Q: 6. Which is the best depiction of (46)-5-hydroxy-4-en-1-yn-3-one? A) OH B) OH C) D) НО OH
A: Stereochemistry is branch of chemistry in which we deal with three dimensional arrangement of atoms…
Q: 55. Stability of the major alkene product I. conc. H,SO, -ÇHCH,4 CHCH,- он он conc. H,SO, -CH,CH II.
A: Kindly get the answer given below.
Q: 8. Prepare S-2-chloropentane from the appropriate alcohol and SOC2. Show appropriate stereochemistry…
A:
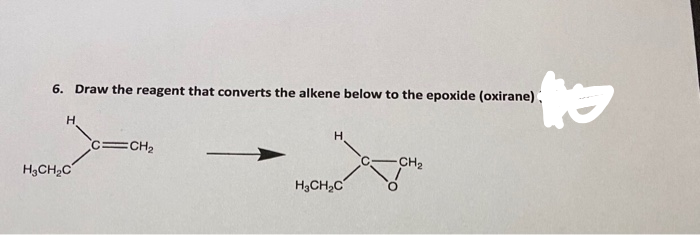
Q: 2 Give reagents that would complete the reaction to give the product with the correct…
A:
Q: 1. KMNO4, HO, Hа0, heat CH3 2. HС H,C- Product 2 ČH3
A:
Q: Which reaction would yield cyclopentanol? (A) (B) -d 8 (C) (D) 1. LiAlH4 2. H+ 1. NaBH4 2. H+ 1.…
A:
Q: A terminal alkyne (RC=CH) is exposed to excess HBr. What rule should be followed to determine the…
A:
Q: What alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict…
A: E1 reactions: These reactions proceed in two parts. Lose of leaving group. Removal of proton These…
Q: 4. (2 pts) Circle the most stable carbocation and cross out the least stable carbocation among the…
A:
Q: -CECH + 2Br, → H,C CH3
A:
Q: 4. Deduce the type of each reaction below: (a) CH2 CH3 CH3 CH,Ċ=CH2 + HBr CH2 CH3 CH3 CH2 CCH3 Br…
A:
Q: Draw the elimination products only for each reaction below. Label any alkene that would be formed…
A: Elimination reactions are of two types; E1 and E2. In In E1 reaction there will be two steps. The…
Q: CH, CH, CH3 エエ DU : I I
A:
Q: Draw the major E2 reaction product formed when cis-1-chloro-2-ethylcyclohexane (shown below) reacts…
A: The given molecule undergoes elimination reaction through E2 mechanism.
Q: Pd/C + H2 CH3 CH(CH;), +
A: Alkene undergoes reduction when they are reacted with H2 in presence of Pd/C. Here a cyclo alkene is…
Q: d. f. + Cl₂ 0 °C HI 85 °C 40 °C
A:
Q: OH b) H' a)
A:
Q: Which of the following carbocations would rearrange after reaction with HX? a. Хон b. ҳ CA ОН 'OH
A: -> When alcohol reacts with hydrogen halide Reaction proceeds through formation of carbocation…
Q: Sometimes carbocation rearrangements can change the size of a ring.Draw a stepwise, detailed…
A: The formation of carbocation is shown below.
Q: 5) Show how cross Claisen condensations could be used to prepare the following: (b) Ph-CH-C-OCH, (a)…
A:
Q: 54. Stability of the carbocation intermediate in an SN1 reaction: CH,=CHCH-OH ČH, I. II.CH2=CHCH2OH
A:
Q: 6. Draw the structure for compound A and for compound B in the following synthetic sequence. 1) Mg.…
A: In this question we have to tell the product of the reaction.
Q: Part IV. Draw the major organic product(s) for each reaction below. Indicate the stereochemistry…
A: These are the examples of Diels-Alder reactions. During this reaction endo product is formed as the…
Q: R'R?CHOH-CR*R* R'R?C=CR°R* t should orientation of each substituent in reactant to convert it into…
A:
Q: R'R°CHOH-CR'R* R'R?C=CR*R* What should orientation of each substituent in reactant to convert it…
A: The given reactant is alcohol. The reaction given above is a dehydrating reaction. Here the alcohols…
Q: Peroxides) CH3 CH3 (b) CHCH3 HI ? (a) HBr CH3CH2C=CHCHCH3
A:
Q: What will happen if the shown hydrogenation reaction is done using Lindlar's catalyst? H2 F it will…
A: Hydrogenation is the process of addition of H2 across carbon-carbon double bond/triple bond. The…
Q: Which of the following alkyl halide undergo rearrangement in S reaction ? CH 3 | C - CH, CH3 (b) (a)…
A:
Q: 4. Rank the following leaving groups from best to worst. OH2 -OH -CH3 -NH2 LG LG LG LG LG A B C D E
A: Ability to leave of a leaving group depends upon: Electronegativity of the leaving group: A high…
Q: (c) Draw a stepwise mechanism that shows how all three alcohols D, E and F are formed from the…
A:
Q: Select the reagent set that would achieve the shown transformation. Do fot CHM 2230 Fall 2020…
A:
Q: For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the…
A: Bulky bases prefer elimination whereas non-bulky bases prefer substitution reaction.
Q: Which pair of reactants is most likely to undergo an E2 reaction? O CH3SH + CH3CH₂CBr(CH3)2 at 20°C…
A: The given problem is based on the chemical properties of alkyl halides.
Q: .. Draw the MAJOR product of the reaction below. 1. NaNH2 2. CH;CH,I 3. HgSO, H,SO4, H,O HC CH
A:
Q: 3. The haloalkane below is heated in a solution of ethanol and undergoes SN1 and EiS a) Draw the…
A: Note: The above alkyl halide is heated with ethanol (weak nucleophile) results E1 products as major…
Q: 2. Which of the following additions to alkenes occur(s) specifically in a syn fashion? A)…
A: Alkenes undergo electrophilic addition reaction due to the presence of π-bonded electrons.…
Q: Please Prouide the missing Feagents, NH2 Please Prouide the missing reagents. OH Br
A:
Q: Show how each of the following compounds can be synthesized from an alkene:
A: Since you have posted multiple sub-parts, we are entitled to answer the first three only.
Q: 9. Draw the constitutional isomer formed when the following alkenes are treated with each set of…
A: Here we have to find the major constitutional isomer formed in the following alkene substrate by…
Q: How to solve ?
A: So alkyne are generally prepared by elimination of dihalides Here the E2 mechanism is followed in…
Q: 10. Assigh E or Z configuration for each alkene shown below. EN H3CH2C. `CH2NH2 H3C HO CI
A: According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority. 2) If first atom…
Q: 4. Draw the appropriate Newman projection (you can just draw the 1 that matters when you determine…
A:
Q: Draw the starting alkene that would lead to this the major product (and its enantiomer) under these…
A: Catalytic hydrogenation is a process in which treatment with H2 in the presence of catalyst like Ni,…
Q: In the elimination of a vicinal dihalide to yield an internal alkyne, two equivalents of NaNH, are…
A: The hydrogen atom bonded to triple bonded carbon atom in alkyne is acidic hydrogen and can be…
Q: 1. CH3CH,MgBr (excess) OCH3 (p) 2. HаО, H+ HCI (a) PPH3 CH;NH2 (r) H+
A:
Q: Q 2(a) Name the following alkenes and rank in order of increasing stability. CH, CH, i) i) CH,…
A: Alkenes is the class of hydrocarbons which contains a carbon-carbon double bond and it is an…
Q: 7. For each reaction, circle the correct product. Br N2OCH3 or NaOCH3 or E2 E2
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: H30* HgSO4 CH3CH2CH,C=CCH2CH2CH3 CH3CH,CH2CH2CCH2CH2CH3 This symmetrical alkyne yields one product
A: Alkynes on hydration(water addition) give carbonyl compounds.Only ethyne in this reaction produces…
Q: H, CH3C=CHCH,CH,CHCH,CH,OH Pt CH3 CH3
A: Ans Reaction is metal catalyst hydrogenation to produce alkane
Q: 5. E2 elimination of the substrate below with potassium hydroxide affords a single diastereomer of…
A: When this type of alkyl halide is treated with base then elimination reaction takes place . By…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What alkene is the major product formed from each alkyl halide in an E1 reaction?What alkene is the major product formed from attached alkyl halide in an E1reaction?The Hofmann product is favored by the E2 mechanism when 2-bromo-3-methylbutane is added: A. MeOH B. MeO:- in MeOH C. Nal in acetona D. (CH 3)3 CO:- in (CH 3),COH
- What other alkene is also formed along with Y in Sample Problem 9.3 (Attached) ? What alkenes would form from X if no carbocation rearrangement occurred?6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, PdWhat alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict the major product.
- What other alkene is also formed along with Y in Sample Problem 9.3? What alkenes would form from X if no carbocation rearrangement occurred?What is the product (A, B, C, or D) of the reaction shown in Image30? a. C b.D c. B d. AA hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.
- The 1,2‑dibromide is synthesized from an alkene starting material. Draw the alkene starting material. Clearly, show stereochemistry of the alkene.5- Draw the mechanism of the E2 Reaction with an Alkyl Halide. Show the correct directions of the arrows, and all reagents.1. What are products formed from the below reaction? 2. Draw the stereoisomers and name them. Hint: There are two different constitutional isomers and each constitutional isomer has 2nstereoisomers.

