Q: How many distinct alkene products are possible when the alkyl iodide below undergoes E2 elimination?…
A: The E2 elimination process is where two substitutes are removed from a molecule in a one or two step…
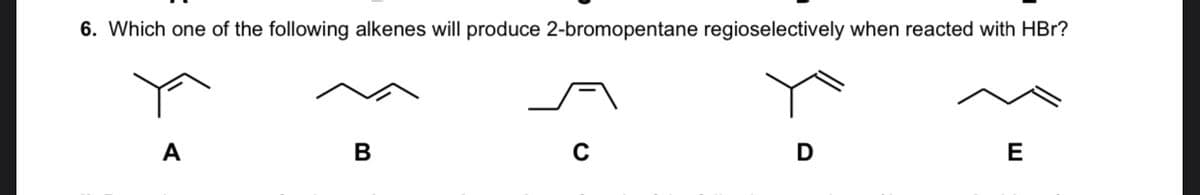
Q: Question is attached
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: 6. Rank the alkenes in order of increasing stability. (1 = least stable, 4 = most stable) |
A: Stability of alkene ∝ Number of hyper conjugative structure ∝ Number…
Q: Which alkyl halides can be prepared in good yield by radical halogenation of an alkane?
A: Alkanes undergo both bromination and chlorination. But Chlorination happens faster than bromination…
Q: 1. KMNO4, HO, Hа0, heat CH3 2. HС H,C- Product 2 ČH3
A:
Q: 6. What alkene would you start with if you wanted to prepare ethyl cyclohexane?
A: What alkene would you start with if you wanted to synthesize ethyl cyclohexane?
Q: Dehydration reactions ALWAY S form the most stable alkene as the major product. What is the major…
A: More stable alkene: Has greater substitution Bulky groups are on opposite side of double bond
Q: Which alkyl halides can be prepared in good yield by radical halogenation of an alkane?
A: Option a is a bromo substituted product and is formed from tertiary free radical. Option b is…
Q: 4. (2 pts) Circle the most stable carbocation and cross out the least stable carbocation among the…
A:
Q: 6. From what alkyl halides are the following alkenes have been made? CH3 CH3 CH3CHCH2CH2ĊHCH=CH2 7.…
A: Dear student, Bartleby platform allow us to only first question. So please repost the other question…
Q: Name the following alkene and draw the product of the reaction with KMnO4.
A: The hydrocarbons in which two of the carbon atoms are bonded to each other by a double bond are…
Q: Which of the following alkenes will yield a meso dihalide when reacted with Br/CCh at room…
A: Alkene reacts with bromine to form dibromoalkane. The reaction proceeds by the bromonium ion…
Q: 11. What is the name of the reaction in the previous question? The major organic product is the less…
A:
Q: Br a) CH3 O: b)
A: Given reactions, (a) (b)
Q: Assign E or Z to the following alkenes
A: The isomers which have a restricted rotation around the double bond is known as geometrical isomers.…
Q: What halides would undergo E2 dehydrohalogenation to give the following pure alkenes?(a) hex-1-ene…
A: Since you have posted multiple sub-parts, we will solve the first three sub-parts for you. To get…
Q: Which is more highly regioselective: reaction of an alkene with BH3 or with 9-BBN?
A: The structure of 9-BBN and BH3 are given below.
Q: 1. Hydroxylation of €-2-pentene with osmium tetroxide yields a different product than the same…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 7. Arrange each of the isomeric alkenes in order of stability: (a) (Ь)
A:
Q: Which of the following diene should give the most stable carbocation when reacts with HBr? 3 4
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Draw the four resonance forms of the allylic carbocation intermediates when the alkene below reacts…
A:
Q: What will be the major product of the reaction of 2-methyl-2-butene with each of the following…
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Arrange the following alkenes in their correct order of stability. 1=Most stable and 4=least stable.
A: Stability in alkenes is determined by the reactivity of the carbons involved in the double bond. The…
Q: Which of the following alkenes undergoes allylic bromination to form a single monobrominated…
A:
Q: What alkenes are formed from attached alkyl halide by an E2 reaction? Use the Zaitsev rule to…
A: E2 elimination is a bimolecular elimination reaction. It is a 2nd order reaction involving the…
Q: Elimination Reactions! 1. Ranking alkene stability: Please rank the following alkenes in order from…
A:
Q: 2. What Alkene would be used to synthesize 3-bromohexane? + HBr CH3CH2CHCH2CH2CH3 Br
A: You can start with 3-hexene. Since the molecule is symmetrical through its double bond, it doesn't…
Q: 2. How would you synthesize each of the following compounds? Include both the reagents and the…
A:
Q: of the following alkenes will undergo a carbocation rearrangement when reacting with H2O in the…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: In the addition reactions of alkynes, the reagents undergo transformation to form first. a. a…
A:
Q: 7. What alkene should be used to synthesize 3-bromohexane? Br ? + HBr CH,CH,CHCH,CH,CHI
A: Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Which alkyl halide(s) would give the following alkene as the only product of an elimination…
A:
Q: The elimination reaction of butan-2-ol will produce butene and but-2-ene via mechanism O a. SN1 O b.…
A:
Q: 9. А. How many different products will result if radical monobromination of the following compound…
A: Monobromination It is a free radical mechanism reaction. Monobromination of alkanes follows three…
Q: 3) Which of the following alkenes would be expected to be stable and, therefore, easily formed?
A: Compound 1 has two six member ring . In compound 1 double bond is not at bridge head carbon so this…
Q: 6. Which of the following alkenes is most reactive in the Heck reaction? in %3D IV A. I B. II C. III…
A: The given structures are represented as follows:
Q: 3. The haloalkane below is heated in a solution of ethanol and undergoes SN1 and EiS a) Draw the…
A: Note: The above alkyl halide is heated with ethanol (weak nucleophile) results E1 products as major…
Q: Which of the following additions to alkenes occur(s) specifically in an anti fashion A)…
A: Solution: Hydroboration-oxidation: in this reaction water molecule adds to alkene in anti…
Q: What alkenes are formed from attached alkyl halide by an E1 reaction? Use the Zaitsev rule to…
A: In an E1 elimination reaction, the more substituted alkene is formed as the major product.
Q: Which of these alkenes would react most slowly with HCI? CH3 (CH3),C=C CH,CH3 O CH;CHCH=CH2 CH3 ©…
A: The addition of HCl to alkene is an example of electrophilic addition reaction. The rate of this…
Q: What is the product of the most non-Zaitsev E2 elimination of the following? (i.e. Which is the…
A: (E)-3,4-dimethylpent-2-ene is the most non-zaitsev product of the reaction.
Q: 5. 2-chloro-2,3-dimethylbutane is the major product from the reaction of three different alkene…
A:
Q: Which rule is followed during this electrophilic addition to the alkene to determine the location of…
A: Addition of HBr on alkene: The reaction of alkene with HBr is a electrophilic addition reaction. In…
Q: 7. Which of the following alkenes is most stable? А. В. С. D.
A: The stability of alkenes depends on the substituents attached to it. More the number of substituents…
Q: Which of the following alkenes will yield a meso dihalide when reacted with Br2/CCI4 at room…
A: Meso compounds are those compound which have more than one stereocenters. They are superimposable.…
Q: Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with a. CH3O-.…
A: An elimination reaction is a reaction in which the atoms or groups of atoms are leaving from the…
Q: Which of the following alkenes produces the highest yield of 2,3-dimethylpentan-3-ol at the fastest…
A: Given: Following alkenes produces the highest yield of 2,3-dimethylpentan-3-ol at the fastest rate:
Q: -Br A B Br-H-
A: A carbocation is a molecule in which a carbon atom with three bonds has a positive charge.
Q: Of the two alkene products that result from this reaction, which is the expected major product? Why?…
A:
Q: Draw the elimination products that are formed when 3-bromo-3-methyl-1-butene reacts with
A: (a) The elimination product formed when 3-bromo-3-methyl-l-butene reacts with CH3O- is shown below.…
Step by step
Solved in 2 steps with 2 images

- 5. Identify the alkene which cannot be made via the Wittig reaction.9. Upon hydrogenation, which of the following alkenes releases the least heat per mole?For each reaction, decide whether substitution or elimination (or both) is possible, andpredict the products you expect. Label the major products. chlorocyclohexane + NaOC(CH3)3 in (CH3)3COH
- Which alkyl bromide(s) can form the alkene under E2 elimination conditions.What starting alkene reacted with I2 in a solution of CH2Cl2 is required to produce 1,2-diiodoocyclopentane?Q7 Consider the reaction, where the alkane shown is subjected to radical bromination at 25 °C. Br₂ light Describe the major monobromination product. product
- 10. Use Hammond's postulate to determine which alkene in each pair would be expected to form a carbocation faster in an electrophilic addition reaction. Choose which is the more reactive compound (in the box) for both pairs.Name the following alkene and draw the product of the reaction with KMnO4.Which of the following alkenes produces the highest yield of 2,3-dimethylpentan-3-ol at the fastest rate?
- Which alkene from the choices can react with excess Hbr to yield the major product shown in the first photo?Draw the four resonance forms of the allylic carbocation intermediates when the alkene below reacts with HBr as shown below.1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. Hyperconjugation

