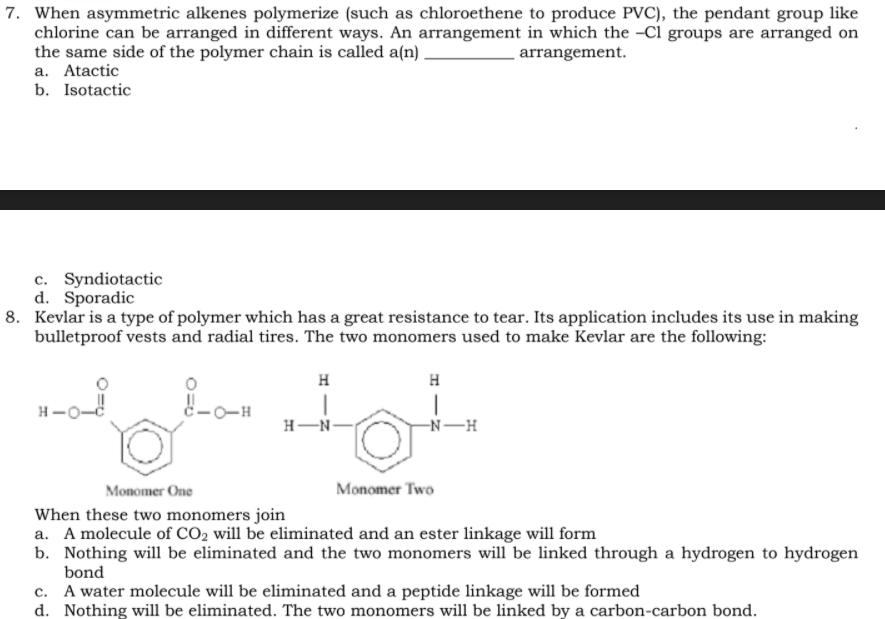
7. When asymmetric alkenes polymerize (such as chloroethene to produce PVC), the pendant group like chlorine can be arranged in different ways. An arrangement in which the -Cl groups are arranged on the same side of the polymer chain is called a(n) . a. Atactic arrangement. b. Isotactic c. Syndiotactic d. Sporadic
7. When asymmetric alkenes polymerize (such as chloroethene to produce PVC), the pendant group like chlorine can be arranged in different ways. An arrangement in which the -Cl groups are arranged on the same side of the polymer chain is called a(n) . a. Atactic arrangement. b. Isotactic c. Syndiotactic d. Sporadic
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter21: Organic And Biological Molecules
Section: Chapter Questions
Problem 10Q
Related questions
Concept explainers
Question
Transcribed Image Text:7. When asymmetric alkenes polymerize (such as chloroethene to produce PVC), the pendant group like
chlorine can be arranged in different ways. An arrangement in which the -Cl groups are arranged on
the same side of the polymer chain is called a(n) ,
a. Atactic
b. Isotactic
arrangement.
c. Syndiotactic
d. Sporadic
8. Kevlar is a type of polymer which has a great resistance to tear. Its application includes its use in making
bulletproof vests and radial tires. The two monomers used to make Kevlar are the following:
H
H
H-O-
c-0-H
H-N-
-N-H
Monomer One
Monomer Two
When these two monomers join
a. A molecule of CO2 will be eliminated and an ester linkage will form
b. Nothing will be eliminated and the two monomers will be linked through a hydrogen to hydrogen
bond
c. A water molecule will be eliminated and a peptide linkage will be formed
d. Nothing will be eliminated. The two monomers will be linked by a carbon-carbon bond.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning