Q: Using the template graph, redraw your graph of the distribution of velocities of Neon at 300K; label…
A:
Q: 2. You are observing a sealed flask of ethyl alcohol at 25°C. The particles in this scenario are…
A: (2) Given that - Boiling point of ethyl alcohol (ethanol) = 78°C And initial temperature is 25°C…
Q: Calculate the relative neutralizing value for a ground limestone with the following characteristics.…
A: Answer- Effective Neutralizing value (ENV)- Relative Neutralizing value-
Q: A certain second-order reaction (B→products) has a rate constant of 1.05x10-³ M-¹.s¯ an initial…
A:
Q: Seawater contains an average of 1.08 x 10^3 ppm of Na+ and 270ppm of SO4^2- . Calculate a) the molar…
A:
Q: Common Polymers 1. teflon 2. starch are made from repeating units of 3. polypropylene 7. cellulose…
A: We are given some polymers and we need to categorize these as polymers found in living things and…
Q: 9) Blast furnaces give off many unpleasant and unhealthy gases. If the total air pressure is 0.99…
A: Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Use the figure below to answer the following questions. Molecule 1 Molecule 2 Molecule 3 Molecule 4…
A:
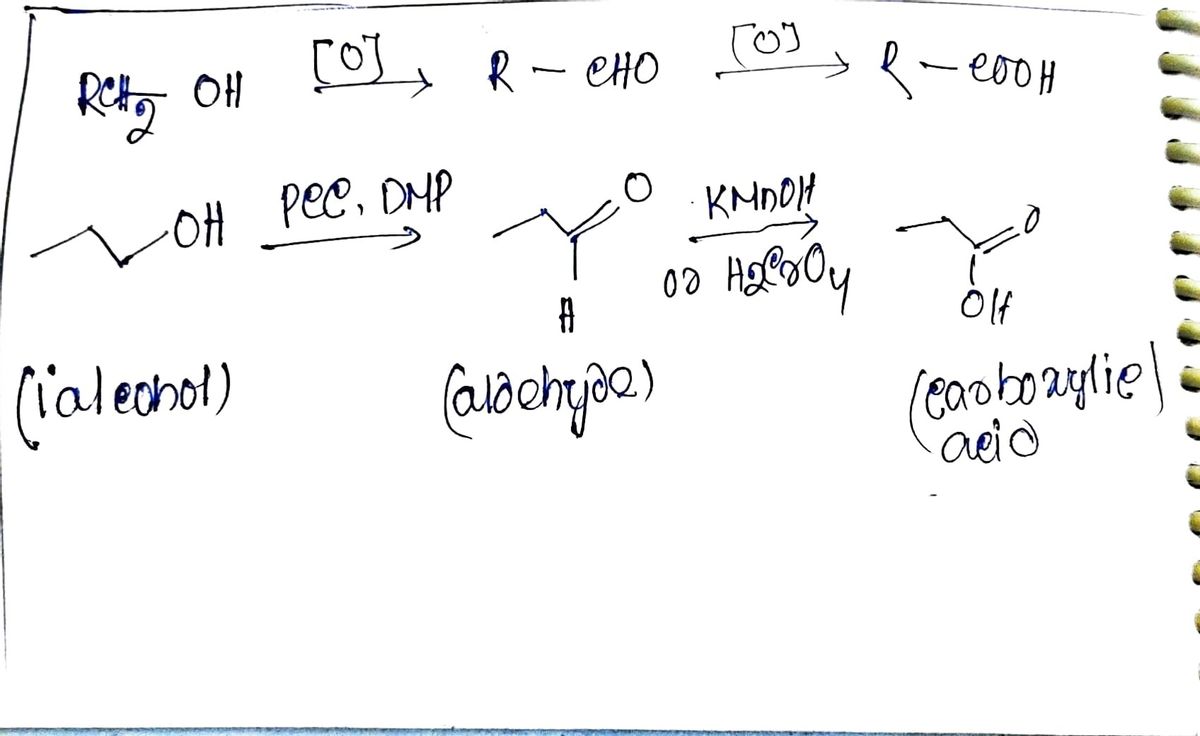
Q: Write equations and answer questions for the following: Step 1: conversion of 1-bromobutane into a…
A:
Q: your answer... choose your answer..... choose your answer... CH3COO CO3 NO2 OH MnO4 PO3 CN choose…
A: 1.Cation are the species which carry posative charge. 2. Anion are the species which carry negative…
Q: Deq xood 90 Textbook pages that helped with this content Element Symbol P S CI 지끅. Ca bo Atomic…
A: For an element the atomic number, number of protons and number of electrons are same. i.e.…
Q: The activation energy for a particular reaction is 102 kJ/mol. If the rate constant is 1.35 x 104 s¹…
A:
Q: According to the following reaction: 2Na (s) + 2H₂0 (1) →→→→ 2NaOH (aq) + H₂ (g) What would you…
A: The mole ratios can be used as conversion factor to convert between amount of any two substances…
Q: Item Mass of empty weigh boat Mass of mixture and weigh boat Total Mass of sample of Iron, Salt, and…
A:
Q: Calculate total vapour pressure above an ideal liquid mixture for which the vapour composition of…
A: •Here given question regarding vapour pressure of solution in which two components present A and B.…
Q: A sample of the compound M2SO4 weighing 0.1561 grams reacts with barium chloride and yields 0.3193…
A:
Q: C6H14NO₂ Cq Hqs Cl 17. What is the molecular geometry and bond angles around the indicated atom in…
A: Given : structure of molecules
Q: Classify these salts as acidic, basic, or neutral. Acidic NaF Basic NH4CIO4 K₂CO3 Neutral KCI LiBr
A: Salts are ionic compounds composed of positively charged cations and negatively charged anions. They…
Q: Name another method that could be used to determine the enantiomeric excess of your resolved amine.
A: Enantiomeric excess (ee) is a measure of the relative amount of one enantiomer present in a mixture…
Q: 17. What is the IUPAC name of the following compounds? 0.3
A: we have to determine the IUPAC name of the given compounds
Q: 6:53 PM Sun Feb 5 Question 11 of 50 What is the vapor pressure of a liquid (in mmHg) at 309.07 K if…
A:
Q: ChemDraw Practice 3. Double click on each of the reactions - because these were made in ChemDraw and…
A: The incomplete reactions given are
Q: Study the following phase diagram of Substance X. pressure (atm) 36 I 18 0 0 solid 200 liquid gas…
A: •Given phase diagram=>
Q: Symbol M 55 Cr³+ A24 ¹40K 19 Zn²+ 3652- Atomic Mass Number Number 24 19 55 40 35 81 4 9 Cu²+ mad…
A:
Q: Question 8 When the following reaction takes place inside a balloon, what will happen to the volume…
A: Based on the reaction we can decide what will happen to baloon.
Q: The half-life for the zero order reaction A → Products is 276 s. What is the value of the rate…
A: Given half life of zero order reaction(t1/2/ = 276 s Initial concentrate (a)= 0.557…
Q: Consider the Macmi MgCl₂(aq) + 2NaOH(aq) Mg(OH)₂ (s) + 2 NaCl(aq) A 138.0 mL solution of 0.123 M…
A: Answer: Limiting reagent is the reactant that exhaust first in the chemical reaction and decide the…
Q: a double replacement reaction between Zinc (ll) chloride and sodium hydroxide: ZnCl2+NaOH ==>…
A: •Given reaction in between:- Zinc(II) Chloride and sodium hydroxide. => ZnCl2 + NaOH ==>…
Q: For each of the following acid-base reactions indicate whether it will favor reactants or products.…
A: Answer: In the given reaction conjugate acid and base formation is taking place and we have to find…
Q: Part 1 On the basis of the frequency factors and activation energy values above, calculate the rate…
A:
Q: needed a hand drawn graph and steps in how you predicted the mass of the 30 marbles from graph.
A: To draw the given data on a graph. The data given is as follows: Number of marbles Mass of…
Q: Part A A certain first-order reaction (A-products) has a rate constant of 3.60x10-3s¹ at 45 °C. How…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1) b b CN The carbon with the smallest delta value (the most shielded) is The carbon with the…
A:
Q: Consider a transition of electron in the hydrogen atom from n=4 to n=7 Determine the wavelength…
A:
Q: Draw the organic product of the Bronsted acid-base reaction. Include all lone pairs and charges as…
A:
Q: Arrange the following in order of increasing AHfus- (1 being the lowest and 4 being the highest)…
A: Answer :
Q: Relate the energies of the orbitals found (these we will use for building Molecular Orbital energy…
A: Principle of Slater's Rule - actual charge felt by an electron is equal to the charge exerted by a…
Q: c) Sketch a complete labelled crystal field splitting diagram for complex ion with 4 cyanide ligands…
A:
Q: Estimate the solubility of KNO3 at 100°C. Show/explain your reasoning. 2 Circle the insoluble salt…
A: (1) Solubility of KNO3 is 169 gm per 100 gm of water at 100°c This is based on the solubility curve.…
Q: The data below were collected for the following reaction: CH3C1(g) + 3Cl2(g) → CCl4 (g) + 3HCl(g)…
A: Answer: Power of concentration term of a reactant in the rate law is the order of reaction with…
Q: Which of the following is NOT a postulate of the Kinetic Molecular Theory? O Gases have tiny volumes…
A: According to kinetic molecular theory, there are 5 main postulates: 1. Gas particles are…
Q: Current Attempt in Progress For a globular protein that is found in the cytosol, would most likely…
A: In the interior of proteins the environment is hydrophobic. So only non-polar amino acids are found…
Q: Pennies currently in circulation are composed of 97.5 % zinc and 2.5 % copper (by mass). What is the…
A: We know that morality = moles of the solute / kilograms of solvent as here copper is in less…
Q: A typical barometric pressure in Kansas City is 747 torr. What is this pressure in atmospheres, in…
A:
Q: 1H 3H 3 PPM 3H CSH 1002 3H
A: To solve this problem (1)find out site of unsaturation Then arrange data as guven in spectrun and…
Q: 15. Write the bondline formula for the following: a) (CH3)3CCH(OH)CH₂C(CH3)=CHCHCICH3 c)…
A: The given molecules are
Q: Click on the site of fastest protonation by HBr. H3C-CH=CH-CH=CH₂
A:
Q: To calculate the half-life, plug the value for k into the half-life equation and solve. What is the…
A: Given : values of rate constant
Q: AucpH of solution AFTER reaction howera Sett nobes19 hsda cl vb89197 Calculate the mass of HC2H302…
A: (a) Given that - Volume percentage of vinegar solution = 5% It means this solution contains 5 mL…
Q: 3. Read the following passage and then solve the problems. In the equation that follows each…
A:

Step by step
Solved in 2 steps with 1 images


