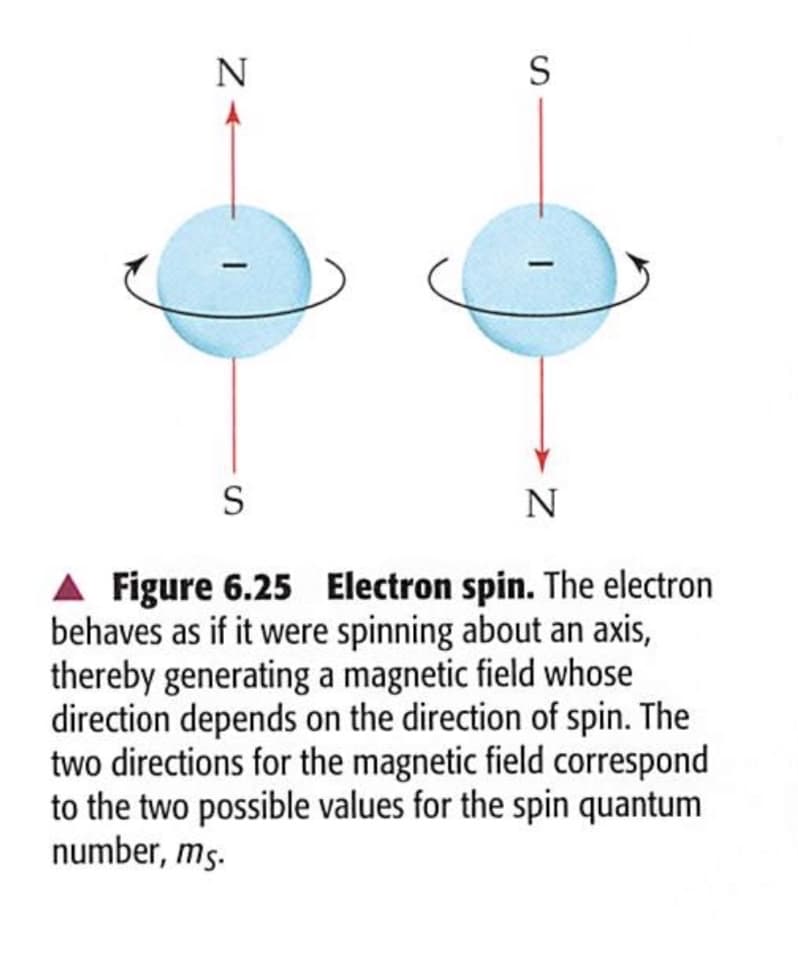
[6.94] As noted in Figure 6.25, the spin of an electron gener- ates a magnetic field, with spin-up and spin-down elec- trons having opposite fields. In the absence of a magnetic field, a spin-up and a spin-down electron have the same energy. (a) Why do you think that the use of a magnet was important in the discovery of elec- tron spin (see the "A Closer Look" box in Section 6.7)? (b) Imagine that the two spinning electrons in Figure 6.25 were placed between the poles of a horseshoe mag- net, with the north pole of the magnet at the top of the figure. Based on what you know about magnets, would you expect the left or right electron in the figure to have the lower energy? (c) A phenomenon called electron spin resonance (ESR) is closely related to nuclear magnetic resonance. In ESR a compound with an unpaired elec- tron is placed in a magnetic field, which causes the un- paired electron to have two different energy states analogous to Figure 6.27. ESR uses microwave radia- tion to excite the unpaired electron from one state to the other. Based on your reading of the "Chemistry and Life" box in Section 6.7, does an ESR experiment re- quire photons of greater or lesser energy than an NMR experiment?
[6.94] As noted in Figure 6.25, the spin of an electron gener- ates a magnetic field, with spin-up and spin-down elec- trons having opposite fields. In the absence of a magnetic field, a spin-up and a spin-down electron have the same energy. (a) Why do you think that the use of a magnet was important in the discovery of elec- tron spin (see the "A Closer Look" box in Section 6.7)? (b) Imagine that the two spinning electrons in Figure 6.25 were placed between the poles of a horseshoe mag- net, with the north pole of the magnet at the top of the figure. Based on what you know about magnets, would you expect the left or right electron in the figure to have the lower energy? (c) A phenomenon called electron spin resonance (ESR) is closely related to nuclear magnetic resonance. In ESR a compound with an unpaired elec- tron is placed in a magnetic field, which causes the un- paired electron to have two different energy states analogous to Figure 6.27. ESR uses microwave radia- tion to excite the unpaired electron from one state to the other. Based on your reading of the "Chemistry and Life" box in Section 6.7, does an ESR experiment re- quire photons of greater or lesser energy than an NMR experiment?
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter1: Chemical Foundations
Section: Chapter Questions
Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and...
Related questions
Question
100%
![[6.94] As noted in Figure 6.25, the spin of an electron gener-
ates a magnetic field, with spin-up and spin-down elec-
trons having opposite fields. In the absence of a
magnetic field, a spin-up and a spin-down electron
have the same energy. (a) Why do you think that the
use of a magnet was important in the discovery of elec-
tron spin (see the "A Closer Look" box in Section 6.7)?
(b) Imagine that the two spinning electrons in Figure
6.25 were placed between the poles of a horseshoe mag-
net, with the north pole of the magnet at the top of the
figure. Based on what you know about magnets, would
you expect the left or right electron in the figure to have
the lower energy? (c) A phenomenon called electron spin
resonance (ESR) is closely related to nuclear magnetic
resonance. In ESR a compound with an unpaired elec-
tron is placed in a magnetic field, which causes the un-
paired electron to have two different energy states
analogous to Figure 6.27. ESR uses microwave radia-
tion to excite the unpaired electron from one state to the
other. Based on your reading of the "Chemistry and
Life" box in Section 6.7, does an ESR experiment re-
quire photons of greater or lesser energy than an NMR
experiment?](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Fdcd1609e-2385-4a24-88bc-c1528e231024%2F80399004-0983-4de5-9a53-b129dc569b0b%2F2d4oxkd_processed.jpeg&w=3840&q=75)
Transcribed Image Text:[6.94] As noted in Figure 6.25, the spin of an electron gener-
ates a magnetic field, with spin-up and spin-down elec-
trons having opposite fields. In the absence of a
magnetic field, a spin-up and a spin-down electron
have the same energy. (a) Why do you think that the
use of a magnet was important in the discovery of elec-
tron spin (see the "A Closer Look" box in Section 6.7)?
(b) Imagine that the two spinning electrons in Figure
6.25 were placed between the poles of a horseshoe mag-
net, with the north pole of the magnet at the top of the
figure. Based on what you know about magnets, would
you expect the left or right electron in the figure to have
the lower energy? (c) A phenomenon called electron spin
resonance (ESR) is closely related to nuclear magnetic
resonance. In ESR a compound with an unpaired elec-
tron is placed in a magnetic field, which causes the un-
paired electron to have two different energy states
analogous to Figure 6.27. ESR uses microwave radia-
tion to excite the unpaired electron from one state to the
other. Based on your reading of the "Chemistry and
Life" box in Section 6.7, does an ESR experiment re-
quire photons of greater or lesser energy than an NMR
experiment?
Transcribed Image Text:N
-
S
N
A Figure 6.25 Electron spin. The electron
behaves as if it were spinning about an axis,
thereby generating a magnetic field whose
direction depends on the direction of spin. The
two directions for the magnetic field correspond
to the two possible values for the spin quantum
number, ms.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781259911156
Author:
Raymond Chang Dr., Jason Overby Professor
Publisher:
McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9780078021558
Author:
Janice Gorzynski Smith Dr.
Publisher:
McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Elementary Principles of Chemical Processes, Bind…
Chemistry
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY