5. The shelf life of pharmaceuticals is an important issue, because drugs may decompose as they age, transforming into inactive or harmful compounds. Shown below are the structures of aspirin (acetylsalicylic acid) and acetaminophen. Refer to the structures to explain the following two observations: • Acetaminophen tablets can be stored for years with no apparent change in activity, while aspirin slowly decomposes over time. • Acetaminophen is routinely included in liquid preparations such as Nyquil, where it is dissolved in water, but aspirin is never sold as a solution. ar or CH3 CH2 соон но acetylsalicylic acid acetominophen
5. The shelf life of pharmaceuticals is an important issue, because drugs may decompose as they age, transforming into inactive or harmful compounds. Shown below are the structures of aspirin (acetylsalicylic acid) and acetaminophen. Refer to the structures to explain the following two observations: • Acetaminophen tablets can be stored for years with no apparent change in activity, while aspirin slowly decomposes over time. • Acetaminophen is routinely included in liquid preparations such as Nyquil, where it is dissolved in water, but aspirin is never sold as a solution. ar or CH3 CH2 соон но acetylsalicylic acid acetominophen
Chapter5: Calculating Adult Dosages: Oral And Parenteral Forms
Section: Chapter Questions
Problem 1RP
Related questions
Question
please explain
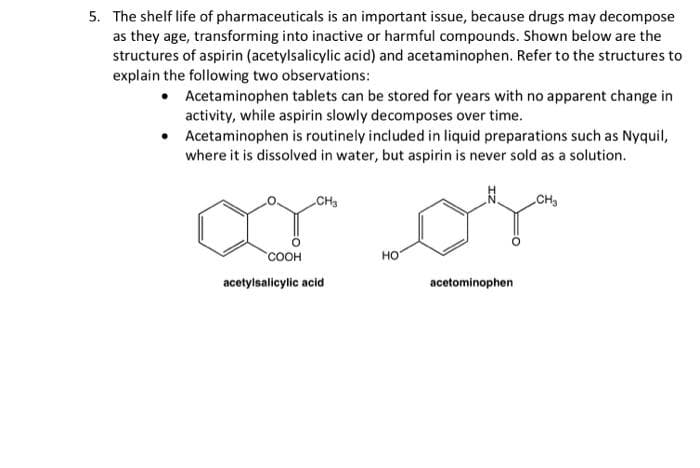
Transcribed Image Text:5. The shelf life of pharmaceuticals is an important issue, because drugs may decompose
as they age, transforming into inactive or harmful compounds. Shown below are the
structures of aspirin (acetylsalicylic acid) and acetaminophen. Refer to the structures to
explain the following two observations:
• Acetaminophen tablets can be stored for years with no apparent change in
activity, while aspirin slowly decomposes over time.
Acetaminophen is routinely included in liquid preparations such as Nyquil,
where it is dissolved in water, but aspirin is never sold as a solution.
ar or
CH3
CH3
соон
но
acetylsalicylic acid
acetominophen
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

