(5,452 unread) - dnld2005@yaho X Microsoft PowerPoint CHEM 14 x Course Home lum.ecollege.com/course.html?courseld%3D15698036&OpenVellumHMAC=Dd20089920194357bb0a06780cebbff9b#10001
(5,452 unread) - dnld2005@yaho X Microsoft PowerPoint CHEM 14 x Course Home lum.ecollege.com/course.html?courseld%3D15698036&OpenVellumHMAC=Dd20089920194357bb0a06780cebbff9b#10001
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Transcribed Image Text:(5,452 unread) - dnld2005@yaho X
Microsoft PowerPoint CHEM 14 x
Course Home
lum.ecollege.com/course.html?courseld%3D15698036&OpenVellumHMAC=Dd20089920194357bb0a06780cebbff9b#10001
<CH 13 HW
Exercise 13.42
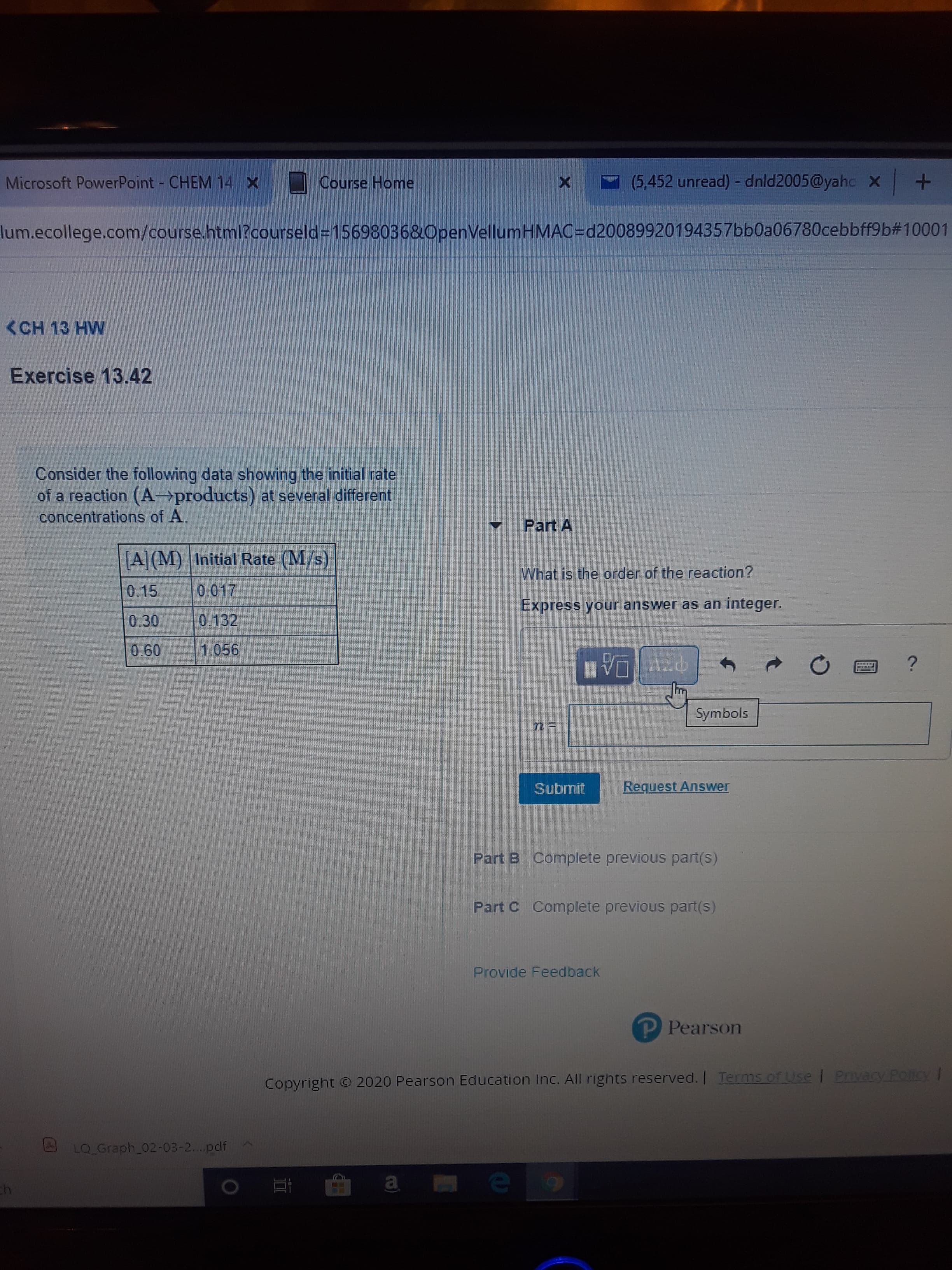
Consider the following data showing the initial rate
of a reaction (A products) at several different
concentrations of A.
Part A
A(M) Initial Rate (M/s)
What is the order of the reaction?
0.15
0.017
Express your answer as an integer.
0.132
0.30
0.60
1.056
ΑΣΦ
Symbols
Request Answer
Submit
Part B Complete previous part(s)
Part C Complete previous part(s)
Provide Feedback
P Pearson
Copyright © 2020 Pearson Education Inc. All rights reserved. I Terms of Use | Privacy Policy1
LQ Graph 02-03-2...pdf
a
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

Recommended textbooks for you

