6. When 60 g of calcium chloride is dissolved in 500 g of Water, greater than (a) exists as a liquid at temperatures (greater than, less than, equal to) 0°C an (greater than, less than, equal to) loss than 100°C. (b) will have a density (greater than, less than, equal to) egual to pure water will not will (c) (will, will not) conduct electricity (d) (will, will not) have a color associated with it. (e) will have a mass of g
6. When 60 g of calcium chloride is dissolved in 500 g of Water, greater than (a) exists as a liquid at temperatures (greater than, less than, equal to) 0°C an (greater than, less than, equal to) loss than 100°C. (b) will have a density (greater than, less than, equal to) egual to pure water will not will (c) (will, will not) conduct electricity (d) (will, will not) have a color associated with it. (e) will have a mass of g
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter10: Solutions
Section: Chapter Questions
Problem 45QAP: Insulin is a hormone responsible for the regulation of glucose levels in the blood. An aqueous...
Related questions
Question
I am working on my homework. Can you check my answers and help? Thanks
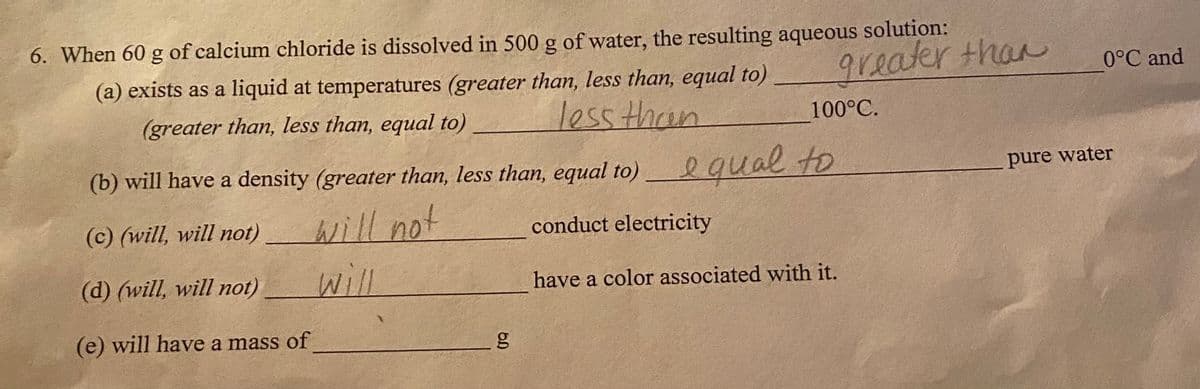
Transcribed Image Text:6. When 60 g of calcium chloride is dissolved in 500 g of water, the resulting aqueous solution:
(a) exists as a liquid at temperatures (greater than, less than, equal to)
greater than
0°C and
(greater than, less than, equal to)
lessthan
100°C.
(b) will have a density (greater than, less than, equal to) qual to
pure water
will not
will
(c) (will, will not)
conduct electricity
(d) (will, will not)
have a color associated with it.
(e) will have a mass of
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT