The following data of decomposition reaction of thionyl chloride (SO2C2) were collected at a certain temperature and the concentration of SO2Cl2 were monitored as shown in the table. SO2Cl2 (g) SO2 (g) Cl2 (g) Time (min) Conc. of SO2CI2 (mol/L) 0.1000 100 0.0876 200 0.0768 300 0.0673 400 0.0590 500 0.0517 600 0.0453 700 0.0397 800 0.0348 900 0.0305 1000 0.0267 1100 0.0234
The following data of decomposition reaction of thionyl chloride (SO2C2) were collected at a certain temperature and the concentration of SO2Cl2 were monitored as shown in the table. SO2Cl2 (g) SO2 (g) Cl2 (g) Time (min) Conc. of SO2CI2 (mol/L) 0.1000 100 0.0876 200 0.0768 300 0.0673 400 0.0590 500 0.0517 600 0.0453 700 0.0397 800 0.0348 900 0.0305 1000 0.0267 1100 0.0234
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.48PAE: 11.48 The following data were collected for the decomposition of NT),-: Time, f (min) [N2Os] (mol...
Related questions
Question
SO2Cl2 (g) ➞ SO2 (g) + Cl2 (g)
What will be the concentration of SO2Cl2 left in the reaction mixture at 1500 minutes?
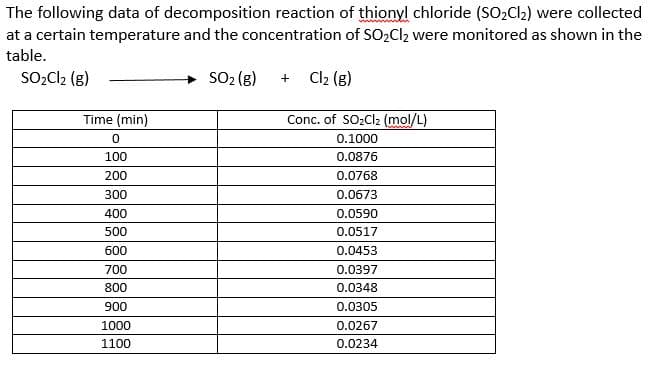
Transcribed Image Text:The following data of decomposition reaction of thionyl chloride (SO2C2) were collected
at a certain temperature and the concentration of SO2Cl2 were monitored as shown in the
table.
SO2Cl2 (g)
SO2 (g)
Cl2 (g)
Time (min)
Conc. of SO2CI2 (mol/L)
0.1000
100
0.0876
200
0.0768
300
0.0673
400
0.0590
500
0.0517
600
0.0453
700
0.0397
800
0.0348
900
0.0305
1000
0.0267
1100
0.0234
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning