: Mobile phase can be : A) Solid or liquid B) Only solid C) Only gas D) Liquid or gas
A: Chromatography is a technique that is used to separate different components of a mixture as a result…
Q: When 5.58 g of a solid mixture composed of NH4CI and CaCl2 was dissolved in 100.0 mL of water, the…
A: 2 questions are asked. According to guidelines, will answer the 1st question only. 100 ml water…
Q: 1. How many mL of a ½ strength magnesium sulfate solution will contain 10 grams of magnesium…
A: As per question, 1ml MgSO4 contain 1/2gm MgSO4 (concentration is 50%) So 10gm MgSO4 will contain ,…
Q: 10. Identify the following as either chemical change (C) or physical change (P) baking a pie melting…
A: A chemical change is a process during which one or more ingredient (reactant) chemically reacts to…
Q: 9. What is the strength (expressed 250 mL solution that contains 5 g of NaCl? as a percent) of a
A: Here we have to calculate the strength of a 250 ml solution that contains 5 g of NaCl.
Q: 4. A bilayer polymer slab (1.2m long, 1.4m wide, 5 cm thick) is placed on a hot plate, which keeps…
A: This answer would have solved so nicely by handwritten note but you aksed for digitally so here you…
Q: 1. Convert the following measurements: Imm 1000um a 92 mm b. 5900 um mm
A: DISCLAIMER: Since you have asked multiple question, we will solve the first question for you. If you…
Q: 8. A Patient received three different I.V. drug, each mixed in 50 ml of fluid, over 8 hours. The…
A: I.V (Intravenous fluid) is the amount of fluid administered through the bloodstream or…
Q: If you need to set an experiment at 0 degree C, what will be the physical state of the water in the…
A: Water is present in three forms: solid, liquid, and gas. Conversion from one form to another depends…
Q: 22) A mixture of NaCl and starch is placed in a dialyzing bag and immersed in distilled water. Which…
A: Dialysis is considered as the process, during which using filters blood is purified. This helps to…
Q: 7. If the concentration of Mg in water is 36.6mg/L, what is the hardness of the water as mg/L CACO3?…
A: Hardness of water is defined as amount of dissolved calcium and magnesium in the water. the degree…
Q: 10. It was found that At coordinates to a metal in a 4:1 stoichiometry. Noting that A has a ba…
A: Biological important of coordination compounds like the heme group, which is Fe-Porphyrin complex…
Q: 8. At 100 °C, 1.00 gram of liquid water has a volume of 1.043 mL. What volume will 1.00 gram of…
A: Internal energy (U) is the sum of kinetic and potential energy associated with the random motion of…
Q: How many mL of distilled water must be added to make the Benzalkonium chloride preparation isotonic
A: Calculate in next step
Q: 05:Calculate the grams of each substance requied to prepare the following solutions:(a)250 ml of 0.1…
A: Multiple questions asked. 1st question will be answered, as permissible by guidelines. Asked :…
Q: 3. Behind Lin's house there is a kiddie pool that holds 180 or 680 units of wate depending on which…
A: Kiddie pool is generally small pool and there depth is low so that kiddie don't get drowned
Q: 1. A solution containing polymer of an unknown structure, and with an approximate molar mass of 1.75…
A: Osmotic pressure can be defined as the minimum pressure that must be applied to halt the flow of…
Q: 3) The Chemical Solution determined by lambda max comaX ) oF a should be 9) The absorbtion specturm…
A: Lambda is considered as the eleventh letter in Greek alphabet. It has been demonstrated on…
Q: 1. Explain the X-ray spectrum given in the figure below. Kaz liigu 80 90 Relative output 0 10 20 30…
A: Introduction An X-ray, also known as X-radiation, is a type of high-energy electromagnetic radiation…
Q: Find the final concentration as a percent (w/v) of dextrose when 75 mL dextrose 50 % (w/v), 50 mL of…
A: There are different ways to denote the concentration of a solution. Per cent (w/v) means there is 1…
Q: 1. An artificial membrane with a pore size of 24 Angstrom (Å) separates two chambers in glassware…
A: For getting the right concentration of the solution we should take right concentration of…
Q: 1) A student measures the mass of an 8 cm3 block of brown sugar to be 12.9 g. What is the density of…
A: Density is a measure of mass per volume. Density and Volume are inversely proportional to each other
Q: 1. A pediatric vitamin drug product contains the equivalent of 0.5 mg of fluoride ion in each…
A: Drug contains equivalent of 0.5 mg fluoride in each milliter. This means that in 1 mL, 0.5 mg…
Q: 3. Two objects are moving at the same speed. The objects have different ar Which of the following…
A:
Q: 6. A student used Dumas procedure to calculate the Molar mass of a volatile liquid and collected…
A: Given, P = 750 mmHg = 750/760 = 0.987 atm R= 0.0821 atm•L/mol•K T T = 100.25 degrees celcius =…
Step by step
Solved in 2 steps

- 6. Your pharmacy has on hand Robitussin CF Syrup (guaifenesin 100 mg/1 tsp).You receive a prescription for Robitussin CF Syrup and the patient is to receive 250 mg of guaifenesin q4h. How many milliliters will the patient receive in 1 dose? If you dispense a 4oz bottle of syrup, how many doses will the patient receive?The distance between k and e would be approximately... a) 6.25 cM b) 6.6 cM c) 4.65 cM d) 5.0 cM e) 11.65 cM9. Find the final concentration as a percent (w/v) of dextrose when 75 mL dextrose 50 % (w/v), 50 mL of sodium chloride 0.9 % (w/v), and 100 mL of dextrose 5 % (w/v) sodium chloride 0.45 % (w/v) solution are mixed together.
- 2. a. What ingredient(s) necessary to white chocolate does this product have? b. Why is this product labeled as “white chocolate style” instead of being simply labeled as “White chocolate”?A solution contains 0.25 M Ni(NO3)2 and 0.25 M Cu(NO3)2. A. Can the metal ions be separated by slowly adding Na2CO3 aq solution? Assume that for successful separation 99% of the metal ion must be precipitated before the other metal ion begins to precipitate, and assume no volume change on addition of Na2CO3 solution. Ksp of CuCO3 = 1.4 x10-10 Ksp of NiCO3 = 1.42X 10-7 B. If you need to redissolve the salt that was first precipitated, what pH will you use and why ? Write your answers in template provided. Upload your step by step solution for A.The iodine contained in every one mL of the 0.010 M iodine solution in the buret will react with 1.76 mg of vitamin C. (In other words, every mL of iodine solution needed for the titration indicates the presence of 1.76 mg of vitamin C in the juice sample.) If your titration used 7.11 mL of iodine solution, what mass of vitamin C is in your fruit juice sample (in milligrams, mg)? Include the unit and two decimal places in your answer.
- 1) the choices for configuration is D or L31. The formula for a cosmetic cream calls for 5% of an emulsifier blend consisting of Span 60 and Tween20. If the Required HLB of the oil phase is 14.0, how much of each of the emulsifiers, respectively, shouldbe used in preparing 500 grams of the cream? (HLB for Span 60 = 4.7; HLB for Tween 20 = 16.7)a. 5.6 grams; 19.4 gramsb. 8.4 grams; 16.6 gramsc. 12.5 grams; 12.5 gramsd. 15.2 grams; 9.8 gramsSpecific rotation of L-alanine [(+)alanine) in water (at 25 °C) is +2.8. Suppose you have 2.00 g of mixture of L and D-alanine [(-)alanine]. You dissolved it in 10 mL of water, and measured rotation in 10 cm cell. The observed rotation was +0.12. Calculate the ee% for the mixture. How many grams of D- and L-alanine do you have in the sample?
- 3. a) An aqueous solution is 3.50% by mass potassium bromide, KBr, and has a density of 1.02 g/mL. The molality of potassium bromide in the solution is _________m. b)An aqueous solution of magnesium nitrate has a concentration of 0.474 molal. The percent by mass of magnesium nitrate in the solution is _________%. c)An aqueous solution of cobalt(II) bromide has a concentration of 0.153 molal. The percent by mass of cobalt(II) bromide in the solution is _________%.1) If you have 2.5g of hydrocortisone POWDER, how many grams of a 0.05% ointment can be compounded? 2) How many mL of a 0.45% saline solution can be made with 6750 mg of sodium chloride? 3) Your pharmacy receives an order to compound 120 g of a 1.25% cream. How many mg of drug are required to compound this order? 4) Is it possible to make 1200 mL of a 30% dextrose solution with only 75 g of dextrose? 5) You are asked to compound a 15% zinc oxide cream. How many grams of this cream can you make using 30,000mg of zinc oxide? 6) Using 45g of acetic acid, how many mL of a 20% acetic acid solution can be made? 7) Calculate the percent strength of a solution when 8 g of drug is dissolved in 400 mL of solution. 8) Calculate the percent strength of a solution when 2.5 g of drug is dissolved in 750 mL of solution. 9) Calculate the percent strength of a solution when 5000 mg of drug is dissolved in 500 mL of solution.Solution A is 20 degrees celsius, Solution B is 80 degrees celsius (both are the same kind of solution). The goal is to make a 50 degree celsius solution; How much of solution A do you need (starting with 100g of solution B)? (cp=4.184 J/g C)





