5.8871 1. Mass NaCi (g) 100.0 2. Volume of water (mL) 51.5023 3. Mass ice (g) -2.85 Freezing point of the solution ('C) Part 1 Calculations What is the predicted value of i for NaC? For electrolytes, assume i the number of ions per formula unit. Calculate the molality of the solution (m) Use the equation: T, (zolution) - T, (zolvent) = -i K; m The freezing point of pure water is 0.00 °C Ky for water is 1.86 'C/m Mass of solvent in grams Add together the masses of the liquid water and ice added to the calorimeter. Convert the volume of liquid water to mass using 1.000 g/ml as the density of water (see procedure for volume used). of
5.8871 1. Mass NaCi (g) 100.0 2. Volume of water (mL) 51.5023 3. Mass ice (g) -2.85 Freezing point of the solution ('C) Part 1 Calculations What is the predicted value of i for NaC? For electrolytes, assume i the number of ions per formula unit. Calculate the molality of the solution (m) Use the equation: T, (zolution) - T, (zolvent) = -i K; m The freezing point of pure water is 0.00 °C Ky for water is 1.86 'C/m Mass of solvent in grams Add together the masses of the liquid water and ice added to the calorimeter. Convert the volume of liquid water to mass using 1.000 g/ml as the density of water (see procedure for volume used). of
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 42QAP: Antifreeze solutions are aqueous solutions of ethylene glycol, C2H6O2(d=1.12g/mL). In Connecticut,...
Related questions
Question
Moles of solute (mol)
Use the molality to convert kg of solvent to moles of solute.
Hints: molality of the solution (m) 0.76612903225806
Mass of solvent in grams 151.5
Mass of solvent in kilograms 0.1515
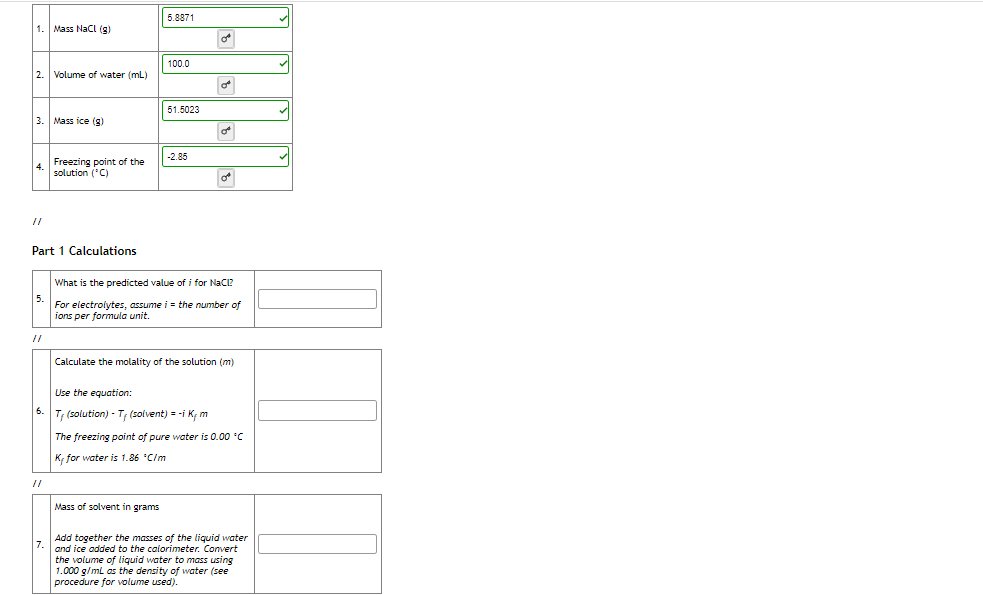
Transcribed Image Text:5.8871
Mass Nacl (g)
100.0
2.
Volume of water (mL)
51.5023
3.
Mass ice (g)
-2.85
Freezing point of the
4.
solution ('C)
Part 1 Calculations
What is the predicted value of i for NaCi?
For electrolytes, assume i = the number of
ions per formula unit.
//
Calculate the molality of the solution (m)
Use the equation:
6.
T; (solution) - T; (solvent) = -i K, m
The freezing point of pure water is 0.00 C
K, for water is 1.86 "C/m
//
Mass of solvent in grams
Add together the masses of the liquid water
and ice added to the calorimeter. Convert
the volume of liquid water to mass using
1.000 g/ml as the density of water (see
procedure for volume used).
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning