For the tetrapeptide YIRG: a. Draw its complete protonic equilibria. Indicate the net charge of each form and encircle the zwitterionic form. b. Give the full name of the tetrapeptide. c. Calculate its IpH. d. If the tetrapeptide was subjected to electrophoresis using a buffer solution at pH 7, at which electrode it will migrate?
For the tetrapeptide YIRG: a. Draw its complete protonic equilibria. Indicate the net charge of each form and encircle the zwitterionic form. b. Give the full name of the tetrapeptide. c. Calculate its IpH. d. If the tetrapeptide was subjected to electrophoresis using a buffer solution at pH 7, at which electrode it will migrate?
Basic Clinical Laboratory Techniques 6E
6th Edition
ISBN:9781133893943
Author:ESTRIDGE
Publisher:ESTRIDGE
Chapter1: The Clinical Laboratory
Section1.7: The Metric System
Problem 4RQ
Related questions
Question
For the tetrapeptide YIRG:
a. Draw its complete protonic equilibria. Indicate the net charge of each form and encircle the zwitterionic form.
b. Give the full name of the tetrapeptide.
c. Calculate its IpH.
d. If the tetrapeptide was subjected to electrophoresis using a buffer solution at pH 7, at which electrode it will migrate?
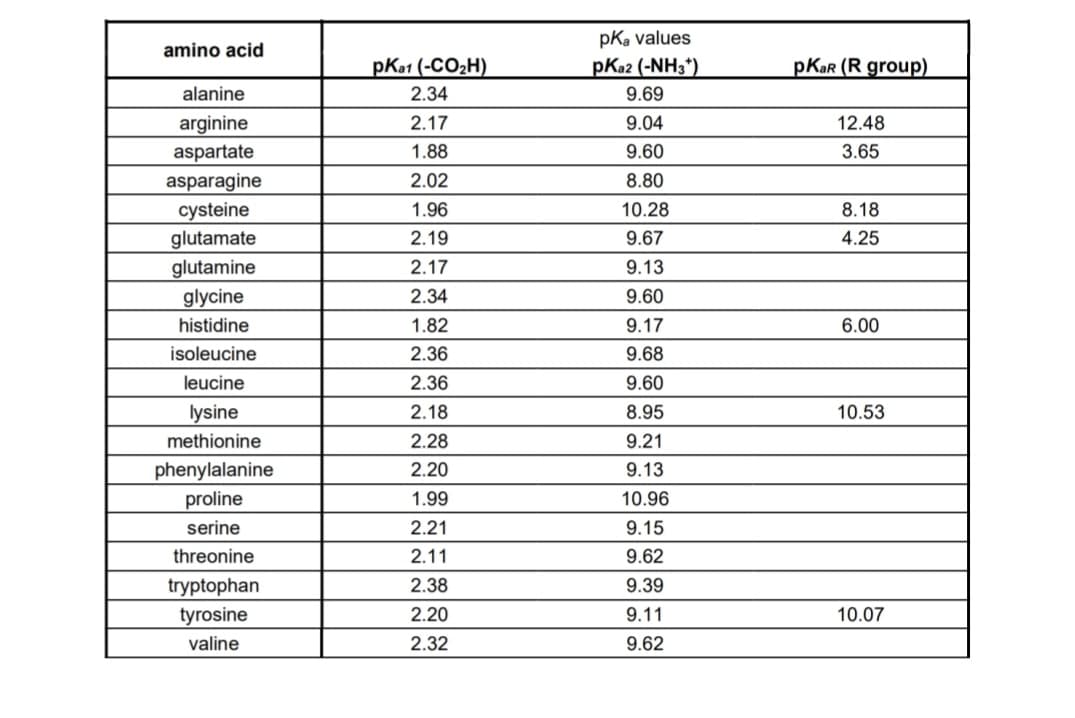
Transcribed Image Text:amino acid
alanine
arginine
aspartate
asparagine
cysteine
glutamate
glutamine
glycine
histidine
isoleucine
leucine
lysine
methionine
phenylalanine
proline
serine
threonine
tryptophan
tyrosine
valine
pka1 (-CO₂H)
2.34
2.17
1.88
2.02
1.96
2.19
2.17
2.34
1.82
2.36
2.36
2.18
2.28
2.20
1.99
2.21
2.11
2.38
2.20
2.32
pKa values
pKa2 (-NH3*)
9.69
9.04
9.60
8.80
10.28
9.67
9.13
9.60
9.17
9.68
9.60
8.95
9.21
9.13
10.96
9.15
9.62
9.39
9.11
9.62
pKar (R group)
12.48
3.65
8.18
4.25
6.00
10.53
10.07
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 5 images

Recommended textbooks for you



