50. Using bond energies given in Table 1 on page 307, determine the enthalpy change for the reaction given by the following balanced chemical equation: (5.3) mm H,(g) + Cl,(g)→ 2 HCI(g)
50. Using bond energies given in Table 1 on page 307, determine the enthalpy change for the reaction given by the following balanced chemical equation: (5.3) mm H,(g) + Cl,(g)→ 2 HCI(g)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter8: Thermochemistry
Section: Chapter Questions
Problem 26QAP: Nitroglycerin, C3H5(NO3)3(l), is an explosive most often used in mine or quarry blasting. It is a...
Related questions
Question
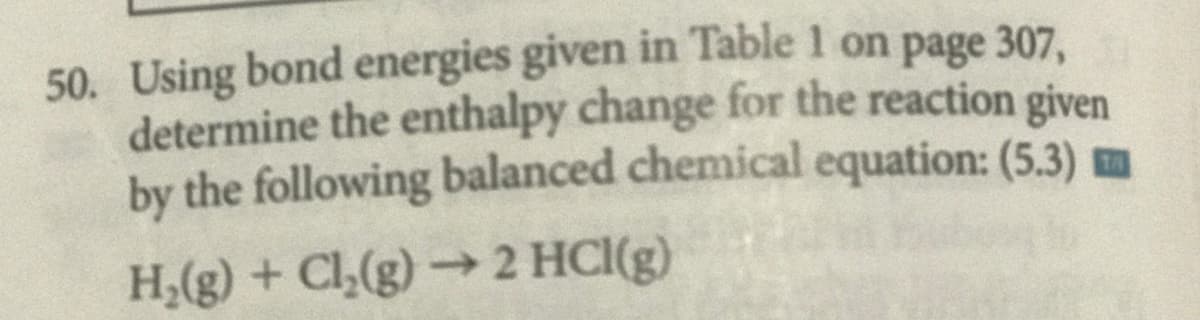
Transcribed Image Text:50. Using bond energies given in Table 1 on page 307,
determine the enthalpy change for the reaction given
by the following balanced chemical equation: (5.3) m
H;(g) + Cl,(g) → 2 HCI(g)
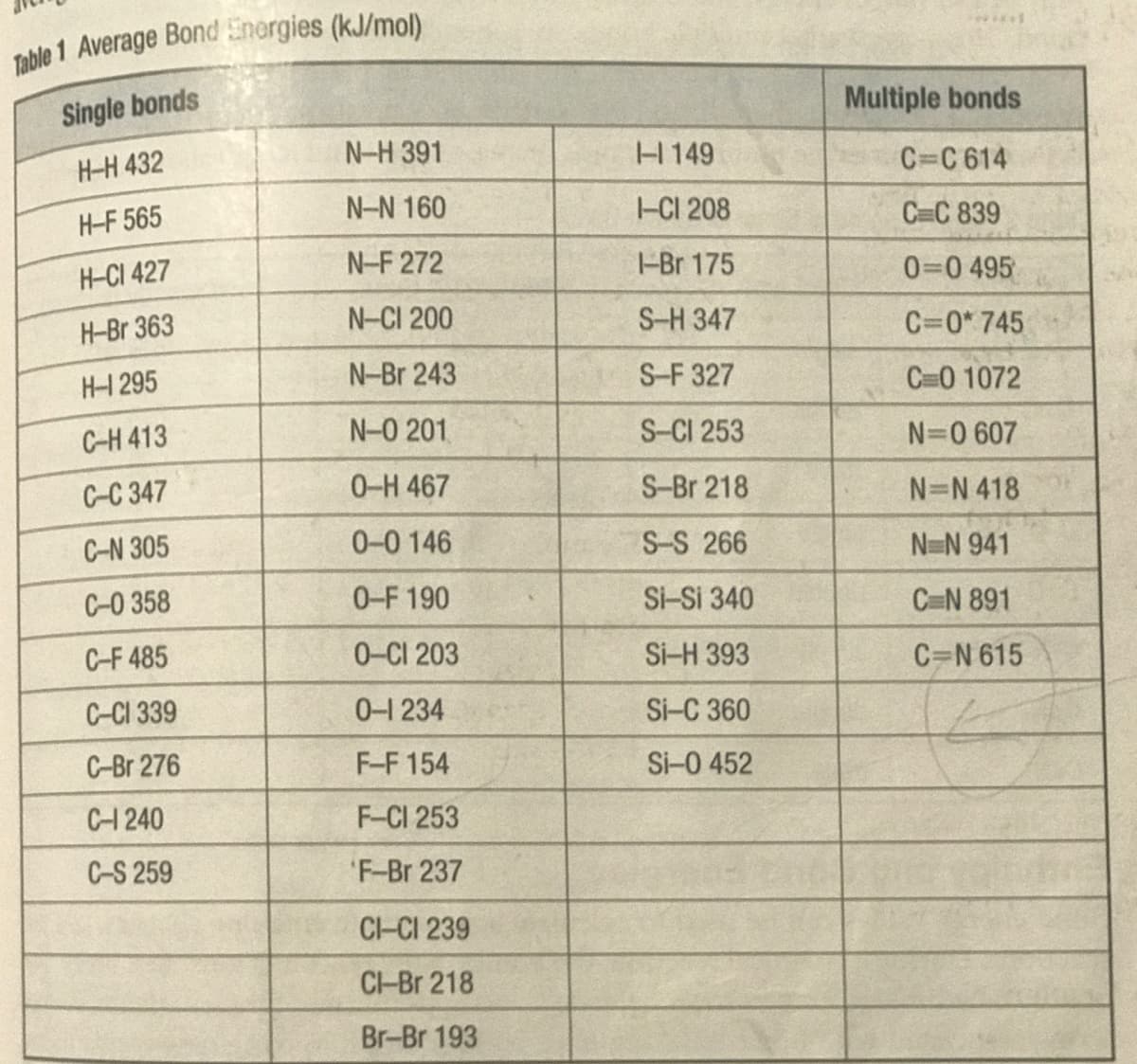
Transcribed Image Text:Single bonds
Multiple bonds
H-H 432
N-H 391
H149
C=C 614
H-F 565
N-N 160
-CI 208
C C 839
H-CI 427
N-F 272
-Br 175
0=0 495
H-Br 363
N-CI 200
S-H 347
C=0* 745
H-1 295
N-Br 243
S-F 327
C=0 1072
C-H 413
N-0 201
S-CI 253
N=0 607
C-C 347
0-H 467
S-Br 218
N=N 418
C-N 305
0-0 146
S-S 266
N-N 941
C-0 358
0-F 190
Si-Si 340
C=N 891
C-F 485
0-CI 203
Si-H 393
C=N 615
C-CI 339
0-1 234
Si-C 360
C-Br 276
F-F 154
Si-0 452
C-1240
F-CI 253
C-S 259
F-Br 237
CI-CI 239
CI-Br 218
Br-Br 193
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning