Q: Draw the structure of (Z)-2,3-dichloro-4-methyl-2-hexene. Consider E/Z stereochemistry of alkenes. •…
A:
Q: What alkenes are formed from attached alkyl halide by an E2 reaction? Use the Zaitsev rule to…
A:
Q: What is the major organic product from the reaction of 3-hexyne with 1 mole of Br2? a. (E)…
A: The solution is shown below
Q: Draw the structure of (E)-1-chloro-3-methyl-2-pentene. Consider E/Z stereochemistry of alkenes. You…
A: Given here the IUPAC name for two molecules and we are asked to draw the structure of the…
Q: Question is attached
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: 1. Assign E or Z to the following alkenes. If the alkene does not have stereochemistry, write…
A: since you have posted a question with multiple subparts we are entitiled to answer only first 3…
Q: 6) Which of the following best describes the geometry about the carbon-carbon double bond in the…
A:
Q: Rank the following groups of alkenes from least stable (4) to most stable (1)
A: Interpretation- To tell about the stability order of alkene in above given question and then in…
Q: Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry. H2C=C=CHCH3
A: We are given an alkene. H2C=C=CHCH3
Q: 6. What alkene would you start with if you wanted to prepare ethyl cyclohexane?
A: What alkene would you start with if you wanted to synthesize ethyl cyclohexane?
Q: 1.. Assign E or Z configuration of the following alkenes; OH H- а) b.
A: E-Z designators are used as like cis-trans terminology for non-similar groups attached alkenes. In…
Q: Which of the following reactions of alkenes takes place with syn stereospecificity? addition of…
A: Answer- Addition of Br2 to alkene always give anti addition. Reaction of alkene with OsO4 give cis…
Q: Rank the following alkenes in terms of increasing stability. (A) (B) (C) OC<A<B OA< C < B OA < B < C…
A: Stability of the alkenes increases on increasing the substitution which means highly substituted…
Q: Using cis- and trans-hex-3-ene, demonstrate that the addition of HCl is not a stereospecific…
A: Since the addition of HCl led to formation of a carbocation in the RDS step. Hence the formed…
Q: 6. Which one of the following alkenes will produce 2-bromopentane regioselectively when reacted with…
A:
Q: Using cis- and trans-hex-3-ene, demonstrate that the addition of HCl is not a stereospecic reaction.…
A: Stereospecific reactions are reactions in which reactants differ only in their configuration are…
Q: Name the following alkene and draw the product of the reaction with KMnO4.
A: The hydrocarbons in which two of the carbon atoms are bonded to each other by a double bond are…
Q: Which of the following alkenes will yield a meso dihalide when reacted with Br/CCh at room…
A: Alkene reacts with bromine to form dibromoalkane. The reaction proceeds by the bromonium ion…
Q: Problem attached
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: What halides would undergo E2 dehydrohalogenation to give the following pure alkenes?(a) hex-1-ene…
A: Since you have posted multiple sub-parts, we will solve the first three sub-parts for you. To get…
Q: Now that you have learned how to name alkenes in Section 10.3, name each of the following epoxides…
A: The name of the epoxide is 1-ethylcyclohexene oxide. The name of the epoxide is 2-methylhex-2-ene…
Q: If the following reaction were carried out, how many isomeric alkene products would be possible?…
A: The elimination reaction of haloalkanes takes place either by Saytzeff's rule or by Hofmann's rule.…
Q: Name the alkene below. Use ONLY E/Z designators to indicate stereochemistry.
A: Alkenes are unsaturated hydrocarbons that have at least one carbon-carbon double bond. It is…
Q: Assign E or Z configurations to alkene below. но-
A: Alkenes are assigned E/Z configuration based on the groups attached on the carbons making the double…
Q: How do you convert the following: (i) Prop-l-ene to 1-fluoropropane (ii) Chlorobenzene to…
A: The objective of the question is to write the mechanism of the conversion reactions given in the…
Q: For the following reaction, looking down the indicated bond, provide the most stable Newman…
A:
Q: (a) CH3 (b) CH3 CH3CHCH=CHCH2CH3
A: NBS is N-bromo succinimide, It is a bromine donor. When NBS is treated with the alkenes, it react…
Q: Which of the following alkenes undergoes allylic bromination to form a single monobrominated…
A:
Q: Name (including E/Z stereochemistry) the five alkenes that can produce 3-bromo-3-methylhexane on…
A: Alkenes undergo electrophilic addition reaction with HBr to form alkyl halide. The H+(aq) formed…
Q: What product will you get when you treat trans-2-butene with HCl/H2O
A:
Q: of the following alkenes will undergo a carbocation rearrangement when reacting with H2O in the…
A: Hello. Since your question has multiple parts, we will solve the first question for you. If you want…
Q: 4. Which of the following alkenes are stable? of
A: The instability of the cyclic and bicyclic compound is due to the angle strain and the bridgehead…
Q: Which alkene gives a racemic compound upon reaction with bromine (Br2)? a. Oc. Od.
A: Alkenes are unsaturated compound which mainly gives addition reaction. Alkene mainly gives…
Q: 6. Which of the following alkenes is most reactive in the Heck reaction? in %3D IV A. I B. II C. III…
A: The given structures are represented as follows:
Q: Draw the structure of (Z)-4-methyl-2-pentene. Consider E Z stereochemistry of alkenes. You do not…
A: The compound given is (Z)-4-methyl-2-pentene.
Q: Which of the following additions to alkenes occur(s) specifically in an anti fashion A)…
A: Solution: Hydroboration-oxidation: in this reaction water molecule adds to alkene in anti…
Q: Draw the structure of (Z)-4-ethyl-3-methyl-3-octene. • Consider E/Z stereochemistry of alkenes. •…
A:
Q: How to figure out E and Z fornalkenes?
A:
Q: Identify the configuration of the alkene: (E) or (Z). Br. OZ Not a case of E vs. Zisomerism. OE
A:
Q: Which rule is followed during this electrophilic addition to the alkene to determine the location of…
A: Addition of HBr on alkene: The reaction of alkene with HBr is a electrophilic addition reaction. In…
Q: E-Z Nomenclature Assign the E-Z designation for the alkenes below „Br -OH SH 71
A:
Q: (а) CH3 (b) CH3 CH3CHCH=CHCH2CH3
A: NBS is used mainly for allylic Bromination. An unsymmetrical alkene will lead to a mixture of…
Q: Draw the products of the attached reaction, including stereochemistry.
A: During Bromination of alkenes, the π-elcetrons of the alkene attack the Br2 from downward position…
Q: is the following alkene E, Z
A: When the molecule has a double bond, the rotation of the group is restricted. This gives rise to…
Q: Assign the alkene stereochemistry as E or Z. CI OE ΟΖ
A: The IUPAC's preferred technique of defining the actual stereo-chemistry of double bonds in organic…
Q: Of the two alkene products that result from this reaction, which is the expected major product? Why?…
A:
Q: Name the alkenes below. Use ONLY E/Z designators to indicate stereochemistry.
A: The given alkene has 3 double bonds with the molecular formula of C10H13.
Assign E or Z to the following
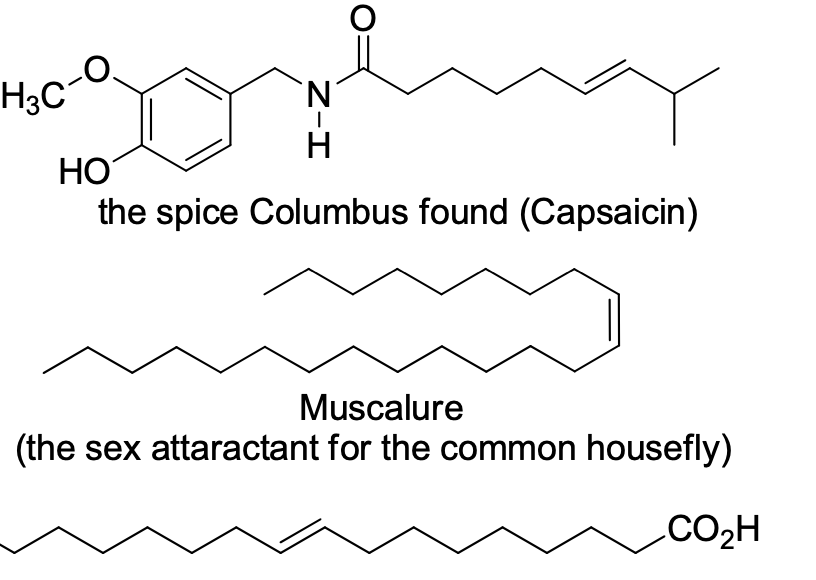
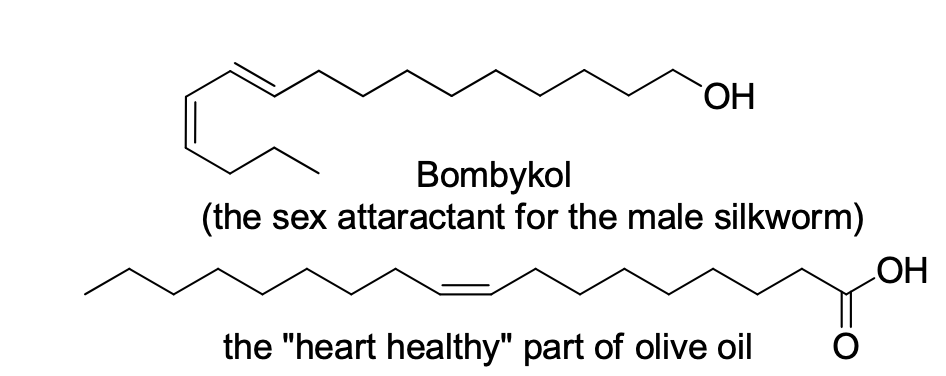
Step by step
Solved in 6 steps with 5 images

- Why are ducks waterproof? It’s because they produce copious amounts of oils from their uropygial glands and spread it across their feathers. In this exercise, we’ll be investigating the molecular structure of one of these preen oils to determine how it keeps ducks dry. Q.5 - Preen oil is actually a complicated mixture of many different organic compounds, such as the structure seen previously.. Ornithologists have determined that birds often use preen oil compounds for scent recognition. Below, several different chemicals isolated from preen oil are shown, along with their vapor pressures at room temperature. p-cymene has the highest vapor pressure, meaning it is the most easily evaporated compound of the three listed. Explain why p-cymene has a higher vapor pressure at room temperature compared to the other compounds. Make sure to explain what holds the p-cymene in the sample. (Image attached)What mass of sucrose, C12H22O11, must be dissolved per liter of water (d= 0.998 g/ml) to obtain asolution with 2.75 mole percent C12H22O11?Sulfur compounds cause "off odors" in wine, so winemakers want to know the odor threshold — the lowest concentration of a compound that the human nose can detect. The odor threshold for dimethyl sulfide (DMS) in trained wine tasters is about 2525 micrograms per liter of wine (?g/L).(�g/L). The untrained noses of consumers may be less sensitive, however. The DMS odor thresholds for 1010 untrained students are given. DMS odor thresholds 30 30 42 35 22 33 31 29 19 23 Assume that the standard deviation of the odor threshold for untrained noses is known to be ?=7 ?g/L. Give a 95% confidence interval for the mean DMS odor threshold among all students. Use the four‑step process. Give your answer as an interval in the form (lower bound, upper bound). Give your numbers to two decimal places. What is the confidence interval ?
- C6H8O6(aq) + C12H7O2NCL2(aq) ----> C6H6O6(aq) + C12H9O2NCL2(aq) - C12H7O2NCL2 is abbreviated as DCIP - Molar Lass of C6H8O6(s) is 176.14 g/mol - Daily recommended intake (DRI) for adults: 90 mg of Vitamin C Askor & Bich found that it required 23.14 mlof 1.32 x 10^-3 M DCIP soliution to reach the endpoint when titrating a 7.50ml aliquot of pear juice. How many mg of ascorbic acid, C6H8O6 are present in the juice aliquot? _5.38_____(can you please show how to calculate this answer) How many ml of this juice would Bich have to drink in order to meet the daily recommended intake (DRI)?__126__ (A. %Na2B4O7 B. %Na2B4O7• 10H20 C.%B2O3 D. %B29. asaaappp plsss

