6. Study the cycle below and answer the following 6.1 Provide a definition for the citric acid cycle 6.2 How many enzymatic reactions occur in this cycle 6.3 name THREE precursors for amino acid synthesis
6. Study the cycle below and answer the following 6.1 Provide a definition for the citric acid cycle 6.2 How many enzymatic reactions occur in this cycle 6.3 name THREE precursors for amino acid synthesis
Anatomy & Physiology
1st Edition
ISBN:9781938168130
Author:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Chapter24: Metabolism And Nutrition
Section: Chapter Questions
Problem 6RQ: The Krebs cycle converts ________ through a cycle of reactions. In the process, ATP, ________, and...
Related questions
Question
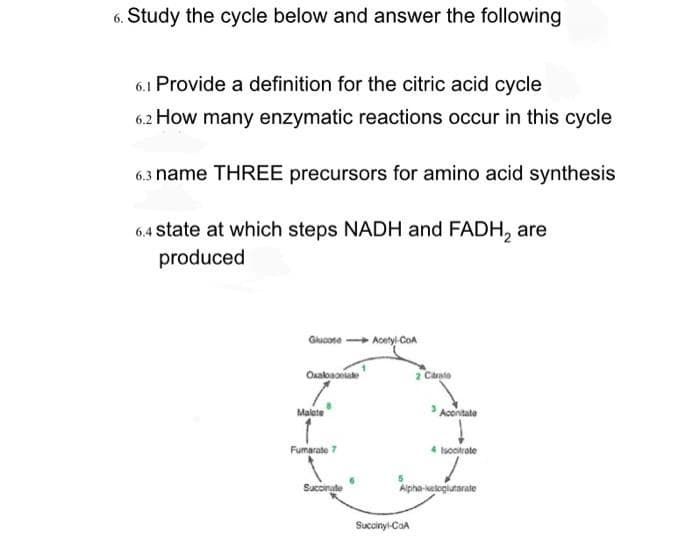
Transcribed Image Text:6. Study the cycle below and answer the following
6.1 Provide a definition for the citric acid cycle
6.2 How many enzymatic reactions occur in this cycle
6.3 name THREE precursors for amino acid synthesis
6.4 state at which steps NADH and FADH₂ are
produced
Glucose-Acetyl-CoA
Oxalbacolade
Malate
Fumarate 7
Succinate
2 Citrato
Succinyl-CoA
Aconitate
4 Isocrate
Alpha-ketoglutarate
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Recommended textbooks for you

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Anatomy & Physiology
Biology
ISBN:
9781938168130
Author:
Kelly A. Young, James A. Wise, Peter DeSaix, Dean H. Kruse, Brandon Poe, Eddie Johnson, Jody E. Johnson, Oksana Korol, J. Gordon Betts, Mark Womble
Publisher:
OpenStax College

Biology 2e
Biology
ISBN:
9781947172517
Author:
Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:
OpenStax

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning