Q: Q2-What is the number of grams of Cas(PO4)2 and grams of KCI can be produced by mixing 5g of CaCl;…
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: N2 + 3H2 2NH3 -> How many liters of NH3 are formed from 9.00 g of H2
A: The number of moles of the substance is provided as the ratio of the mass(m) and its molar mass…
Q: a sample of ammonia (NH3) Liberates 5.59 KJOF heat as it solidifies at its meining point Whar is the…
A: Given : Amount of heat liberated = 5.59 KJ And the heat of fusion of ammonia = 5.66 KJ/mol.
Q: If 2.88 moles of AlPO4 were needed for a reaction what mass of AlPO4 would need to be measured out
A: The moles of AlPO4 is = 2.88 moles The mass of AlPO4 in grams is =?
Q: Ton.) 83: The combustion of gasoline produces carbon dioxide and water. Assume gasoline to be pure…
A:
Q: e ethyl alcohol(Mw=46.06 g/mol) content of many juice produced in the United States is 4.05%…
A: The given data: The (v/v%) of ethyl alcohol = 4.05% The density of ethyl alcohol = 0.7893 g/mL To…
Q: 5.00 g of copper is reacted with 5.00 g of sulfur in a crucible and acompound composed of copper and…
A: The question is based on the concepts of reaction Stoichiometry. we know the amount of product…
Q: Given the data: 1. Mass of test tube + MNO² = 21.5403g 21.5403g 2. Mass of…
A: Given : Mass of test tube + MNO2 = 21.5403 g And mass of the test tube + MnO2 + KClO3 + KCl =…
Q: Magnesium at start is 0.33g mass at end is 0.49 g Determine the number of moles of magnesium…
A: The no of moles can be calculated by the following formula. no of moles=massmolar mass Molar mass of…
Q: A sealed chamber contains 7.50 g CH4 and 14.5 g O2. The mixture is ignited producing CO2 and H2O.…
A: According to the balanced chemical reaction shown as follows:
Q: Solid lithium hydroxide is used to remove exhaled carbon dioxide from space vehicles by forming…
A: Lithium hydroxide reacts with carbon dioxide to form lithium carbonate and water. The equation for…
Q: Magnesitim will burn in air to form both Mg3N2 and Mgo. Vwhat mass oI each product would Ioune 3.42…
A:
Q: See picture below
A: The balanced reaction is given below.
Q: 500g of Sodium(Na) reacts with 750g of Calcium Chloride (CaCl½) to form Sodium Chloride (NaCI) and…
A:
Q: In the preparation of brass, which is a copper-zinc alloy,19.0g copper is added to enoigh zinc to…
A:
Q: Mg + 2HCl ---> MgCl2 + H2 If 24.31g of Mg react completely with 72.92g of HCl, according to the…
A: Since in any reaction, the mass of reactants reacted will always be equal to the mass of products…
Q: An organic liquid is either methyl alcoholethyl alcohol or a mixture of the two.A 0.220 g sample of…
A: The combustion reaction of methyl alcohol and ethyl alcohol:
Q: 102.0g of tricarbon Octahydrogren is completely burned. How much carbon Dioxide is produced?
A: C3H8 is tricarbon Octahydrogen.
Q: 1.08 g Epsomsalt (or hydrated magnesium sulfate)is heated,and 0.57g of dried Epsom…
A: Given: 1.08 g Epsomsalt (or hydrated magnesium sulfate)is heated and 0.57g of dried Epsom…
Q: The ethyl alcohol(Mw=46.06 g/mol) content of many juice produced in the United States is 3.07%…
A: Given: The concentration of the ethyl alcohol in juice = 3.07% (vol/vol) The density of ethyl…
Q: ny moles of HI will be formed?
A:
Q: What is the percent by weight (w/w%) of sugar in soda? Assume the average mass of sugar in soda is…
A: % by weight of sugar in soda=weight of sugarTotal massx100 given weight of sugar=35 g , Total…
Q: 500g of Sodium(Na) reacts with 750g of Calcium Chloride (CaCl2) to form Sodium Chloride (NaCI) and…
A: Given:mass of Na= 500gmass of CaCl2 = 750galso, we knowmolar mass of Na =22.98gmolar mass of CaCl2…
Q: A biogas-plant converts biomass into methane. From a certain substrate, 52% (w/w) methane can be…
A:
Q: Elemental sulfur occurs as octatomic molecules, S₈. Whatmass of fluorine gas is needed to react…
A: To determine the mass of the fluorine gas required to react completely with 17.8 g of sulfur. The…
Q: Part B: Percentage of Baking Soda in an Unknown Mixture UNKNOWN # Mass of test tube and beaker…
A: Given that - Mass of unknown mixture used = 1.40 g Mass of H2CO3 released = 0.51 g From the…
Q: what is the number of moles of calcium carbonate reacted?
A:
Q: Aqueous hydrochloride a if (HCl) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A: The number of moles for a given compound or a molecule refers to the ratio of the mass to that of…
Q: Conversions from moles to molecules involve how many steps? A. 1-step because 1 mole = 6.02X10^23…
A:
Q: Solid lithium hydroxide is used in space vehicles to remove exhaled carbon dioxide from the living…
A: Mass of LiOH = 1kg
Q: 3.230g sample of a compound containing on the carbon hydrogen and oxygen is burning an excess…
A: Given: Mass of sample = 3.230 g Mass of CO2 = 7.343 g Mass of H2O= 3.005 g To Calculate: Mass of…
Q: If 56.0 g of O2 is mixed with 56.0 g of H2 and the mixture is ignited, A. what is the maximum mass…
A: Reaction: 2H2 + O2 →2H2O 2moles of H2 reacts with 1 mole of O2 to give 2 moles of H2O
Q: Use the moles of carbonate ion, CO,²-, to determine the moles of MgCO, and CaCO, present in the…
A: Part 1 : a.) As in limestone there are both CaCO3 and MgCO3 . So CO32- will come from both CaCO3…
Q: 48.1g of aluminum hydroxide to molecules of aluminum hydroxide
A: Consider the given information is as follows; Mass of aluminum hydroxide = 48.1 g…
Q: Compute for the mass in grams of NaCl produced by reacting 1.50 g of NHẠCI with 3.0 g NaOH. NH4 +…
A:
Q: the reaction C2H4 + 3O2 ---> 2CO2 + 2H2O, does this mean that ten (10) grams of CO2 produced reuired…
A: C2H4 + 3O2 ----> 2CO2 + 2H2O Mass of CO2 = 10 g Number of moles of CO2= 10g ÷ 44g/mol = 0.227…
Q: Consider the following unbalanced chemical reaction: Br2 + F2 → BrF5 How many grams of fluorine…
A: the balanced equation for given reaction is Br2 + 5F2 → 2BrF5 here 1 mole of Br2 needs 5 moles of F2…
Q: During the analysis, 0.163 g H,0 and 0.600 g CO2 are produced. Calcuate the amount (mol) C in the…
A: The combustion Reaction of the hydrocarbon Hydrocarbon + O2 → H2O + CO2 By this combustion…
Q: Methane is burned completely with 25% excess air. Compute the number of moles of nitrogen in the…
A:
Q: ,how many moles of dichloromethane (CH,Cl) will be formed
A: Number of moles = mass/molar mass Molar mass of CH4 = 16.0425 g/mol
Q: Mass Salicylic Acid used 0.1498 g Mass Acetic Anhydride used 0.3417 g Mass filter…
A: Theoretical yield of aspirin can be obtained by determining the limiting reagent of the reaction.
Q: Solid lithium hydroxide is used to remove exhaled carbon dioxide from space vehicles by forming…
A:
Q: he information below Ws some volumes of
A: Hydration means the substance combines with water. Cement undergoes hydration and forms hardened…
Q: A 36.7g sample of HCl is reacted with 27.5 grams of 02. Calculate the grams of CL2 produced
A: The balanced reaction is given as: Here, the limiting reagent would be that, which less in…
Q: What volume of 0, gas is produced if _Sr(CIO3)2>_SrCI, + _02
A: Strontium chloride (SrCl2) is a salt of strontium and chloride. Like all compounds of Sr, this also…
Q: In one sentence explain what's wrong with this equation, and rewrite it so that the coefficients are…
A: Answer:- C3H6(g) + 4.5 O2(g) -------> 3CO2(g) + 3H2O(g) In this equation the…
Q: Octane gasoline C2 H5OH, is burned with theoretical air. Calculate a. A/F ratio on mole basis b. A/F…
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Given that 4 NH3 + 5 O2 ----> 4 NO + 6 H2O, when 4.50 mol of H2O are formed, the amount of NO formed…
A:
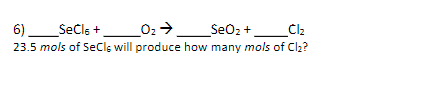
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Evaluate the molar ratio of the below reaction: Xe+F2 ----> XeF2How to manipulate formula mgh=1/2mv2 to find velocity and massand Employees CH STATE GE 90 ENDULERSE IS FOREVER "ON-Tame Name Jodi Rayal Molarity = wt. x1000 3. Calculate the massof Potassium hydrogen phthalate (KHP) to prepare a 250.0 mL solution of 0.1000 M KHP solution. Mol ut. x Voluse the 2500×1000 204.22250 mass Initial Buret Reading Final Buret Reading Vol NaOH Added Moles of NaOH Moles of HCI Volume of unknown HCI solution Concentration of HCI Average concentration of HCI of KHP = 5.11g 4. Complete the table below for the following neutralization reaction of an unknown concentration of hydrochloric acid with 0.100 M sodium hydroxide: Mu spate solution Trial 1 0 11.15 го data and calculate the concentration 15 Trial 2 11.15 22.15 Trial 3 22.15 33.25 abyh 067 LANDINUKASHU TROWTHCENTISSATE ARAD Ma That par މއކނި REMURNAR MISAMARALISERDRY Tile
- Masses : Ca = 40.08 g S= 32.07 g O= 16.00 g A 35.0 mL sample of CaSO4 was evaporated to dryness, leaving 0.967 g of residue. What was the molarity of the original solution? step by step pleaseWhy do you multipy the # of H2S mols by 4, when 4 corresponds to the reactory amount of O2?The logic there is somewhat confusing to me.I understand how/why you found out the mols of the equation but dividing the sample's MM by the equation confuses me.50.00 cm3 of a 1.5784 mol.dm-3 solution of potassium hydroxide is transferred to an empty 700.00 cm3 volumetric flask. This flask is made up to the mark with distilled water and then shaken well. The concentration of the potassium hydroxide in this second flask is:
- Recall that the formula for ΔHrxn = [(-Ccal)(moles of limiting reactant)/(ΔT)]If the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value, will the value of Ccal increase, decrease, or remain the same? Please explain. How about if the actual concentration of the sodium hydroxide used in the calibration part of the experiment was lower than the stated value. However, the NaOH solution used in the determination of ΔHrxn was of the correct concentration. Will the magnitue of ΔHrxn increase, decrease, or remain the same? Please explain.To prepare a very dilute solution of K2Cr2O7 (294.18 g/mol), 0.661 g of K2Cr2O7 was transferred to a 250.0-mL volumetric flask and adding water to the mark. A sample of this solution of volume 1.000 mL was then transferred to a 500.0-mL volumetric flask and diluted to the mark with water. Subsequently, 10.0 mL of the diluted solution was transferred to a 250.0-mL flask and diluted to the mark with water. What is the final concentration of K2Cr2O7 in this last solution?What is the concentration of a 62.0 % (w/w) concentrated nitric acid (HNO3, Mr = 63.0 g/mol) solution (density: 1.32 g/cm3) in mol/dm3 (molarity) and g/dm3 (mass concentraion) units? How many cm3 do you need to dilute into 200.0 cm3 from that concentrated nitric acid if you would like to have a solution with a concentration of 3.0 mol/dm3 ?
- How many milimoles of Ca(NO3)2 contains 4.78 x10^22 formula units of Ca(NO3)2?what is the correct sig figs?Molarity: 0.148 M Na2S2O3 Data for Unknown Sample Trial 1 Trial 2 Mass of Sample 0.081 g 0.083 g Volume of Na2S2O3 ----------------- ------------------- Final Buret Reading 0.78 mL 0.84 mL Initial Buret Reading 0.07 mL 0.10 mL Net Volume of Na2S2O3 _____mL ______mL Calculation of Percentage NaOCl -------------- ------------- Moles of Na2S2O3 used _______mol _______mol Moles of NaOCl reacted _______mol _______mol Mass of NaOCl in sample _______g _______g Percentage by weight NaOCl (g NaOCl/g sample) X 100% _______% _______%

