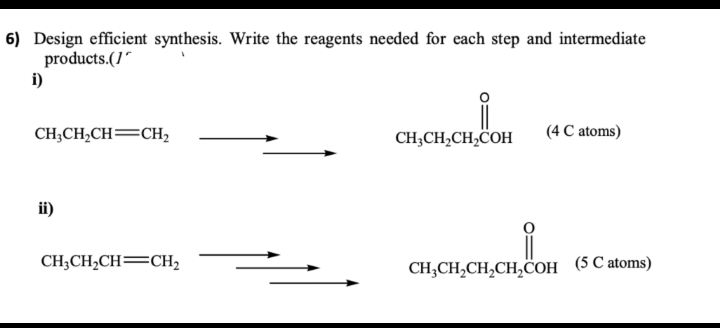
6) Design efficient synthesis. Write the reagents needed for each step and intermediate products.(1 i) CH3CH₂CH=CH₂ ii) CH3CH₂CH=CH₂ i CH3CH₂CH₂COH (4 C atoms) CH3CH₂CH₂CH₂COH (5 C atoms)
Q: 1. (A) HO OH (B) NHẠNH, KOH, heat (C)
A: In question 1, the 1st example is the ketal formation reaction, the 2nd example is the imine…
Q: CH3CHCH=CH₂ CH3 2 1 B₂H6 H₂O₂/OH*™
A: A question based on the reaction of alkene. For a given alkene the product of hydroboration…
Q: Q7. A sample consisting of 1 mol of perfect gas atoms (for which Cy=R) is taken through the cycle…
A: The main aim of this question is to calculate the q. w, AU, and H for each step and for the overall…
Q: Use of Baking Powder in baking (2NaHCO3 -> Na₂CO3 + CO₂ + H₂O )Identify what type of reaction it is.…
A: The objective of the question is to identify the type of chemical reaction involved in the use of…
Q: O C 1) NH3 2) H₂, Pd /C 3) aq. HCI но, Olese an HO !!!! NH₂ CH3
A: The objective of this question is to show product formation by using three different reagents. It…
Q: 9- Which of the following carbonyl groups exhibits the highest wavenumber in infrared spectroscopy?…
A: Two questions based on IR spectroscopy of organic compounds. One question is about the impact of…
Q: QUESTION 20 Which of these formulas is the expanded structural formula for an alkane with three…
A: Alkane is an organic compound containing only carbon and hydrogen atoms. The atoms are connected via…
Q: (iii) Discuss the mechanism of the reaction sequence shown in Figure 2. Make sure to include all…
A: The objective of the question is to discuss the mechanism of the given reaction sequence.
Q: What is name of this structure? Give explantion and steps for supporting.
A: IUPAC naming is a way to get the name of any compound depending on the number of carbon, functional…
Q: (a) A sample of linear polyethylene is dissolved in a large excess of xylene at 130 C, and dilute…
A: (a) The fine white suspension obtained after cooling the diluted solution of linear polyethylene in…
Q: Draw the mechanism of the addition of glucose to the cellulose chain. Note: Please provide a…
A: Cellulose is considered to be a straight chain polymers in which multiple glucose molecules are…
Q: A bowling ball that has a radius of 11.0 cm and a mass of 7.50 kg rolls without slipping on a level…
A: In the given scenario, we are presented with a bowling ball rolling without slipping on a level…
Q: Given the struct re of Katrinose below OH OH C HO- OH HO HO- OH OH OH OH A. Redraw the structure and…
A: The objective of the question is to find the monomer of the raffinose also the type of glycosidic…
Q: 22-Acetyl CoA can be produced from the metabolism of which of the following? A. Glucose B. Fatty…
A: The objective of this question is to determine the correct option which show the acetyl CoA produced…
Q: 4. Consider the system described by the following reaction: C₂H6(g) + O2(9) CO2(g) + H₂O AH-2847.6…
A: A question based on equilibrium. Based on the Le-Chatelier principle, the effect of various…
Q: q(t) h(t) h .g. (t) qo= R! To establish the non-sedentary and sedentary mass balance for the liquid…
A:
Q: For the reaction; CO (g) + Cl2 (g) -> COCl2 (g) Kc = 215 If I start with 2.00 M CO and 3.00 M…
A: Concentration of CO = 2.00 MConcentration of Cl2 = 3.00 MKc = 215
Q: Draw resonance structures for the following compounds and in each case specify the major and minor…
A: If a molecule consists of lone pair or pi electrons and they are in conjugation then the…
Q: What is an acceptable Newman projection for the molecule shown below, looking down the indicated…
A:
Q: Consider the following two possibilities for electron tran- sitions in a hydrogen atom, pictured…
A: The question pertains to the study of electron transitions within a hydrogen atom, particularly in…
Q: e. CH3 CH3 C=C CH3 -CH3 -CH3 CH3 Cl₂ CCI4 C11H17CI
A: The purpose of this mechanism involved chlroination in presence of solvent carbon tetrachloride.
Q: In a coffee cup calorimeter, 32.0 mL of 0.71 M nitric acid (HNO3) and 32.0 mL of 0.71 M potassium…
A: The objective of this question is to calculate the ∆Hrxn by using given data. The calorimetry method…
Q: Explain the stereoselecticity of this reaction
A: In stereoselective there is a choice of pathway and a more favourable product is formed in the…
Q: Rewrite this measurement with a simpler unit, if possible. kg-m 7.7 S'S Note: If you can simplify…
A: The question is based on the concept of unit conversions.we need to simplify the given expression in…
Q: xH1³~~xx- H Me MeLi 40% H₂SO4 LOH
A: In the first case there occurs 1,2 addition of carbonyl group as here methyl lithium is present.In…
Q: The "leaning" or "roof" effect Why equivalent protons do not show coupling VAB = 50 Hz VAB = 40 Hz B…
A: 1H-NMR is a spectroscopic analysis to analyze the structure of the molecules depending on the…
Q: d. CH3 CH3 + NBS A peroxide
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: (a) First, add enough electrons as either lone pairs or r bonds to give an overall negative charge.…
A: In drawing Lewis structure, total valence electrons of all the atoms should be equal to total…
Q: For EACH of the following reactions (a)-(d) provide a curved arrow mechanism and where appropriate…
A: The aim is to explain the product formation by using different mechanism in organic synthesis. It…
Q: Give detailed Solution with explanation needed...don't give Handwritten answer. Choose the correct…
A: From the above reaction scheme we see that , In the first step presence of base it abstract acidic…
Q: 15. What type of nucleophilic aromatic substitution is the reaction of m-dinitrophenol with NaOH…
A: The objective of the question is to predict the type of nucleophilic aromatic substitution for the…
Q: Propose a curved arrow mechanism to account for the following transformation. ².8 MeO "Bu3P HO 38…
A: A question based on reaction mechanism. The reactant of nucleophilic addition reaction is given…
Q: What vibrational frequency change can be generally expected upon an isotopic exchange of vibrating…
A: Isotopic substitution refers to the replacement of one isotope of an element with another in a…
Q: You perform a concentration of a single component using displacement chromatography, and the…
A: The aim is to explain the data includes the concentration of binding sites, dissociation constants…
Q: reaction
A:
Q: the poh of 25% titration of 50ml of 0.2 m hcl with 0.1m nacl is
A: The objective of the question is to find the pOH of the solution when 25% titration of 50ml of 0.2 m…
Q: Exercice 5 Complete the following reactions, giving details of the reaction mechanisms Exercice 6…
A: The aim is to show the synthesis of carbonyl compound and cyano group attached by using kCN. It show…
Q: 10. Explain the reaction shown below. Include the following pieces in your explanation. Identify the…
A: is a strong reducing agent which is used for the reduction of carbonyl compounds, amides, cyanides,…
Q: Energy (CH3)3C-OH (CH3)3C-OH₂ + r IH + (CH3)3C+ + H₂O + F a) Write the overall balanced chemical…
A: The energy diagram represents the energy value of reactants and products on the y-axis. It is used…
Q: The van der Waals equation of state was designed (by Dutch physicist Johannes van der Waals) to…
A: a = 1.337b = 0.0320R = 0.08206 Latm/molKn = 1 as the gas is idealV = 0.384 L/mol
Q: Nitrogen monoxide reacts with hydrogen at 500 °C as in the equation below. 2NO(g) + 2H₂(g) → N₂(g) +…
A: The rate equation for a particular reaction is the mathematical expression of the reaction rate in…
Q: Br e Br 1 mol CH3CH₂NH₂ (A) aq. NaOH (B)
A: In the given reaction sequence the primary amine undergoes mono-alkylation with the alkyl bromide to…
Q: : Please draw a mechanism for the reaction shown below. In the second box please draw a potential…
A: Exothermic reactions are the reactions in which heat is released. The heat is released when stable…
Q: g) HaC OCH3 OCH₂Ph OsO4/ NalO4 acetone /ether
A: The aim of this question is to show the aldehyde formation, C=C formation and C-C coupling product.
Q: Which of the following statements correctly describe the Bom-Haber cycle? Select all that apply. 0…
A: The aim of this question is to explain the correct statement which is related to born haber cycle.…
Q: 2. When an F ion-selective electrode was immersed in unfluorinated ground water, the potential was…
A: The objective of the question is to determine the concentration of the fluoride ion in the…
Q: Rank the following five mass quantities in order from the largest to the smallest. If two of the…
A: The conversion factors we must use:1 kg = 1000 g1 g = 1000 mg
Q: Answer the following questions: 1. Name the following compounds according to IUPAC rules. A, ( C, (…
A: nomenclature rules A) Identify the longest continuous carbon chain.B) Identify the substituent…
Q: 5. Determine the Miller indices for the planes shown in the following unit cell: 6. Determine the…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: 8.142 a. Write the equilibrium equation for the acid-base reaction that takes place between aniline…
A: The objective of this question is to explain the equilibrium equation for the acid base reaction…
Step by step
Solved in 3 steps with 2 images

- Finished A-C need help with the remaining D-IIdentify the reagents needed to come up with the given compounds. Pls Separate the reagents with a comma and a space. (ex. HNO3, H2SO4). For a multi-step reaction, separate the reagents with a semi-colon. (ex. HNO3, H2SO4; CH3Cl, AlCl3) There should be 9 reagentsWhich compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaF
- For the nitration of a monosubstituted benzene, which substituent, when attached to benzene, produces the fastest reaction rate? —Br —CH3 -OCOCH3 —OCH3Draw a stepwise mechanism for the attached reaction that illustrateshow two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reactionconditions, even though it is a 1 ° alkyl halide.which c-h bond has the lowest BDE (bond disassociation energy)?
- Please help with this ochem mechanism... 1. Provide the stepwise mechanisms of the given reaction a) o,p-dinitrochlorobenzene + NaOHIn some nucleophilic substitutions under SN1 conditions, complete racemization does not occur and a small excess of one enantiomer is present. For example, treatment of optically pure 1-bromo-1-phenylpropane with water forms 1-phenylpropan-1-ol. (a) Calculate how much of each enantiomer is present using the given optical rotation data. (b) Which product predominates—the product of inversion or the product of retention of configuration? (c) Suggest an explanation for this phenomenon.Reagents 1 and 2
- ONLY USE THINGS LEARNED IN ORGANIC CHEM 1 Show stepwise mechanism by pushing arrows (Fishhook) and drawing all intermediates: Draw a detailed mechanism for the addition of HBr to 3,3-dimethyl-1-butene in the presence of dimethylperoxide. Show and label all steps including initiation, propagation and termination.Identify the correct ranking of the following molecules from most prone to nucleophilic attack at a sp^2 carbon to least. The correct answer is D, but please explain.Rearrangements can occur during the dehydration of 1° alcohols eventhough no 1° carbocation is formed—that is, a 1,2-shift occurs as the C—OH2+ bond is broken, forming a more stable 2° or 3° carbocation, asshown in Equation [1]. Using this information, draw a stepwisemechanism for the reaction shown in Equation [2]. We will see anotherexample of this type of rearrangement

