7. Calculate the enthalpy change following the combustion of 1 mol of acetone using the bond energies of competent atoms: C3H60 (g) + 4O2 (g) ЗСО (g) + 3H2О (g) 0=0 O=C=O H H- Acetone Охуgen Carbon dioxide Water Bond Bond energy C-C 347 kJ/mol С-Н 413 kJ/mol 745 kJ/mol (799 kJ/mol in CO2) 498 kJ/mol C=O O=0 O-H 467 kJ/mol A. -1687 kJ/mol B. 1687 kJ/mol C. -2926 kJ/mol D. 2926 kJ/mol E. -5909 kJ/mol
7. Calculate the enthalpy change following the combustion of 1 mol of acetone using the bond energies of competent atoms: C3H60 (g) + 4O2 (g) ЗСО (g) + 3H2О (g) 0=0 O=C=O H H- Acetone Охуgen Carbon dioxide Water Bond Bond energy C-C 347 kJ/mol С-Н 413 kJ/mol 745 kJ/mol (799 kJ/mol in CO2) 498 kJ/mol C=O O=0 O-H 467 kJ/mol A. -1687 kJ/mol B. 1687 kJ/mol C. -2926 kJ/mol D. 2926 kJ/mol E. -5909 kJ/mol
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter6: Thermochemistry
Section: Chapter Questions
Problem 76E: Combustion reactions involve reacting a substance with oxygen. When compounds containing carbon and...
Related questions
Question
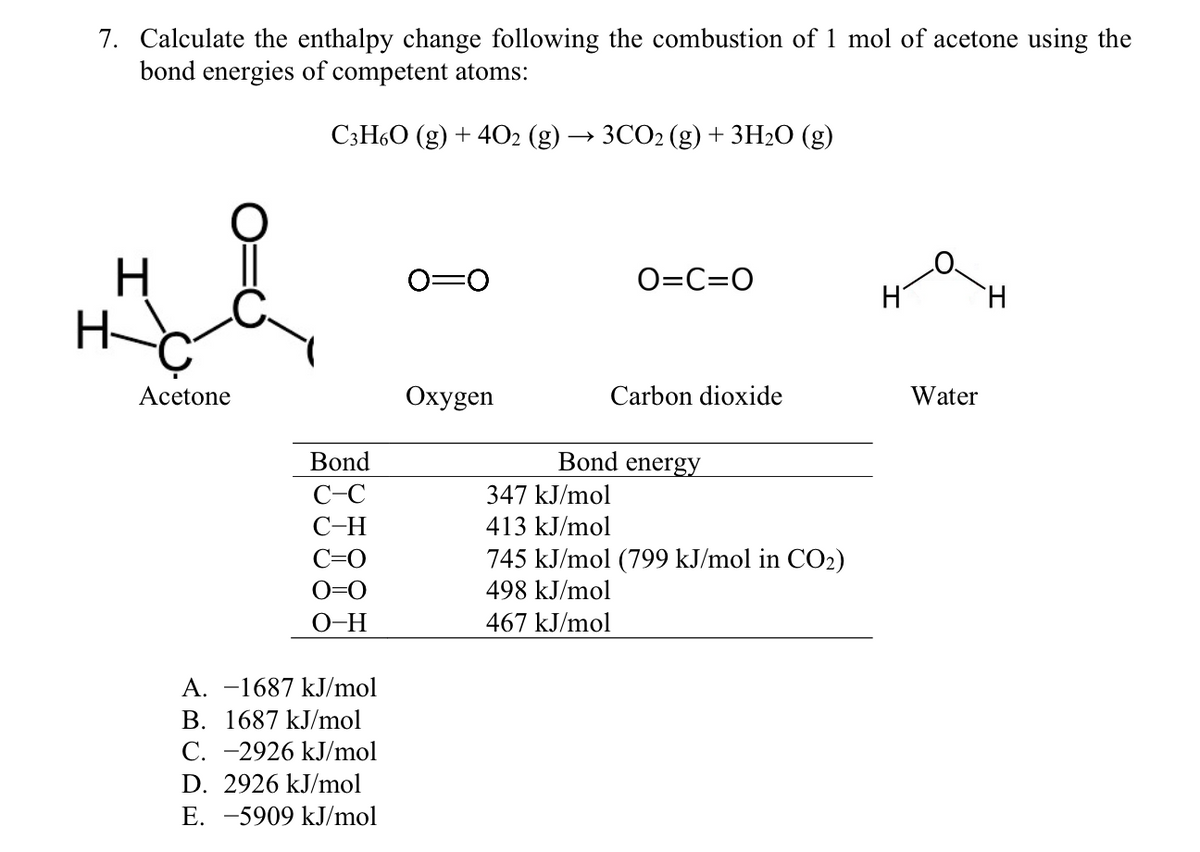
Transcribed Image Text:7. Calculate the enthalpy change following the combustion of 1 mol of acetone using the
bond energies of competent atoms:
СзН6О (g) + 4О2 (g) —> 3СО2 (g) + 3H20 (g)
H
0=0
O=C=0
Acetone
Охудen
Carbon dioxide
Water
Bond
Bond energy
C-C
347 kJ/mol
С-Н
413 kJ/mol
C=0
745 kJ/mol (799 kJ/mol in CO2)
O=0
498 kJ/mol
O-H
467 kJ/mol
A. -1687 kJ/mol
B. 1687 kJ/mol
C. -2926 kJ/mol
D. 2926 kJ/mol
E. -5909 kJ/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT