Chapter19: Aldehydes And Ketones: Nucleophilic Addition Reactions
Section19.SE: Something Extra
Problem 38MP
Related questions
Question
help with my

Transcribed Image Text:The product from a) with the molecular formula C9H10 can be
polymerized under radical conditions. Write the mechanism for the
polymerization reaction and show the polymer product structure clearly
showing the repeat unit of the structure.
Transcribed Image Text:6
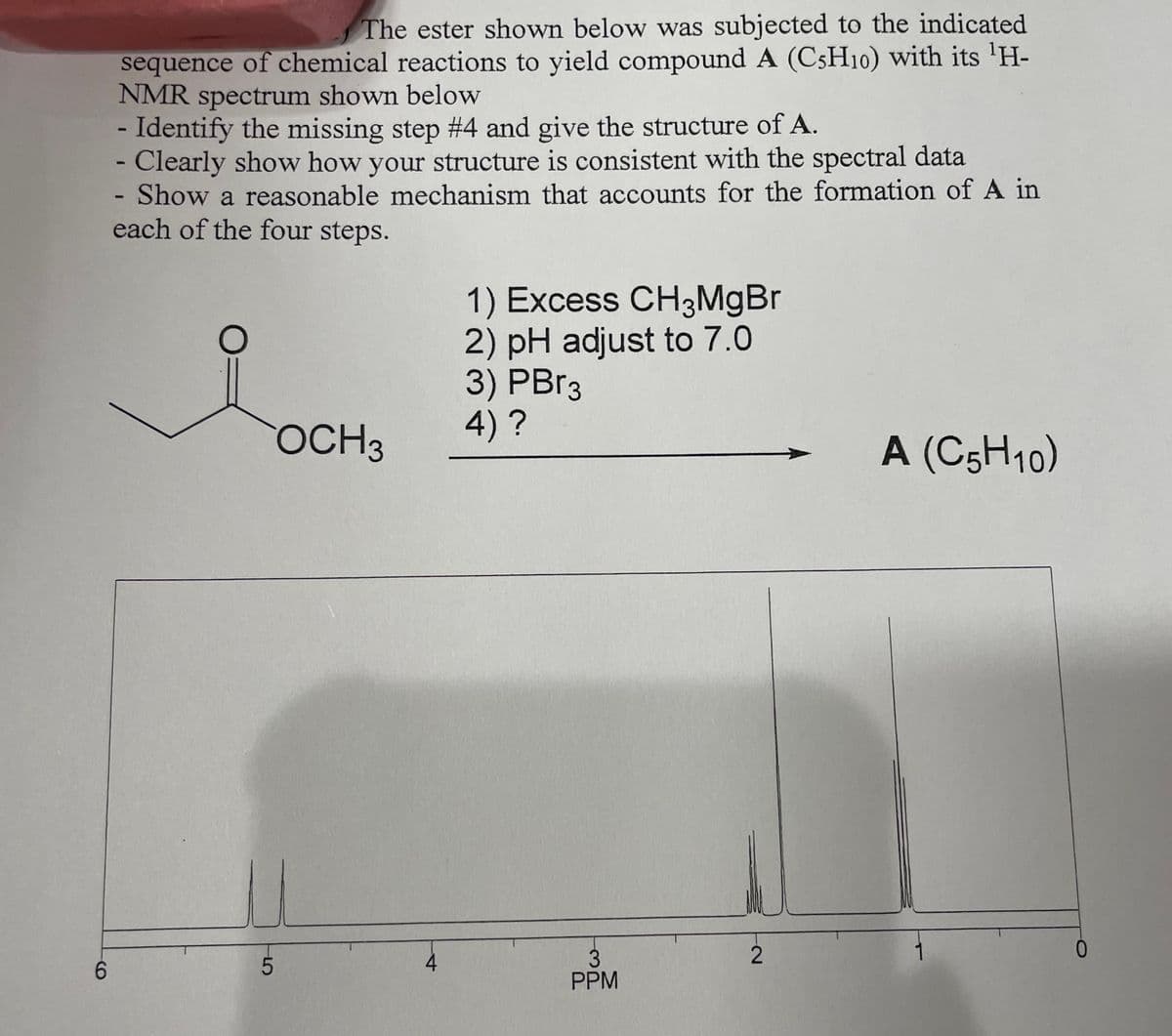
The ester shown below was subjected to the indicated
sequence of chemical reactions to yield compound A (C5H10) with its ¹H-
NMR spectrum shown below
- Identify the missing step #4 and give the structure of A.
- Clearly show how your structure is consistent with the spectral data
Show a reasonable mechanism that accounts for the formation of A in
each of the four steps.
O
CT
OCH 3
4
1) Excess CH3MgBr
2) pH adjust to 7.0
3) PBr3
4) ?
3
PPM
2
A (C5H10)
0
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

